
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg | |||
| 250mg | |||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
SB408124 HCl is a potent, novel, selective, non-peptide antagonist for OX1 receptor with Ki of 57 nM and 27 nM in both whole cell and membrane, respectively, it exhibits 50-fold selectivity over OX2 receptor. In primary astrocyte cultures from the rat cerebral cortex, pretreatment with SB408124 significantly reduced the orexin A a stimulating effect on basal-induced cAMP production and forskolin.
| Targets |
OX1 Receptor ( Ki = 57 nM ); OX1 Receptor ( Ki = 27 nM )
|
||
|---|---|---|---|
| ln Vitro |
|
||
| ln Vivo |
|
||
| Enzyme Assay |
SB-408124 is a non-peptide antagonist that shows 50-fold selectivity over OX2 receptor and has a Ki of 57 nM and 27 nM in whole cell and membrane, respectively, for the OX1 receptor.
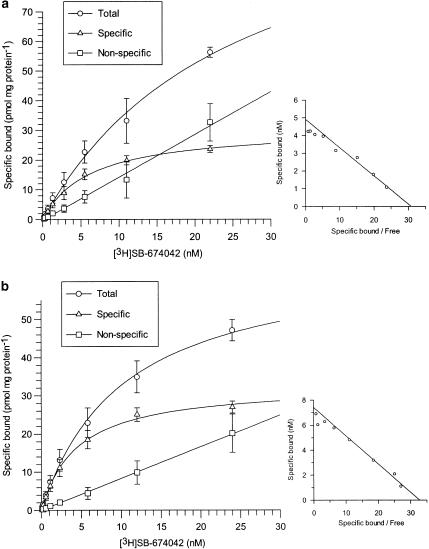
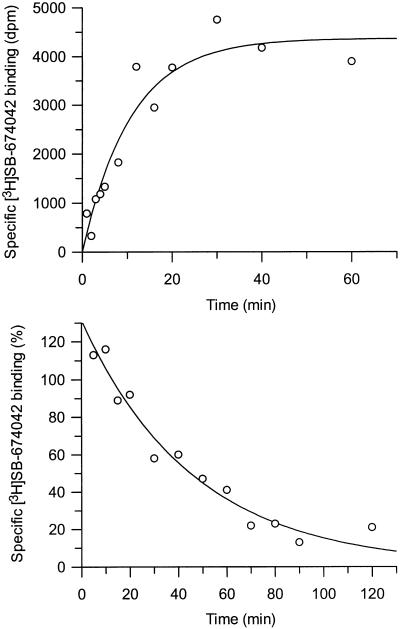
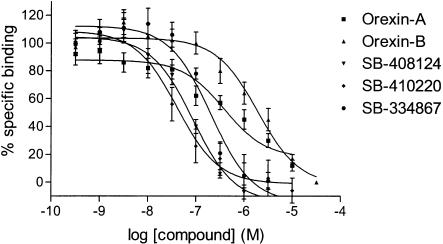
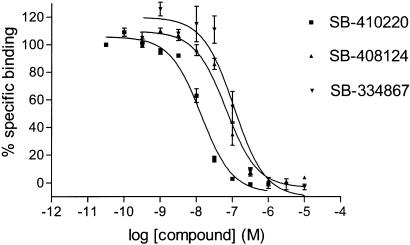
[3H]SB-674042 whole cell binding assays[1] After overnight culture in 96-well Packard Cultur plates, the medium was discarded and cells were incubated in buffer containing 150 mM NaCl, 20 mM HEPES and 0.5% bovine serum albumin (pH 7.4) for 60 min at 25°C. Saturation studies were carried out by incubating cells with a range of concentrations of [3H]SB-674042 (0.2–24 nM); the total assay volume was 250 μl. Protein content was assayed by lysing cells with 0.1 M NaOH and using the Bradford method (Bradford, 1976) with bovine serum albumin (BSA) as a standard. Association kinetic studies were performed by measuring the specific binding of [3H]SB-674042 (3 nM) at 1–60 min after addition of [3H]SB-674042. For dissociation studies, cells were first incubated with [3H]SB-674042 (3 nM) for 60 min. Specific binding was then measured at 2–120 min after the addition of 3 μM SB-408124. Competition studies were performed by incubating cells with [3H]SB-674042 (3 nM) and a range of concentrations of the test compound. All assays were terminated by washing the cells three times with 250 μl ice-cold phosphate-buffered saline. A volume of 100 μl of Microscint 40 was added to each well and the plate was left at room temperature for 2 h. Cell-associated radioactivity was then measured using a Packard Topcount, with a count time of 2 min well−1. [3H]SB-674042 membrane-based SPA binding assays[1] CHO-K1_OX1 cell membranes (75 μg ml−1) were precoupled by shaking with wheatgerm-agglutinin polyvinyltoluene (WGA-PVT) scintillation proximity assay (SPA) beads (5 mg ml−1) in buffer containing 25 mM HEPES, 2.5 mM MgCl2, 0.5 mM EDTA and 0.025% bacitracin (pH 7.4) at 4°C for 1 h. The bead-membrane suspension was centrifuged at 300 × g and resuspended in the same volume of room temperature assay buffer. A volume of 100 μl of bead-membrane suspension was incubated with [3H]SB-674042 (5 nM) in a total assay volume of 200 μl in a 96-well Packard Optiplate to give a final protein concentration of 7.5 μg well−1. Nonspecific binding was measured as that remaining in the presence of 3 μM SB-408124. Assay plates were shaken for 10 min and then incubated at room temperature for 4 h before being counted on a Packard TopCount scintillation counter (count time 2 min well−1). Saturation studies were carried out by incubating bead-membranes (equivalent to 7.5 μg protein well−1 and 2.5 mg beads ml−1) with a range of concentrations of [3H]SB-674042 (0.1–20 nM). Protein content was assayed using the Bradford method (Bradford, 1976) using bovine serum albumin as a standard. Association kinetic studies were performed by measuring specific binding of [3H]SB-674042 (5 nM) at 1–30 min after addition of bead-membranes (equivalent to 7.5 μg protein well−1 and 2.5 mg beads ml−1). For dissociation studies, bead-membranes were first incubated with [3H]SB-674042 (5 nM) for 30 min. Specific binding was then measured at 2–120 min after the addition of 3 μM SB-408124. Competition studies were performed by incubating bead-membranes (equivalent to 7.5 μg protein well−1 and 2.5 mg beads ml−1) with [3H]SB-674042 (5 nM) and a range of concentrations of the test compound. |
||
| Cell Assay |
SB-408124 has a pKi of 7.57 when it comes to binding the hypocretin type 1 receptor (HcrtR1). According to studies on calcium mobilization, SB-408124 functions as a functional antagonist of the OX1 receptor and has an affinity that is roughly 50 times more selective than the OX2 receptor. According to a recent study, the stimulatory action of Orexin A on basal and forskolin-acivated cAMP production was significantly reduced when primary cultures of rat astrocytes were pretreated with SB-401824 prior to Orexin A administration.
Measurement of orexin A-induced AVP mRNA expression in cultured brain neurons.[3] Primary neuronal cultures from the hypothalamus were incubated with vehicle control or differing concentrations of orexin A (10 nM, 100 nM, 1 μM, or 10 μM) in DMEM-HSPS for 6 h. The culture medium was removed, and cells were washed with cold PBS, collected, and subjected to RNA isolation. Real-time PCR was performed to measure mRNA levels of AVP. To test which orexin receptor mediates the AVP increase induced by orexin, we coincubated neuronal cultures with 1 μM orexin A with or without the OX1R antagonist SB-408124 (100 µM) or OX2Ra TCS-OX2-29 (100 µM) for 6 h. The mRNA level of AVP was determined by real-time quantitative PCR. Each set of experiments was performed using 3 culture wells in a 24-well plate, and all cDNA samples were assayed in duplicate. The whole experiment was repeated two to three times. Data were normalized to GAPDH mRNA. |
||
| Animal Protocol |
|
||
| References |
|
||
| Additional Infomation |
The orexin system is involved in the regulation of arginine vasopressin (AVP), and its overactivation is associated with hypertension. However, its role in salt-sensitive hypertension (SSHTN) remains unclear. This study aimed to investigate whether overactivation of the paraventricular nucleus (PVN) orexin system leads to SSHTN by enhancing AVP signaling. Eight-week-old male Dahl salt-sensitive (Dahl S) rats and age- and sex-matched Sprague-Dawley (SD) rats were fed high-salt (HS; 8% NaCl) or normal-salt (NS; 0.4% NaCl) diets for four weeks, respectively. Results showed that the high-salt diet did not alter mean arterial pressure (MAP), or the mRNA levels of orexin receptor 1 (OX1R) or OX2R in the PVN, but slightly increased AVP mRNA expression in the PVN of SD rats. The high-salt diet significantly increased the mRNA levels of OX1R, OX2R, and AVP in both mean arterial pressure (MAP) and the paraventricular nucleus (PVN) of Dahl S rats. Intraventricular injection of orexin A (0.2 nmol) significantly increased the mRNA level and immunoreactivity of avascular vasopressin (AVP) in the paraventricular nucleus (PVN) of SD rats. Incubation of cultured hypothalamic neurons from neonatal SD rats with orexin A increased AVP mRNA expression, while this increase was attenuated by OX1R blockers. Furthermore, intraventricular injection of sodium chloride solution (4 µmol) to increase cerebrospinal fluid sodium ion concentration increased the mRNA levels of OX1R and AVP in the PVN of SD rats, as well as immunoreactivity. In addition, microinjection of the OX1R antagonist SB-408124 into the bilateral paraventricular nucleus (PVN) significantly reduced the mean arterial pressure (MAP) in the high-salt intake group (-16 ± 5 mmHg) compared to the normal-salt intake group (-4 ± 4 mmHg). These results suggest that enhanced PVN OX1R activation may lead to salt-sensitive hypertension (SSHTN) by enhancing angiotensin (AVP) signaling. New findings and significance: To our knowledge, this study is the first to investigate the role of the orexin system in salt-sensitive hypertension. Our results suggest that the orexin system may participate in the Dahl salt-sensitive hypertension model by enhancing vasopressin signaling in the paraventricular nucleus of the hypothalamus. [3]
1. This study characterized the binding of a novel non-peptide antagonist radioligand [(3)H]SB-674042 (1-(5-(2-fluorophenyl)-2-methylthiazolyl-4-yl)-1-((S)-2-(5-phenyl-(1,3,4)oxadiazol-2-ylmethyl)-pyrrolidine-1-yl)-methyl ketone) to the human orexin-1 (OX(1)) receptor stably expressed in Chinese hamster ovary (CHO) cells. Two methods were used: whole-cell assay and cell membrane-based scintillation proximity assay (SPA). 2. The specific binding of [(3)H]SB-674042 was saturated in both whole-cell and cell membrane assays. Analysis revealed the existence of a high-affinity binding site, with Kd values of 3.76±0.45 nM and 5.03±0.31 nM in the whole-cell and membrane-bound states, respectively, and corresponding Bmax values of 30.8±1.8 pmol/mg protein and 34.4±2.0 pmol/mg protein, respectively. Kinetic studies also yielded similar Kd values. 3. Whole-cell competition experiments showed that natural orexin peptides have low affinity for the OX1 receptor, with orexin A having an affinity approximately five times that of orexin B (Ki values of 318±158 nM and 1516±597 nM, respectively). 4. SB-334867, SB-408124 (1-(6,8-difluoro-2-methyl-quinoline-4-yl)-3-(4-dimethylaminophenyl)-urea) and SB-410220 (1-(5,8-difluoro-quinoline-4-yl)-3-(4-dimethylaminophenyl)-urea) all showed high affinity for the OX(1) receptor in whole-cell (K(i) values of 99±18, 57±8.3 and 19±4.5 nm, respectively) and membrane (K(i) values of 38±3.6, 27±4.1 and 4.5±0.2 nm, respectively). 5. Calcium mobilization studies have shown that SB-334867, SB-408124, and SB-410220 are all functional antagonists of the OX(1) receptor, with efficacy consistent with their affinity (determined by radioligand binding assays), and a selectivity of approximately 50-fold for orexin-2 receptors. 6. These studies have shown that [(3)H]SB-674042 is a specific, high-affinity radioligand for the OX(1) receptor. The availability of this radioligand will be a valuable tool for studying the physiological function of the OX(1) receptor. [1] |
| Molecular Formula |
C19H19CLF2N4O
|
|
|---|---|---|
| Molecular Weight |
392.83
|
|
| Exact Mass |
392.121
|
|
| CAS # |
1431697-90-3
|
|
| Related CAS # |
SB-408124; 288150-92-5
|
|
| PubChem CID |
71576692
|
|
| Appearance |
White to gray solid powder
|
|
| Hydrogen Bond Donor Count |
3
|
|
| Hydrogen Bond Acceptor Count |
5
|
|
| Rotatable Bond Count |
3
|
|
| Heavy Atom Count |
27
|
|
| Complexity |
484
|
|
| Defined Atom Stereocenter Count |
0
|
|
| InChi Key |
DIHXPSGMLOETTI-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C19H18F2N4O.ClH/c1-11-8-17(15-9-12(20)10-16(21)18(15)22-11)24-19(26)23-13-4-6-14(7-5-13)25(2)3;/h4-10H,1-3H3,(H2,22,23,24,26);1H
|
|
| Chemical Name |
1-(6,8-difluoro-2-methylquinolin-4-yl)-3-[4-(dimethylamino)phenyl]urea;hydrochloride
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5456 mL | 12.7282 mL | 25.4563 mL | |
| 5 mM | 0.5091 mL | 2.5456 mL | 5.0913 mL | |
| 10 mM | 0.2546 mL | 1.2728 mL | 2.5456 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
 |
|---|
 |
  |