
| Size | Price | Stock | Qty |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
Ropinirole (also known as AS-041164; SKF101468; Requip) a potent and selective dopamine D2 receptors agonist (Ki = 29 nM) of the non-ergoline class of medications with anti-PD effects. Ropinirole is mainly used for Parkinson's disease, RLS-restless legs syndrome and extrapyramidal symptoms. Additionally, it can lessen the side effects of selective serotonin reuptake inhibitors (SSRIs) and antipsychotics, such as Parkinsonism syndrome, erectile dysfunction, and sexual dysfunction. In the Fe2+–H2O2 reaction system, ropinirole scavenges free radicals and inhibits lipid peroxidation.
| Targets |
D2 Receptor ( Ki = 29 nM ); hD2 Receptor ( pEC50 = 7.4 ); hD3 Receptor ( pEC50 = 8.4 ); hD4.4 Receptor ( pEC50 = 6.8 )
|
|
|---|---|---|
| ln Vitro |
|
|
| ln Vivo |
|
|
| Animal Protocol |
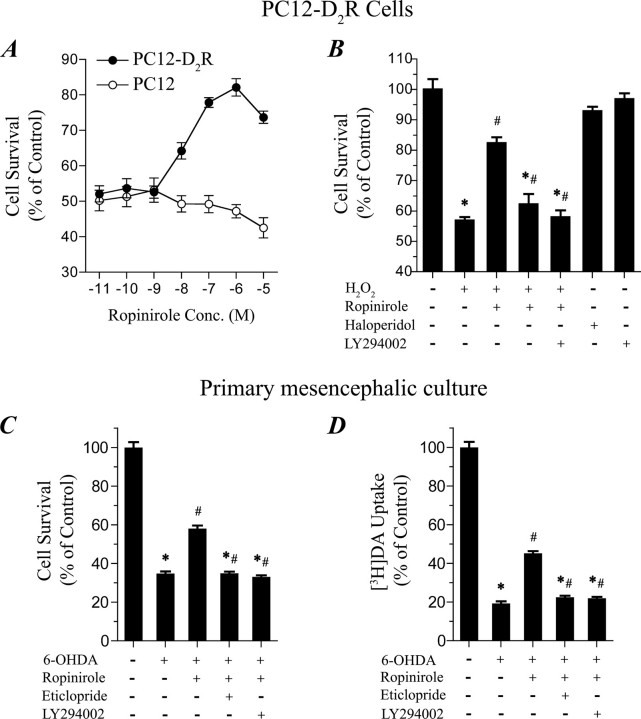
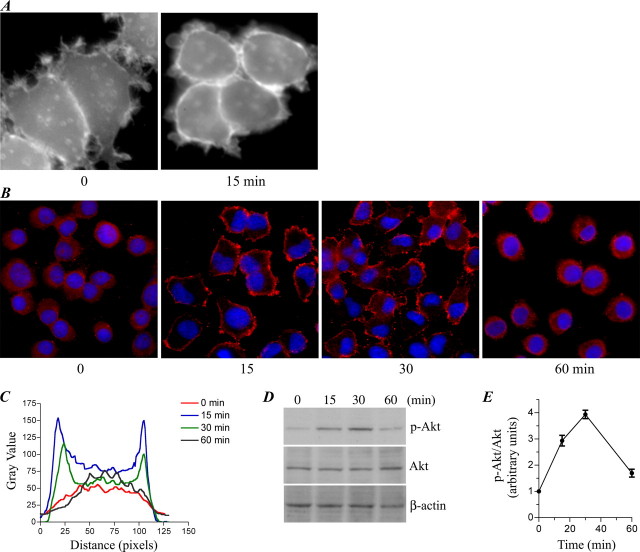
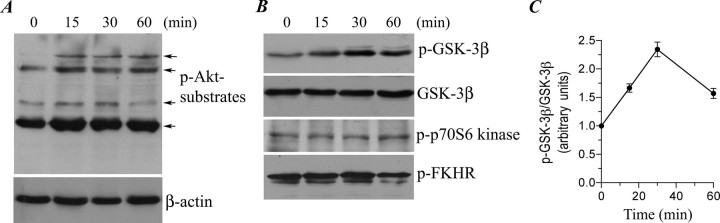
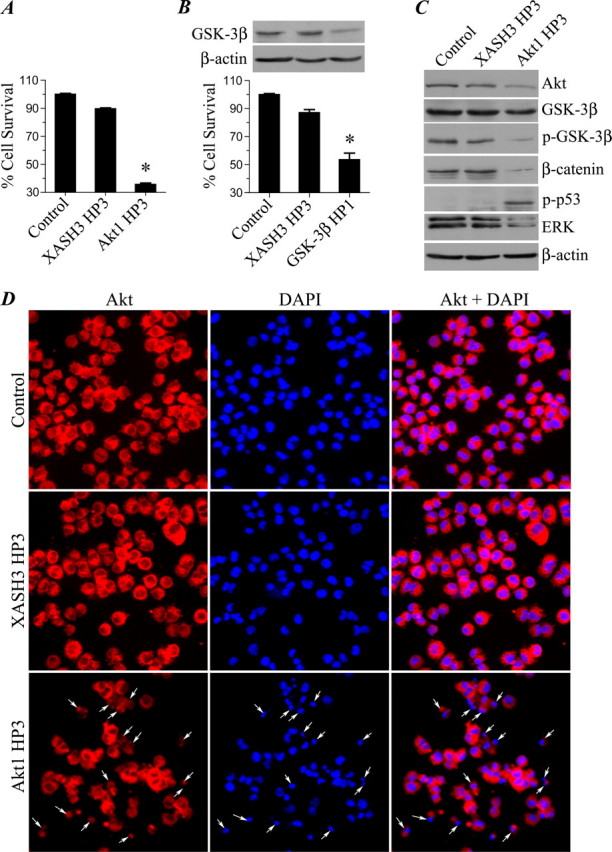
For the Irwin primary screen in mice, male mice (27-31 g) received Ropinirole (SK&F 101468-A) at doses of 1.0, 10.0, and 100 mg/kg PO. Behavioral parameters were observed continuously for 30 minutes and then at 1 and 2 hours after dosing. Body temperature was measured using a rectal thermometer at each observation period. [1]
For the spontaneous locomotor activity (SLA) test in mice, male mice (25-35 g) were placed in Perspex cages with two photocells. Locomotor activity was recorded for 30 minutes after IP dosing of ropinirole (1-100 mg/kg) and expressed as a cumulative count. [1] For the stereotypic behavior test in rats and mice, animals were placed individually in Perspex cages after treatment with ropinirole (1-100 mg/kg IP for mice). The degree of stereotypy was assessed at 5-minute intervals for 30 minutes using a scoring system (0-4), and effects were followed for up to 2.5 hours. [1] For the intrastriatal administration study in rats, under chloral hydrate anesthesia, chronic bilateral cannulae were implanted into the center of the caudate-putamen. After a 14-21 day recovery, ropinirole (1 or 10 μg) was injected unilaterally into one striatum and vehicle into the other in a volume of 1 μl over 60 seconds. Asymmetric behavior was assessed at 15-minute intervals using a scoring system (0-3). [1] For the unilateral 6-OHDA-lesioned mouse model, male mice (30-35 g) received a unilateral administration of 6-OHDA (8 μg/0.5 μl) into the substantia nigra. After 7-25 days, ropinirole was administered acutely IP over the dose range 0.001-100 mg/kg, or chronically for 14 days at 1.0 mg/kg IP, b.i.d. Asymmetry was assessed using a scoring system (0-3) and circling was measured in revolutions per minute. [1] For the MPTP-lesioned marmoset model, marmosets received a unilateral, 13-day infusion of MPTP (20 μg/24 hours) into the zona compacta of the substantia nigra via an Alzet osmotic minipump. Drug effects commenced 7-10 days after the start of the infusion. Ropinirole (SK&F 101468-A) was administered SC (0.01-1.0 mg/kg), PO by gastric gavage (0.1-1.0 mg/kg), or chronically PO b.i.d. (0.5 mg/kg for 4 days). Behavioral parameters measured included spontaneous locomotor activity (recorded for two 2-minute test periods), head movement, jumps, and a behavioral scoring system (0-6) for parameters like speed of movement, interest in novel stimuli, and facial expression. [1] |
|
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Ropinirome is rapidly absorbed after oral administration, reaching peak plasma concentration in approximately 1 to 2 hours. Absolute bioavailability is 45% to 55%, suggesting a first-pass effect of approximately 50%. Compared to immediate-release tablets, the bioavailability of extended-release ropinirome tablets is approximately 100%. Food intake does not affect the absorption of ropinirome, but when taken with a high-fat meal, the time to peak concentration (Tmax) is prolonged by 2.5 hours, and the peak plasma concentration (Cmax) is reduced by approximately 25%. Most of the absorbed dose is cleared by the liver. In clinical trials, over 88% of the radiolabeled dose was recovered in the urine. Less than 10% of the administered dose is excreted unchanged in the urine. The main metabolite in urine is despropylropinirome (40%), followed by carboxylic acid metabolites (10%) and glucuronide of the hydroxyl metabolite (10%). Ropinirome is widely distributed throughout the body, with an apparent volume of distribution of 7.5 L/kg. The clearance of ropinirole via oral administration was 47 L/h. Ropinirole is a novel compound used to treat the symptoms of Parkinson's disease. This study investigated the in vivo distribution and metabolism of ropinirole hydrochloride in mice, rats, cynomolgus monkeys, and humans via oral and intravenous administration. In all animals, almost all doses (94%) following oral administration were rapidly absorbed from the gastrointestinal tract (14C-ropinirole hydrochloride). In rats and monkeys, the compound rapidly distributed outside the body fluids and was shown to cross the blood-brain barrier. The compound exhibited high hemolytic clearance, approximately half that of hepatic blood flow in monkeys and similar to hepatic blood flow in rats. The terminal elimination half-life of the compound was relatively short (0.5 h in rats and 1.3 h in monkeys), although there is evidence of a second elimination phase in monkeys, with an elimination half-life of approximately 5–11 h. In mice, plasma concentrations of ropinirole following intravenous injection were not determined; however, at the study dose described herein, plasma concentrations of ropinirole in mice were below the limit of quantification (0.08 ng/mL). Ropinirole is extensively metabolized in both animals and humans. In rats, the primary metabolic pathway is hydroxylation of the aromatic ring to 7-hydroxyropinirole. In mice, monkeys, and humans, the primary metabolic pathway is N-depropylation. The N-depropylate metabolite is further metabolized to 7-hydroxy and carboxylic acid derivatives. In all species, the generated metabolites are generally further metabolized via glucuronidation. 7-Hydroxyropinirole is the only ropinirole metabolite previously shown to have significant in vivo dopamine agonist activity. In all species, the primary route of excretion of ropinirole-related substances following oral or intravenous administration is the kidneys (60–90% of the dose). Ropinirole is a selective non-ergot dopamine D2 receptor agonist indicated for the treatment of Parkinson's disease. After oral administration, ropinirole is rapidly and almost completely absorbed and widely distributed throughout the bloodstream. Its bioavailability is approximately 50%. Ropinirole has low plasma protein binding. The drug is inactivated after hepatic metabolism, and its major circulating metabolites are pharmacologically inactive. The main metabolic enzyme is the cytochrome P450 (CYP) isoenzyme CYP1A2. The pharmacokinetics of ropinirole are approximately linear with single or repeated administration, with a half-life of approximately 6 hours. Population pharmacokinetic studies have shown that sex, mild or moderate renal impairment, Parkinson's disease stage, comorbidities, or several common concomitant medications do not affect the pharmacokinetics of ropinirole. Clearance is slower in patients over 65 years of age compared to younger patients; clearance is also slower in women receiving hormone replacement therapy compared to women not receiving hormone replacement therapy. When the CYP1A2 inhibitor ciprofloxacin is co-administered with ropinirole, the plasma concentration of ropinirole increases, but no interaction is observed when co-administered with theophylline (which, like ropinirole, is a CYP1A2 substrate). The plasma concentration-effect relationship of ropinirole is not significant. Ropinirole is widely distributed throughout the body, with an apparent volume of distribution of 7.5 L/kg. Its plasma protein binding rate is as high as 40%, and the ratio of plasma concentration to blood concentration is 1:1. After oral administration, ropinirole is rapidly absorbed, reaching peak plasma concentration in approximately 1–2 hours. In clinical trials, over 88% of the radiolabeled dose was recovered in the urine, with an absolute bioavailability of 45%–55%, indicating a first-pass effect of approximately 50%. The relative bioavailability of tablets compared to oral solutions is 85%. Food does not affect the absorption of ropinirole, but when taken with a high-fat meal, its time to peak concentration (Tmax) is prolonged by 2.5 hours, and its peak concentration (Cmax) is reduced by approximately 25%. For more complete data on the absorption, distribution, and excretion of ropinirole (6 items), please visit the HSDB record page. Metabolism/Metabolites ropinirole is primarily metabolized in the liver. The most important metabolic pathways are N-depropylation and hydroxylation, producing N-depropyl and hydroxy metabolites, both of which are inactive. The N-depropyl metabolite is subsequently converted to carbamoyl glucuronide, carboxylic acid, and N-depropyl hydroxy metabolite. Following this process, the hydroxy metabolite of ropinirole undergoes rapid glucuronidation. In vitro studies have shown that the main cytochrome P450 enzyme involved in ropinirole metabolism is CYP1A2. Ropinirole is primarily metabolized in the liver. The N-depropyl metabolite is the main circulating metabolite in plasma. Based on AUC data, the plasma concentration of this metabolite was consistently higher than that of the parent drug, suggesting that the conversion of ropinirole to the N-depropyl metabolite is unsaturated. The N-depropyl metabolite has a lower affinity for the human clonal D2 receptor than ropinirole. Furthermore, this metabolite cannot cross the blood-brain barrier; therefore, it is unlikely to contribute to the therapeutic effect of ropinirole. The plasma concentration of the hydroxylated metabolite was low, approximately 1-5% of the ropinirole concentration. Although the hydroxylated metabolite showed higher activity than ropinirole in in vitro D2 receptor binding studies, it is not expected to contribute to the activity of ropinirole at therapeutic doses. Ropinirole is a novel compound used to treat the symptoms of Parkinson's disease. This study investigated its in vivo distribution and metabolic pathways in mice, rats, cynomolgus monkeys, and humans via oral and intravenous administration of ropinirole hydrochloride. …In rats, the main metabolic pathway is hydroxylation of the aromatic ring to 7-hydroxyropinirole. In mice, monkeys, and humans, the main metabolic pathway is N-depropylation. The N-depropyl metabolite is further metabolized to 7-hydroxy and carboxylic acid derivatives. In all species, the generated metabolites are typically further metabolized via glucuronidation. 7-hydroxyropinirole is the only ropinirole metabolite previously shown to have significant in vivo dopamine agonist activity. ... The dopamine receptor agonist ropinirole (SKF-101468) is used to treat Parkinson's disease. Ropinirole can be metabolized through two pathways to produce a series of different metabolites, but the dominant metabolic pathway varies by species. It is currently unclear whether any of these metabolites contributes to its anti-Parkinson's activity, or whether D3 or D2 receptor agonist activity plays a dominant role. Therefore, this study tested ropinirole and its major metabolites SKF-104557, SKF-97930, and SKF-96990, as well as the rat metabolite SKF-89124, in a 6-hydroxydopamine-induced Parkinson's disease model. SKF-89124 and SKF-96990 were also subjected to radioligand binding and microphysiological assays, detecting cloned human dopamine D2 and D3 receptors. Ropinirole and SKF-89124 showed comparable potency in vivo, both increasing circling behavior in a dose-dependent manner, with subcutaneous doses of 0.05–0.8 mg kg⁻¹ for ropinirole and 0.05–0.75 mg kg⁻¹ for SKF-89124. SKF-96990 and SKF-97930 did not increase circling frequency at doses up to 15 mg kg⁻¹. Some circling behavior was observed in SKF-104557 at a dose of 15 mg kg⁻¹, but its response was less than half that of ropinirole (0.8 mg kg⁻¹). SKF-104557 is 150 times less potent than ropinirole. In radioligand binding assays, SKF-89124 showed a 30-fold higher affinity for the D3 receptor than for the D2 receptor, but no selectivity was observed in functional microphysiological assays. In radioligand binding assays, SKF-96990 showed a 10-fold higher selectivity for the D3 receptor than for the D2 receptor. In radioligand binding assays, both ropinirole and SKF-104557 showed a 20-fold higher selectivity for the D3 receptor than for the D2 receptor, but only a 10-fold selectivity in microphysiological assays. SKF-97930 showed no activity in either radioligand binding assays or microphysiological assays. The major metabolites of ropinirole contributed little to its activity in this Parkinson's disease model. Ropinirone lacks selectivity for dopamine D3/D2 receptors, thus ruling out the possibility of linking the level of D2 or D3 receptor activity to the behavioral therapeutic effects of ropinirone. This study aimed to identify the cytochrome P450 enzymes responsible for the biotransformation of ropinirone by metabolizing it in vitro. The metabolic pathway of ropinirone after incubation with human liver microsomes involves N-depropylation and hydroxylation. Enzyme kinetics indicated that each metabolic pathway involves at least two enzymes. There is a high-affinity fraction (Km of 5–87 μM) and a low-affinity fraction (Km of approximately two orders of magnitude higher). Pre-incubation of microsomes with furazolidone eliminated the high-affinity fraction. Furthermore, incubation of ropinirone with microsomes derived from CYP1A2-transfected cells rapidly produced N-depropylation and hydroxylation metabolites. Ketoconazole was also observed to inhibit ropinirone metabolism, suggesting a smaller role for CYP3A. Multivariate correlation data are consistent with the involvement of cytochrome P450 enzymes 1A2 and 3A in ropinirole metabolism. Therefore, it can be concluded that at lower (clinically relevant) concentrations, the primary P450 enzyme responsible for ropinirole metabolism is CYP1A2, while the role of CYP3A is primarily observed at higher concentrations. Known metabolites of ropinirole include 4-[2-(dipropylamino)ethyl]-7-hydroxy-1,3-dihydro-2H-indole-2-one and 4-(2-(propylamino)ethyl)indolin-2-one. In the liver, ropinirole is primarily metabolized to inactive metabolites via N-depropionylation and hydroxylation pathways, mainly catalyzed by the P450 isoenzyme CYP1A2. N-Depropylropinirole is the major metabolite in urine (40%), followed by the carboxylic acid metabolite (10%) and the glucuronide of the hydroxyl metabolite (10%). Elimination pathway: Ropinirole is primarily metabolized in the liver to inactive metabolites, with less than 10% of the administered dose excreted unchanged in the urine. Half-life: 6 hours (bioactive half-life): approximately 6 hours. Terminal elimination half-life: approximately 6 hours (range 2-27 hours)... Ropinirole is a novel compound used to treat the symptoms of Parkinson's disease. This study investigated its distribution and metabolic pathways in mice, rats, cynomolgus monkeys, and humans via oral and intravenous administration of ropinirole hydrochloride. The compound has a relatively short terminal elimination half-life (0.5 hours in rats and 1.3 hours in monkeys), although there is evidence of a second elimination phase in monkeys, with an elimination half-life of approximately 5-11 hours. |
|
| Toxicity/Toxicokinetics |
Toxicity Summary
Identification and Uses: Ropinirone hydrochloride is a dipropylaminoethyl indoleone derivative belonging to the non-ergot dopamine receptor agonist class. It is used to treat symptoms of idiopathic Parkinson's syndrome. It is also used to treat symptoms of moderate to severe primary restless legs syndrome (RLS). Human Exposure and Toxicity: The maximum reported overdose of ropinirone in clinical trials was 435 mg (62.1 mg/day) over 7 days. Symptoms reported by patients taking more than 24 mg daily include common side effects during dopaminergic therapy (nausea, dizziness), as well as visual hallucinations, hyperhidrosis, claustrophobia, chorea, palpitations, weakness, and nightmares. Other symptoms reported by patients taking 24 mg or less or overdoses (dose unknown) include vomiting, worsening cough, fatigue, syncope, vasovagal syncope, movement disorders, agitation, chest pain, orthostatic hypotension, drowsiness, and confusion. Post-marketing reports indicate that patients may experience new or worsening mental and behavioral changes, which can be severe, including psychotic-like behaviors, while taking ropinirole or after starting or increasing the dose of ropinirole. These abnormal thoughts and behaviors may include one or more manifestations such as delusions, illusions, hallucinations, confusion, psychotic-like behaviors, disorientation, aggressive behavior, agitation, and delirium. Case reports indicate that patients taking one or more drugs that enhance central dopaminergic tone, including ropinirole, may experience intense gambling urges, increased libido, intense spending urges, binge eating or compulsive eating, and/or other intense and uncontrollable urges. These drugs are commonly used to treat Parkinson's disease and restless legs syndrome. In some cases (but not all), these urges have been reported to disappear upon dose reduction or discontinuation. Ropinirole does not cause chromosomal aberrations in human lymphocytes. Animal studies: Ropinirole has a biphasic effect on motor activity. Low doses inhibit spontaneous movement, while high doses stimulate movement. In mice, intraperitoneal injections of 10 mg/kg and 100 mg/kg caused inhibition and stimulation, respectively. In rats, decreased activity was observed in the 0.3 mg/kg dose group, while hyperactivity was observed in the 1–30 mg/kg dose groups. Ropinirole caused a dose-dependent decrease in blood pressure and a decrease in heart rate in anesthetized and awake spontaneously hypertensive rats. Single-dose studies were conducted in both mice and rats. Clinical symptoms were significantly dose-dependent, including hyperactivity, dyskinesia, stereotyped behaviors, tremors, convulsions, and ultimately death. A two-year carcinogenicity study of ropinirole was conducted in mice and rats at oral doses up to 50 mg/kg/day. In rats, all tested doses resulted in an increased incidence of testicular interstitial cell adenomas. In mice, a dose of 50 mg/kg/day resulted in an increased incidence of benign endometrial polyps. The endocrine mechanisms underlying these tumors in rats are considered to be unrelated to those in humans. Ropinirole was administered to mated female rats. No maternal deaths or abortions were observed. A dose-dependent increase in post-implantation embryo loss was observed (up to 43%), along with a decrease in average fetal weight. Delayed ossification of the hind limb metatarsals and other malformations, including phalangeal abnormalities, neural tube defects, and cardiovascular abnormalities, were observed in the fetuses. Rats were administered ropinirole at doses of 0.1, 1.0, and 10 mg/kg/day from day 15 of gestation until weaning. No maternal death or abortion was observed. Although pups in the high-dose group had higher weights than the control group at 1–2 days after birth, their weight subsequently decreased, reaching 18% less than the control group by day 14. Female offspring exhibited reduced startle responses to auditory and tactile stimuli, while this was not observed in male offspring. Administration of ropinirole to female rats before mating, during mating, and throughout gestation resulted in implantation failure. This effect in rats is thought to be due to ropinirole's reduction of prolactin levels. In rats during the early prolactin-dependent period of pregnancy, oral administration of low-dose (5 mg/kg) ropinirole showed no effect on fertility in female rats at doses up to 100 mg/kg/day. No effect on male rat fertility was observed at doses up to 125 mg/kg/day. Ropinirole was not found to be mutagenic or chromosomally breakable in vitro (Ames test, mouse lymphoma Tk test) or in vivo mouse micronucleus assays. Ropinirole binds to dopamine receptors D3 and D2. Although the exact mechanism of action of ropinirole in treating Parkinson's disease is unclear, it is generally believed to be related to its ability to stimulate these receptors in the striatum. Animal electrophysiological studies support this conclusion, showing that ropinirole affects the firing frequency of striatal neurons by activating dopamine receptors in the striatum and substantia nigra (the site of neurons projecting to the striatum). Hepatotoxicity Ropinirone has been reported to cause elevated serum transaminases or alkaline phosphatase in a small number of patients, but these abnormalities are usually mild, asymptomatic, and resolve spontaneously, even without dose adjustment. Ropinirone has been associated with a small number of cases of acute liver injury, but its clinical features and typical enzyme elevation patterns are not well understood. In one case report, the onset was 2 months, with a mixed pattern of liver enzyme elevations and marked jaundice. No immune hypersensitivity or autoimmune features were observed. The liver injury resolved within 2 months after discontinuation of the drug. Therefore, ropinirone can cause acute, clinically significant liver injury with jaundice, but this is rare. Probability score: D (Possible, but rare, cause of clinically significant liver injury). Pregnancy and Lactation Effects ◉ Overview of Use During Lactation There is currently no information on the use of ropinirone during lactation, but the drug suppresses serum prolactin levels, which may affect breastfeeding. Especially during the nursing of newborns or premature infants, alternative medications may be necessary. ◉ Effects on breastfed infants No published information found as of the revision date. ◉ Effects on lactation and breast milk No published information found regarding breastfeeding mothers as of the revision date. Ropinirole can lower serum prolactin levels. Clinically, it has been successfully used to lower prolactin levels in women with hyperprolactinemia and alleviate galactorrhea symptoms. For mothers who have established lactation, prolactin levels may not affect their ability to breastfeed. Protein binding 40% bound to plasma proteins, with a serum-to-plasma ratio of 1:1. Drug interactions Because ropinirole is a dopamine agonist, dopamine antagonists (such as neuroleptics (e.g., phenothiazines, butyrophenones, thioxanone) or metoclopramide) may reduce the efficacy of Requip. Population pharmacokinetic analysis showed that higher doses of estrogen (often associated with hormone replacement therapy (HRT)) reduced ropinirole clearance. Starting or stopping HRT may require dose adjustment of Requip. There is currently no information regarding potential interactions between Requip and alcohol. As with other central nervous system active drugs, patients should be advised to avoid alcohol consumption while taking Requip. This study investigated the effects of Requip (2 mg, three times daily) on the pharmacokinetics of digoxin (0.125–0.25 mg, once daily) in patients with Parkinson's disease (n=10, mean age 72 years). Results showed that, at steady state, co-administration with Requip resulted in a 10% decrease in digoxin AUC, but no change in mean trough concentration. However, the effect of higher recommended doses of Requip on digoxin pharmacokinetics remains unclear. For more complete data on interactions with ropinirole (12 items in total), please visit the HSDB record page. Non-human toxicity values Rat intravenous LD50: 66 mg/kg Rat oral LD50: 862 mg/kg Mouse intravenous LD50: 46 mg/kg Mouse oral LD50: 657 mg/kg |
|
| References | ||
| Additional Infomation |
Therapeutic Uses
Anti-Parkinson's Disease Drug; Dopamine Agonist /Clinical Trials/ ClinicalTrials.gov is a registry and results database that lists human clinical studies funded by public and private institutions worldwide. The website is maintained by the National Library of Medicine (NLM) and the National Institutes of Health (NIH). Each record on ClinicalTrials.gov includes a summary of the study protocol, including: the disease or condition; the intervention (e.g., the medical product, behavior, or procedure under investigation); the title, description, and design of the study; participation requirements (eligibility criteria); the location of the study; contact information for the study location; and links to relevant information from other health websites, such as the NLM's MedlinePlus (for providing patient health information) and PubMed (for providing citations and abstracts of academic articles in the medical field). Ropinirole is listed in this database. Requip is indicated for the treatment of Parkinson's disease. /US Product Label Contains/ Requip is indicated for the treatment of moderate to severe primary restless legs syndrome (RLS). /US product label contains/ For more complete data on the therapeutic uses of ropinirole (ROPINIROLE) (out of 7), please visit the HSDB record page. Drug Warnings Post-marketing reports indicate that new or worsening changes in mental status and behavior, including psychotic-like behaviors, may occur during treatment with Requip or after starting or increasing the dose of Requip. These changes may be serious. Other medications used to improve symptoms of Parkinson's disease may also have similar effects on thought and behavior. Such abnormal thought and behavior may include one or more manifestations such as delusional ideas, illusions, hallucinations, confusion, psychotic-like behaviors, disorientation, aggressive behavior, agitation, and delirium. Ropinirole (Requip) should generally not be used to treat patients with severe psychosis due to the risk of exacerbating psychosis. Additionally, some medications used to treat psychosis may worsen symptoms of Parkinson's disease and may reduce the effectiveness of ropinirole. The safety and efficacy of ropinirole in pediatric patients have not been established. Ropinirone inhibits the secretion of prolactin in the human body and may suppress lactation. Ropinirone has been detected in rat milk. It is currently unknown whether this drug is excreted into human milk. Because many drugs are excreted into breast milk, caution should be exercised when breastfeeding women take ropinirone (Requip). For more complete data on drug warnings for ropinirone (21 in total), please visit the HSDB record page. Pharmacodynamics Effects on Parkinson's Disease and Restless Legs Syndrome This drug relieves or improves symptoms of Parkinson's disease or restless legs syndrome by stimulating dopamine receptors (which regulate movement). Effects on Blood Pressure Clinical experience with dopamine agonists (including ropinirone) suggests an association with impaired blood pressure regulation, leading to orthostatic hypotension, particularly in dose-escalating patients. In some clinical studies, changes in blood pressure have been associated with orthostatic symptoms, bradycardia, and one case of transient sinus arrest with syncope occurred in a healthy volunteer. The mechanism by which ropinirole induces orthostatic hypotension is believed to be due to a weakened D2 receptor-mediated noradrenergic response, leading to a decrease in peripheral vascular resistance. Nausea is also a common symptom associated with orthostatic hypotension. Effects on prolactin Oral doses as low as 0.2 mg of ropinirole can suppress serum prolactin concentrations in healthy male volunteers. In the dose range of 0.01 to 2.5 mg, ropinirole has no dose-related effects on ECG waveforms and rhythms in young, healthy male volunteers. Effects on QT interval At doses up to 4 mg/day, ropinirole has no dose- or exposure-related effects on the mean QT interval in healthy male and female volunteers. The effects of ropinirole on the QTc interval at high exposures (possibly due to drug interactions, hepatic impairment, or higher doses) have not been adequately evaluated. |
| Molecular Formula |
C16H24N2O
|
|---|---|
| Molecular Weight |
260.37456
|
| Exact Mass |
260.188
|
| Elemental Analysis |
C, 73.81; H, 9.29; N, 10.76; O, 6.14
|
| CAS # |
91374-21-9
|
| Related CAS # |
Ropinirole hydrochloride; 91374-20-8; Ropinirole-d14; 1132746-05-4
|
| PubChem CID |
5095
|
| Appearance |
Solid powder
|
| Density |
1.0±0.1 g/cm3
|
| Boiling Point |
410.5±45.0 °C at 760 mmHg
|
| Melting Point |
243-250°C
|
| Flash Point |
202.0±28.7 °C
|
| Vapour Pressure |
0.0±1.0 mmHg at 25°C
|
| Index of Refraction |
1.539
|
| LogP |
3.19
|
| Hydrogen Bond Donor Count |
1
|
| Hydrogen Bond Acceptor Count |
2
|
| Rotatable Bond Count |
7
|
| Heavy Atom Count |
19
|
| Complexity |
287
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
O=C1CC2C(=CC=CC=2CCN(CCC)CCC)N1
|
| InChi Key |
UHSKFQJFRQCDBE-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C16H24N2O/c1-3-9-18(10-4-2)11-8-13-6-5-7-15-14(13)12-16(19)17-15/h5-7H,3-4,8-12H2,1-2H3,(H,17,19)
|
| Chemical Name |
4-[2-(dipropylamino)ethyl]-1,3-dihydroindol-2-one
|
| Synonyms |
Ropinirole; AS-041164; SKF 101468; AS 041164; SKF-101,468; AS041164; SKF 101468
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.8407 mL | 19.2034 mL | 38.4069 mL | |
| 5 mM | 0.7681 mL | 3.8407 mL | 7.6814 mL | |
| 10 mM | 0.3841 mL | 1.9203 mL | 3.8407 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Cognitive Remediation for Cocaine Dependence
CTID: NCT01393457
Phase: Phase 2 Status: Completed
Date: 2018-03-29
 |
|---|
  |
 |