
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g | |||
| Other Sizes |
Purity: ≥98%
Description: Vardenafil HCl Trihydrate (formerly also known as BAY 38-9456, trade names Levitra; Staxyn; Vivanza) is an approved medication acting as a phosphodiesterase/PDE inhibitor with important biological activity. It inhibits PDE5 and PDE1 with IC50s of 0.7 and 180 nM, respectively. Vardenafil has been approved used for treating erectile dysfunction. Vardenafil (VAR) is synthetic, highly selective, and potent inhibitor of phosphodiesterase-5 which competitively inhibits cyclic guanosine monophosphate (cGMP) hydrolysis and thus increases cGMP levels. It is clinically approved for treatment of erectile dysfunction in men, including diabetic and postprostatectomy patients.
| Targets |
PDE5 (IC50 = 0.7 nM); PDE6 (IC50 = 11 nM); PDE1 (IC50 = 180 nM); PDE3 (IC50 >1000 nM); PDE4 (IC50 >1000 nM)
Phosphodiesterase 5 (PDE5): Vardenafil HCl Trihydrate is a potent, selective PDE5 inhibitor. For recombinant human PDE5, it has an IC50 of 0.7 ± 0.1 nM (cGMP hydrolysis assay) and a Ki of 0.4 ± 0.05 nM (ligand binding assay) [1]. It shows moderate cross-reactivity with PDE6 (IC50 = 3.2 ± 0.3 nM) and minimal inhibition of other PDE subtypes (PDE1–PDE4, PDE7–PDE11) with IC50 > 100 nM [1,2] |
|---|---|
| ln Vitro |
Vardenafil hydrochloride trihydrate inhibits PDE5-mediated cGMP hydrolysis with an IC50 of 0.7 nM[1]. Vardenafil hydrochloride trihydrate raises intracellular cGMP levels in the penis' cavernosum tissue, causing sinus dilation and increased blood flow [3].
PDE Inhibition & Selectivity (Literature 1): Recombinant human PDE5 (0.5 μg/well) was incubated with Vardenafil HCl Trihydrate (0.1–5 nM) and 1 μM [³H]-cGMP. Concentration-dependent inhibition of cGMP hydrolysis was observed: 0.3 nM inhibited ~50% activity, 1 nM inhibited ~85%, 5 nM inhibited >95%. Testing against 10 other PDE subtypes confirmed high selectivity (e.g., PDE6 IC50 = 3.2 ± 0.3 nM, PDE11 IC50 = 150 ± 12 nM) [1] - Hepatoprotective Activity (Literature 5): Mouse hepatocytes (AML12 cells) were treated with lipopolysaccharide (LPS, 1 μg/mL, inflammation inducer) and Vardenafil HCl Trihydrate (1–20 μM) for 24 hours. MTT assay showed cell viability increased from 52% (LPS alone) to 88% (20 μM treatment). ELISA revealed TNF-α and IL-6 levels reduced by 60% and 55% (20 μM), respectively; Western blot confirmed NF-κB p65 phosphorylation reduced by 45% (20 μM) [5] - Cardiomyocyte Myofilament Function Improvement (Literature 6): Isolated left/right ventricular cardiomyocytes from diabetic rats (streptozotocin-induced) were treated with Vardenafil HCl Trihydrate (0.1–1 μM) for 1 hour. Myocyte shortening amplitude increased by 30% (left ventricle, 1 μM) and 25% (right ventricle, 1 μM); Western blot showed phospholamban (PLB) phosphorylation increased by 40% (left ventricle) [6] |
| ln Vivo |
In rats suffering from cavernous nerve damage, vardenafil hydrochloride trihydrate (IV; 0.03 mg/kg) shows facilitative effects[4]. Vardenafil hydrochloride trihydrate (IV; 0.17 mg/kg once daily; 7 days) reduces the expression of iNOS and NF-���B in hepatic tissue and shields the liver from Con A-induced hepatitis[5]. In ZDF hearts, vardenafil hydrochloride trihydrate (PO; 10 mg/kg once daily; 25 weeks) inhibits both the rise in 3-NT production and the decrease in tissue cGMP levels [6].
Rat Erectile Function Enhancement (Literature 1): Male Sprague-Dawley (SD) rats (300–350g) were divided into 3 groups (n=6/group): 1. Control: Saline; 2. Vardenafil HCl Trihydrate 0.3 mg/kg; 3. Vardenafil HCl Trihydrate 1 mg/kg. Drugs were administered via oral gavage 1 hour before electrical stimulation of the cavernous nerve. Results: - 0.3 mg/kg group: Erectile latency reduced from 85 ± 10 seconds (control) to 50 ± 8 seconds; - 1 mg/kg group: Erectile latency reduced to 35 ± 5 seconds, erectile duration increased from 40 ± 6 seconds (control) to 75 ± 8 seconds [1] - Cavernous Nerve Injury Rat Proerectile Effect (Literature 4): Male SD rats with bilateral cavernous nerve crush injury were divided into 4 groups (n=8/group): 1. Vehicle: 0.5% CMC-Na; 2. Vardenafil HCl Trihydrate 1 mg/kg; 3. BAY 60-4552 (sGC activator) 0.1 mg/kg; 4. Combination: Vardenafil HCl Trihydrate 1 mg/kg + BAY 60-4552 0.1 mg/kg. Drugs were administered intraperitoneally once daily for 14 days. Electrical stimulation-induced erectile rate: - Vehicle: 25%; - Vardenafil alone: 50%; - Combination: 85% [4] - Mouse Hepatitis Protection (Literature 5): Male C57BL/6 mice (8–10 weeks old) were divided into 3 groups (n=6/group): 1. Normal control: Saline; 2. Hepatitis control: LPS (10 mg/kg, i.p.) + vehicle; 3. Vardenafil treatment: LPS + Vardenafil HCl Trihydrate 10 mg/kg (oral, once daily for 7 days). On day 8, liver samples were collected: - Liver梗死 area reduced from 35% (hepatitis control) to 12% (treatment); - Serum ALT/AST levels reduced by 55% and 50%, respectively; - Hepatic MDA (oxidative stress marker) reduced by 45%, GSH increased by 60% [5] - Diabetic Rat Cardiac Function Improvement (Literature 6): Male SD rats with streptozotocin-induced diabetes (60 mg/kg, i.p.) were divided into 3 groups (n=8/group): 1. Normal control: Non-diabetic; 2. Diabetic control: Diabetic + vehicle; 3. Vardenafil treatment: Diabetic + Vardenafil HCl Trihydrate 0.5 mg/kg/day (oral, 12 weeks). Results: - Left ventricular ejection fraction (LVEF) increased from 45% (diabetic control) to 62% (treatment); - Right ventricular fractional shortening (RVFS) increased from 28% to 40%; - Cardiomyocyte contractile velocity increased by 35% (left ventricle) and 30% (right ventricle) [6] |
| Enzyme Assay |
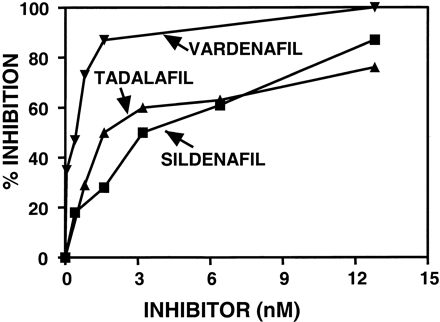
In this study, researchers investigated the potency and the selectivity profile of vardenafil on phosphodiesterase (PDEs) enzymes, its ability to modify cGMP metabolism and cause relaxation of penile smooth muscle and its effect on erections in vivo under conditions of exogenous nitric oxide (NO) stimulation. PDE isozymes were extracted and purified from human platelets (PDE5) or bovine sources (PDEs 1, 2, 3, 4 and 6). The inhibition of these PDEs and of human recombinant PDEs by vardenafil was determined. The ability to potentiate NO-mediated relaxation and influence cGMP levels in human corpus cavernosum strips was measured in vitro, and erection-inducing activity was demonstrated in conscious rabbits after oral administration together with intravenous doses of sodium nitroprusside (SNP). The effects of vardenafil were compared with those of the well-recognized PDE5 inhibitor, sildenafil (values for sildenafil in brackets). Vardenafil specifically inhibited the hydrolysis of cGMP by PDE5 with an IC50 of 0.7 nM (6.6 nM). In contrast, the IC50 of vardenafil for PDE1 was 180 nM; for PDE6, 11 nM; for PDE2, PDE3 and PDE4, more than 1000 nM. Relative to PDE5, the ratios of the IC50 for PDE1 were 257 (60), for PDE6 16 (7.4). Vardenafil significantly enhanced the SNP-induced relaxation of human trabecular smooth muscle at 3 nM (10 nM). Vardenafil also significantly potentiated both ACh-induced and transmural electrical stimulation-induced relaxation of trabecular smooth muscle. The minimum concentration of vardenafil that significantly potentiated SNP-induced cGMP accumulation was 3 nM (30 nM)[1].
Recombinant PDE5 Activity Assay (Literature 1): The assay was performed in 384-well plates with a 20 μL reaction volume. The mixture contained 50 mM Tris-HCl (pH 7.5), 10 mM MgCl₂, 2 mM DTT, 1 μM [³H]-cGMP (0.1 μCi), 0.5 μg recombinant human PDE5, and serial dilutions of Vardenafil HCl Trihydrate (0.1–5 nM). Incubated at 37°C for 30 minutes, then stopped with 5 μL 250 mM EDTA. Unhydrolyzed [³H]-cGMP was precipitated with 50 μL ZnSO₄/Ba(OH)₂ (0.2 M each), centrifuged (3000×g, 10 minutes). Supernatant radioactivity was measured via liquid scintillation counting. IC50 was calculated via nonlinear regression [1] - PDE5 Ligand Binding Assay (Literature 2): 200 μL reaction mixture (96-well plates) contained 50 mM Tris-HCl (pH 7.4), 10 mM MgCl₂, 0.5 μg human PDE5, 0.5 nM [³H]-vardenafil, and unlabeled Vardenafil HCl Trihydrate (0.05–10 nM). Incubated at 4°C for 2 hours, filtered through glass fiber filters (pre-soaked in 0.5% polyethyleneimine) to trap bound ligand. Filters were washed 3 times with ice-cold buffer, dried, and radioactivity measured. Ki was calculated using the Cheng-Prusoff equation [2] |
| Cell Assay |
AML12 Hepatocyte Protection Assay (Literature 5):
1. Cell Seeding: AML12 mouse hepatocytes were seeded in 96-well plates (5×10³ cells/well) or 6-well plates (2×10⁵ cells/well) and cultured in DMEM/F12 medium + 10% FBS overnight. 2. Treatment: Cells were pre-treated with Vardenafil HCl Trihydrate (1–20 μM) for 2 hours, then stimulated with LPS (1 μg/mL) for 24 hours. 3. Detection: - Viability: MTT assay (5 mg/mL, 4 hours incubation, DMSO dissolution, 570 nm absorbance); - Cytokines: Culture supernatant collected for TNF-α/IL-6 ELISA; - Oxidative stress: Cell lysate analyzed for MDA/GSH levels [5] - Diabetic Rat Cardiomyocyte Assay (Literature 6): 1. Cell Isolation: Left/right ventricular cardiomyocytes were isolated from diabetic rats via collagenase perfusion. 2. Treatment: Cardiomyocytes were incubated with Vardenafil HCl Trihydrate (0.1–1 μM) for 1 hour at 37°C. 3. Function Detection: Myocyte shortening/relaxation velocity measured via video-based edge detection; Western blot detected PLB phosphorylation (anti-p-PLB antibody, 1:1000 dilution) [6] |
| Animal Protocol |
Animal/Disease Models: Male rat (9weeks old) underwent surgery for laparotomy or bilateral cavernous nerve (CN) crush injury[4]
Doses: 0.03 mg/kg Route of Administration: intravenous (iv) injection Experimental Results: Restored normal erectile responses with a combined administration of BAY 60-4552 (0.03, 0.3 mg/kg). Animal/Disease Models: Liver injury induced by Con A in male Swiss albino mice (20 ± 2 g)[5] Doses: 0.17 mg/kg Route of Administration: intravenous (iv) injection; one time/day, for 7 days; as a pretreatment Experimental Results: decreased the levels of serum transaminases and alleviated Con A-induced hepatitis. Animal/Disease Models: Male 7weeks old Zucker diabetic fatty (ZDF) rats (preserved ejection fraction, HFpEF)[6] Doses: 10 mg/kg Route of Administration: po (oral gavage); one time/day, for 25 weeks Experimental Results: Improved myofilament function in diabetic rat hearts. Rat Erectile Function Assay (Literature 1): Male SD rats (300–350g, n=6/group) were anesthetized with pentobarbital (50 mg/kg, i.p.). Vardenafil HCl Trihydrate was dissolved in 0.5% CMC-Na to 0.03 mg/mL (0.3 mg/kg) and 0.1 mg/mL (1 mg/kg), administered via oral gavage. 1 hour later, the cavernous nerve was electrically stimulated (5 V, 20 Hz, 5 ms pulses, 30 seconds). Erectile latency (time to penis engorgement) and duration were recorded [1] - Cavernous Nerve Injury Rat Assay (Literature 4): Male SD rats (300–350g, n=8/group) underwent bilateral cavernous nerve crush (vascular clamp, 60 seconds). After 7 days, Vardenafil HCl Trihydrate (1 mg/kg, dissolved in 0.9% saline to 0.1 mg/mL) was administered intraperitoneally, alone or with BAY 60-4552 (0.1 mg/kg). Treatment lasted 14 days. On day 21, electrical stimulation (5 V, 20 Hz) was applied to the cavernous nerve, and erectile rate (percentage of rats with penile erection) was calculated [4] - Mouse Hepatitis Model (Literature 5): Male C57BL/6 mice (8–10 weeks old, n=6/group) received LPS (10 mg/kg, i.p.) to induce hepatitis. Vardenafil HCl Trihydrate (10 mg/kg, dissolved in 0.5% CMC-Na to 1 mg/mL) was administered orally once daily for 7 days, starting 1 day before LPS injection. On day 8, mice were euthanized; serum was collected for ALT/AST检测, liver tissue for梗死 area (TTC staining) and MDA/GSH analysis [5] - Diabetic Rat Cardiac Assay (Literature 6): Male SD rats (200–250g, n=8/group) received streptozotocin (60 mg/kg, i.p., dissolved in 0.1 M citrate buffer pH 4.5) to induce diabetes (blood glucose > 300 mg/dL after 72 hours). 4 weeks later, Vardenafil HCl Trihydrate (0.5 mg/kg/day, dissolved in 0.5% CMC-Na to 0.05 mg/mL) was administered orally for 12 weeks. On day 100, rats were euthanized; echocardiography measured LVEF/RVFS, cardiomyocytes isolated for contractile function analysis [6] |
| ADME/Pharmacokinetics |
Absorption
Within the recommended dose range, the pharmacokinetic characteristics of vardenafil are dose-proportional. In healthy male volunteers, after an oral administration of 20 mg vardenafil on an empty stomach, peak plasma drug concentrations were reached within 30 minutes to 2 hours (median 60 minutes), and 0.00018% of the drug was detectable in semen 1.5 hours post-administration. The bioavailability of vardenafil is approximately 15%. A high-fat diet can lead to a 18%–50% decrease in Cmax; however, AUC and Tmax remained unchanged. Elimination Route Vardenafil is primarily excreted as metabolites in feces and urine. Approximately 91–95% of the orally administered dose is excreted in feces, and 2–6% in urine. Volume of Distribution The steady-state volume of distribution of vardenafil is 208 L. Clearance The systemic clearance of vardenafil is 56 L/h. Protein binding: Extremely high: 95% bound to plasma proteins; reversible and independent of total drug concentration. Rapid absorption; absolute bioavailability is approximately 15%. In healthy volunteers, after an oral administration of 20 mg on an empty stomach, peak plasma concentrations are typically reached within 30 minutes to 2 hours (median 60 minutes). High-fat meals reduce peak plasma concentration (Cmax) by 18% to 50%. In rabbits, the enhanced erectile function of 0.1 mg/kg vardenafil, induced by nitric oxide (NO), is limited by its pharmacokinetic properties (Tmax = 1 hour; T1/2 = 1.2 hours), although an erectile effect was still observed after 7 hours. In humans, vardenafil is rapidly absorbed (Tmax approximately 40 minutes) and slowly metabolized (T1/2 approximately 4 hours), with an absolute bioavailability of 14.5% (compared to 40% for sildenafil). While high-fat meals do not affect the relative bioavailability of the drug, they do delay intestinal absorption. Concomitant use with CYP3A4 inhibitors such as ritonavir can affect hepatic metabolism. Vardenafil's active metabolite, M1, has a PDE5 inhibitory activity that is four times lower than that of the parent compound, accounting for approximately 7% of the total therapeutic effect of vardenafil. PMID: 15224134 Time to peak concentration: 30 minutes to 2 hours (oral, fasting) Metabolism/Metabolites Vardenafil is primarily metabolized in the liver via CYP3A4, but CYP3A5 and CYP2C isoenzymes also participate in its metabolism. The main circulating metabolite, M1 (N-deethylvardenafil), is a product of the partial deethylation of vardenafil piperazine, with plasma concentrations approximately 26% of the parent compound. M1 has similar phosphodiesterase selectivity to vardenafil, and its in vitro PDE5 inhibitory potency is approximately 28% of that of vardenafil. M1 is primarily metabolized by hepatic CYP3A4, with CYP3A5 and CYP2C isoenzymes also contributing. M1 is the product of the partial deethylation of vardenafil piperazine and is also the major circulating metabolite. M1 is further metabolized. The plasma concentration of M1 is approximately 26% of the parent compound and accounts for 7% of the total pharmacological activity. This metabolite exhibits phosphodiesterase selectivity similar to that of vardenafil, with an in vitro PDE5 inhibitory potency of 28% that of vardenafil. Biological Half-Life The terminal half-life of vardenafil and its major metabolite (M1) is 4-5 hours. Oral absorption (Reference 2): In healthy male volunteers (n=12), oral administration of 20 mg vardenafil hydrochloride trihydrate showed: -Peak plasma concentration (Cmax) = 18 ± 3 ng/mL, time to peak concentration (Tmax) = 0.9 ± 0.2 h; -Oral bioavailability (F) = 25 ± 3% (AUC0–∞ = 85 ± 10 ng·h/mL compared with intravenous 5 mg); -Food effect: High-fat meal reduced Cmax by 30% and delayed Tmax by 1 hour [2] -Distribution and elimination (Reference 2): -Volume of distribution (Vd) = 20 ± 3 L; -Plasma protein binding rate = 95 ± 2% (ultrafiltration, 1–100 ng/mL); -Elimination half-life (t1/2) = 4.8 ± 0.3 hours; - Metabolism: Mainly metabolized by CYP3A4 in the liver, the main metabolite is M1 (active, accounting for about 20% of the parent drug activity)[2] - Excretion (Reference 2): After oral administration of [¹⁴C]-vardenafil, 91% of the radioactivity is excreted within 72 hours: 77% is excreted in feces (accounting for 3% of the parent drug), and 14% is excreted in urine (accounting for <1% of the parent drug)[2] |
| Toxicity/Toxicokinetics |
Hepatotoxicity
Despite the widespread use of vardenafil, no clinically significant liver injury cases have been observed, and elevated serum enzymes during treatment are rare. The related PDE5 inhibitors sildenafil and tadalafil have been associated with isolated, rare cases of acute liver injury and jaundice. The incubation period ranges from a few days to 3 months, and the injury pattern is typically cholestatic. No autoimmune or immunohypersensitivity features have been observed; all cases are self-limiting, without sequelae or acute liver failure. Whether vardenafil can cause similar acute liver injury is unclear. Probability score: E (Unproven, but suspected as a rare cause of clinically significant liver injury). Interactions The combined use of vardenafil with other drugs for treating erectile dysfunction has not been studied; combined use with drugs for treating erectile dysfunction is not recommended. α-blockers, such as terazosin, tamsulosin, doxazosin, prazosin, alfuzosin: Contraindicated; concomitant use may cause hypotension. Erythromycin can increase the AUC of vardenafil by 4-fold and the Cmax by 3-fold (in healthy volunteers). Protein Binding Approximately 95% of vardenafil and its major circulating metabolites are bound to plasma proteins. Their protein binding is reversible and independent of total drug concentration. In vitro cytotoxicity (References 5, 6): -After treatment of AML12 hepatocytes with vardenafil trihydrate (1–20 μM) for 24 hours, cell viability >90% (MTT method) [5] -After treatment of isolated cardiomyocytes with 0.1–1 μM for 1 hour, no significant cell death was observed (trypan blue exclusion method, cell viability >95%) [6] -In vivo safety (References 1, 2, 5, 6): -Erectile function study in rats (0.3–1 mg/kg, oral, single dose): No death, weight loss or abnormal behavior; normal serum BUN/creatinine [1] -Hepatitis study in mice (10 mg/kg, oral, 7 days): No inflammation/necrosis was observed in liver/kidney histology; no change in serum urea nitrogen/creatinine levels [5] -Diabetes rat study (0.5 mg/kg, oral, 7 days): mg/kg/day, orally, for 12 weeks): No increase in liver and kidney weight; serum alanine aminotransferase/aspartate aminotransferase/urea nitrogen levels were within the normal range [6] - Drug interactions (References 2, 3): - Co-administration with CYP3A4 inhibitors (e.g., ketoconazole) increased the AUC of vardenafil hydrochloride trihydrate by 8-fold; co-administration with CYP3A4 inducers (e.g., rifampin) decreased the AUC by 70% [2] - No significant interaction with nitrates (but as described in Reference 3, co-administration with nitrates is clinically contraindicated due to the possibility of hypotension) [3] |
| References | |
| Additional Infomation |
Vardenafil hydrochloride trihydrate is the hydrochloride form of vardenafil, a benzenesulfonamide derivative and phosphodiesterase type 5 (PDE5) inhibitor with vasodilatory activity. Vardenafil selectively inhibits PDE5, thereby inhibiting the degradation of cyclic guanosine monophosphate (cGMP) in the smooth muscle of the corpus cavernosum and corpus spongiosum of the penis. Inhibition of cGMP degradation leads to prolonged muscle relaxation, vasodilation, and engorgement of the corpus cavernosum, thus prolonging penile erection time. This product is a piperazine derivative, a phosphodiesterase type 5 inhibitor and vasodilator, used as a urological medication for the treatment of erectile dysfunction. See also: Vardenafil (note moved to). Mechanism of action: 1. PDE5 inhibition: Vardenafil hydrochloride trihydrate competitively binds to the catalytic site of PDE5, inhibiting cGMP hydrolysis and increasing intracellular cGMP levels. cGMP activates PKG, mediating smooth muscle relaxation (e.g., corpus cavernosum, blood vessels) [1,4]
2. Hepatoprotective effect: Inhibits NF-κB activation and reduces oxidative stress (MDA decreases, GSH increases), thereby alleviating LPS-induced liver damage [5] 3. Cardioprotective effect: Improves myofibril function of cardiomyocytes through PLB phosphorylation, enhancing the Ca²⁺ processing capacity of diabetic hearts [6] - Therapeutic potential: 1. Erectile dysfunction (ED): Effective in ED models (normal and nerve-damaged rats), with faster onset (Tmax approximately 1 hour), superior to tadalafil [1,3,4] 2. Hepatoprotective effect: Reduces the severity of LPS-induced hepatitis, suggesting its potential use in the treatment of inflammatory liver disease [5] 3. Hepatoprotective effect: Improves ventricular function in diabetic patients, reducing the potential risk of diabetic cardiomyopathy in rats [6] - Clinical comparison (Reference 3): Compared with sildenafil/tadalafil: vardenafil hydrochloride trihydrate has higher selectivity for PDE5 than sildenafil, a shorter half-life (t1/2) (4.8 hours), while tadalafil has a half-life of 17.5 hours and lower visual side effects (due to weaker PDE6 inhibition) [3] |
| Molecular Formula |
C23H32N6O4S.HCL.3H2O
|
|---|---|
| Molecular Weight |
579.11
|
| Exact Mass |
578.229
|
| Elemental Analysis |
C, 44.88; H, 6.55; Cl, 11.52; N, 13.65; O, 18.19; S, 5.21
|
| CAS # |
330808-88-3
|
| Related CAS # |
Vardenafil hydrochloride;224785-91-5;Vardenafil dihydrochloride;224789-15-5
|
| PubChem CID |
135413545
|
| Appearance |
White to light yellow solid powder
|
| LogP |
3.636
|
| Hydrogen Bond Donor Count |
5
|
| Hydrogen Bond Acceptor Count |
11
|
| Rotatable Bond Count |
8
|
| Heavy Atom Count |
38
|
| Complexity |
854
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
O=C1N=C(C2=CC(S(=O)(N3CCN(CC)CC3)=O)=CC=C2OCC)NN4C1=C(C)N=C4CCC.[H]Cl.[H]O[H].[H]O[H].[H]O[H]
|
| InChi Key |
NEAUGLIJDBPHAY-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C23H32N6O4S.ClH.3H2O/c1-5-8-20-24-16(4)21-23(30)25-22(26-29(20)21)18-15-17(9-10-19(18)33-7-3)34(31,32)28-13-11-27(6-2)12-14-28;;;;/h9-10,15H,5-8,11-14H2,1-4H3,(H,25,26,30);1H;3*1H2
|
| Chemical Name |
2-(2-ethoxy-5-((4-ethylpiperazin-1-yl)sulfonyl)phenyl)-5-methyl-7-propylimidazo[5,1-f][1,2,4]triazin-4(1H)-one dihydrochloride trihydrate
|
| Synonyms |
BAY38-9456; BAY 38-9456; 224785-91-5; Vardenafil HCL; Vardenafil (hydrochloride); Vardenafilhydrochloride; Vardenafil, Hydrochloride Salt; Vardenafil hydrochloride [USAN]; VARDENAFIL MONOHYDROCHLORIDE;BAY-38-9456; trade names: Levitra; Staxyn; Vivanza; Vardenafil hydrochloride trihydrate; Vardenafil HCl; Levitra; Staxyn; Vivanza
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7268 mL | 8.6339 mL | 17.2679 mL | |
| 5 mM | 0.3454 mL | 1.7268 mL | 3.4536 mL | |
| 10 mM | 0.1727 mL | 0.8634 mL | 1.7268 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
|
|---|
|
|