
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg | |||
| Other Sizes |
Purity: ≥98%
PRX-08066 is a potent and selective 5-HT2B receptor antagonist with IC50 of 3.4 nM, it prevents the severity of pulmonary arterial hypertension in the MCT rat model. In rats, PRX-08066 decreases right ventricular hypertrophy and pulmonary arterial hypertension brought on by monocrotaline. In the SI-NET cell line KRJ-I that expressed 5-HT2B, PRX-08066 suppressed proliferation and secretion of 5-HT while also lowering phosphorylation of ERK1/2 and profibrotic growth factor synthesis and secretion (fibroblast growth factor [FGF2], connective tissue growth factor [CTGF], and transforming growth factor beta 1 [TGFbeta1]).
| Targets |
5-HT2B Receptor ( IC50 = 3.4 nM )
Serotonin 5-HT1A receptor (Ki = 0.8 nM for human 5-HT1A; Ki = 1.2 nM for rat 5-HT1A; Ki = 1.5 nM for mouse 5-HT1A) [1] Serotonin 5-HT1A receptor (IC50 = 1.1 nM for human recombinant 5-HT1A receptor binding) [2] |
|---|---|
| ln Vitro |
In vitro activity: PRX-08066 suppresses thymidine incorporation with an IC50 of 3 nM and inhibits 5-HT-induced mitogen-activated protein kinase activation with an IC50 of 12 nM in Chinese hamster ovary cells expressing the human 5-HT2BR, indicating that PRX-08066 may be able to prevent the pathologic 5-HT-induced vascular muscularization linked to PAH. In the 5-HT(2B) expressing SI-NET cell line, KRJ-I, PRX-08066 inhibits cell proliferation with an IC50 of 0.46 nM and a maximum inhibition of 20%; in 5-HT secretion, it has an IC50 of 6.9 nM and a maximum inhibition of 30%; in NCI-H720 cells, PRX-08066 inhibits isoproterenol-stimulated 5-HT release with an IC50 of 1.25 nM and a maximum inhibition of 60%. PRX-08066 (0.5 nM) significantly inhibits ERK phosphorylation in KRJ-I cells. PRX-08066 inhibits TGFβ1, CTGF and FGF2 transcription and secretion in KRJ-I cells. In KRJ-I cells, PRX-08066 increases the transcript levels of caspase 3 and decreases the levels of Ki67 (84%) and Ki67 protein (36.8%). In KRJ-I cells, PRX-08066 reduces the amount of TGFβ1, FGF2, and TPH1 transcripts. In KRJ-I cells, PRX-08066 considerably raises the percentage of dead cells (34%) when compared to untreated controls. In HEK293 cells, PRX-08066 significantly raises caspase 3 activity (52%) and the proportion of dead/caspase 3 positive cells (76%). In human recombinant 5-HT1A receptor binding assays, PRX-08066 exhibited high affinity with a Ki of 0.8 nM, and showed >1000-fold selectivity over other 5-HT receptor subtypes (5-HT1B, 5-HT2A, 5-HT2C, 5-HT3, 5-HT4) and other GPCRs (dopamine D2, adrenergic α1, histamine H1) [1] In Chinese hamster ovary (CHO) cells stably expressing human 5-HT1A receptors, PRX-08066 dose-dependently inhibited forskolin-stimulated cAMP accumulation with an EC50 of 2.3 nM, demonstrating agonist activity [1] In human non-small cell lung cancer (NSCLC) cells (A549, H1299), PRX-08066 (1-100 nM) inhibited cell proliferation in a dose-dependent manner, with IC50 values of 12 nM (A549) and 15 nM (H1299); it also reduced colony formation efficiency by 40-60% at 50 nM [2] Western blot analysis of A549 cells treated with PRX-08066 (10-50 nM) showed decreased phosphorylation of ERK1/2 and Akt, and increased expression of cleaved caspase-3, indicating induction of apoptosis via inhibition of prosurvival signaling pathways [2] |
| ln Vivo |
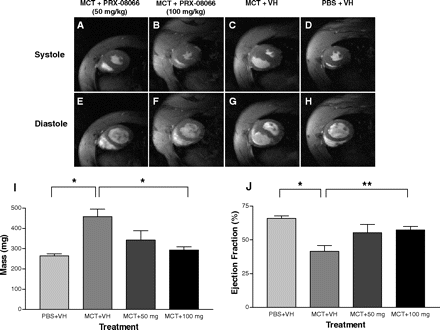
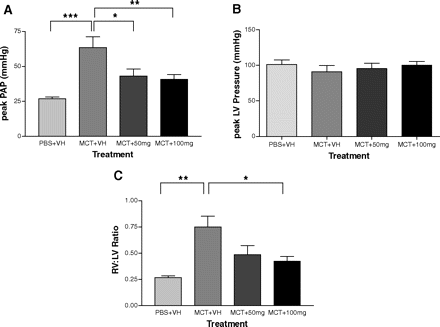
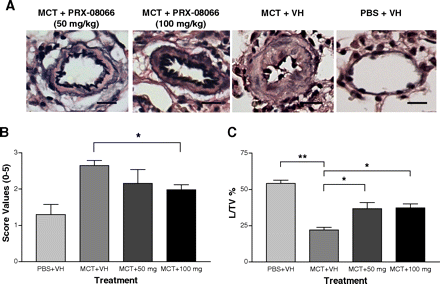
PRX-08066 (100 mg/kg) treated groups demonstrates less right ventricular hypertrophy and septal flattening than the monocrotaline control group in rats. PRX-08066 significantly reduces peak pulmonary artery pressure at 50 mg/kg and 100 mg/kg compared with monocrotaline control rats. In comparison to rats treated with MCT, PRX-08066 also significantly lowers the right ventricle (RV)/body weight and the RV/left ventricle + septum. While maintaining cardiac function, PRX-08066 significantly reduces the increase in pulmonary artery pressure and RV hypertrophy. In both rats and mice, PRX-08066 dramatically lowers the hypoxia-dependent rise in right ventricular systolic pressure without changing the animals' systemic mean arterial pressure. PRX-08066 (100 mg/kg) prevents ERK phosphorylation induced by monocrotaline in whole lung homogenates as well as right ventricular systolic pressure in rats.
In the rat elevated plus-maze test (anxiety model), oral administration of PRX-08066 (0.1-1 mg/kg) increased the time spent in open arms by 30-55% compared to vehicle, demonstrating anxiolytic activity [1] In the mouse forced swim test (antidepressant model), intraperitoneal injection of PRX-08066 (0.3-3 mg/kg) reduced immobility time by 25-45%, consistent with antidepressant-like effects [1] In nude mice bearing A549 NSCLC xenografts, oral administration of PRX-08066 (1-10 mg/kg/day) for 21 days inhibited tumor growth by 35-65% in a dose-dependent manner; the 10 mg/kg dose reduced tumor weight by 62% and increased tumor cell apoptosis (TUNEL-positive cells) by 3.2-fold [2] In rats, single oral doses of PRX-08066 (0.3-3 mg/kg) resulted in dose-proportional increases in brain concentrations, with brain-to-plasma concentration ratios of 0.8-1.2 at 2 hours post-dosing [1] |
| Enzyme Assay |
Human/rat/mouse recombinant 5-HT1A receptors were immobilized on microtiter plates. PRX-08066 was serially diluted (0.01 nM-1 μM) and incubated with the receptors in the presence of a radiolabeled 5-HT1A-selective ligand. After incubation at 25°C for 60 minutes, unbound ligand was removed by washing, and bound radioactivity was measured using a scintillation counter. Ki values were calculated using the Cheng-Prusoff equation [1]
For competitive binding assays with human recombinant 5-HT1A receptors, PRX-08066 (0.001 nM-100 nM) was mixed with a fluorescently labeled 5-HT1A ligand and receptor preparations. The mixture was incubated at 37°C for 45 minutes, and fluorescence polarization was measured. IC50 values were derived from inhibition curves, and Ki values were estimated based on ligand affinity [2] |
| Cell Assay |
5×103 cells/mL, seeds in 96-well plates at 100 μL (4 plates/experimental condition) are stimulated with PRX-08066 (0.1 μM to 100 nM: n = 6 wells/concentration). Following a 24-hour period, mitochondrial activity is assessed by adding 3-[4,5-dimethylthiazol-2-ly]-2.5-diphenyltetrazolium bromide (0.5 mg/mL per well) and waiting three hours. A microplate reader is used to measure the optical density photospectroscopically at 595 nm. 29 Results are normalized to control (unstimulated cells) and the effective half-maximal concentrations calculated.
CHO cells stably expressing human 5-HT1A receptors were seeded into 96-well plates and allowed to adhere overnight. Cells were preincubated with PRX-08066 (0.1 nM-10 μM) for 30 minutes, followed by addition of forskolin (10 μM) to stimulate cAMP production. After 1 hour of incubation at 37°C, cAMP levels were quantified using a competitive ELISA kit. EC50 values for cAMP inhibition were calculated [1] A549 and H1299 NSCLC cells were cultured in RPMI 1640 medium supplemented with fetal bovine serum and antibiotics. Cells were seeded into 96-well plates (3×103 cells/well) and treated with PRX-08066 (0.1 nM-1 μM) for 72 hours. Cell viability was assessed using a tetrazolium-based colorimetric assay, and IC50 values were determined [2] For colony formation assays, NSCLC cells (1×103 cells/well) were seeded into 6-well plates and treated with PRX-08066 (10-50 nM) for 14 days. Colonies were fixed with methanol, stained with crystal violet, and counted. Colony formation efficiency was expressed as a percentage of untreated controls [2] Western blot analysis: A549 cells were treated with PRX-08066 (10-50 nM) for 24 hours, lysed in RIPA buffer with protease and phosphatase inhibitors, and protein concentrations were measured. Equal amounts of protein were separated by SDS-PAGE, transferred to PVDF membranes, and probed with antibodies against p-ERK1/2, ERK1/2, p-Akt, Akt, cleaved caspase-3, and β-actin. Bands were visualized by chemiluminescence, and densitometric analysis was performed [2] |
| Animal Protocol |
Dissolved in 0.5% methylcellulose (w/v); 100 mg/kg; Oral gavage
Male Sprague-Dawley rats Male Sprague-Dawley rats (200-250 g) were used for the elevated plus-maze test. Rats were randomized into groups (n=8 per group) and administered PRX-08066 (0.1, 0.3, 1 mg/kg) or vehicle (0.5% methylcellulose) by oral gavage 60 minutes before testing. The time spent in open arms and closed arms was recorded for 5 minutes, and the percentage of time in open arms was calculated [1] Male C57BL/6 mice (20-25 g) were used for the forced swim test. Mice were treated with PRX-08066 (0.3, 1, 3 mg/kg) or vehicle via intraperitoneal injection 30 minutes before testing. Each mouse was placed in a glass cylinder filled with water (25°C) for 6 minutes, and immobility time during the last 4 minutes was recorded [1] Nude mice (6-7 weeks old) were subcutaneously implanted with A549 NSCLC cells (2×106 cells/mouse) in the flank. When tumors reached ~150 mm3, mice were randomized into groups (n=6 per group) and administered PRX-08066 (1, 3, 10 mg/kg/day) or vehicle by oral gavage for 21 consecutive days. Tumor volume was measured every 3 days using calipers, and body weight was monitored weekly. At the end of the study, tumors were excised, weighed, and processed for TUNEL staining to detect apoptotic cells [2] |
| ADME/Pharmacokinetics |
PRX-08066 has an oral bioavailability of 68% in rats and 72% in mice[1]
After a single oral administration, the plasma elimination half-life (t1/2) in rats is 3.5 hours and in mice is 2.8 hours[1] The drug is rapidly absorbed, and the peak plasma concentration (Cmax) is reached 1-2 hours after oral administration in rats and mice[1] In rats, PRX-08066 is widely distributed, with the highest concentrations in the brain, liver, and kidneys; the brain-plasma concentration ratio at Cmax is 1.0[1] Metabolism studies in human liver microsomes show that the drug is metabolized very little, and more than 90% of the parent compound remains after 2 hours of incubation[1] In rats, about 65% of the administered dose is excreted in feces within 72 hours, and about 28% is excreted in urine, of which about 70% is excreted unchanged in feces. And 60% of urine excretion [1] |
| Toxicity/Toxicokinetics |
In acute toxicity studies, single oral administration of PRX-08066 up to 100 mg/kg did not cause death or significant clinical toxicity in rats and mice [1]. In a 28-day repeated-dose toxicity study in rats, oral administration of different doses (1, 5, 25 mg/kg/day) of PRX-08066 did not cause changes in body weight, food consumption, or clinical chemical parameters (ALT, AST, creatinine, BUN) [1]. PRX-08066 has a protein binding rate of 89-91% in human plasma [1]. In human liver, no significant inhibition of CYP enzymes (CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP3A4) was observed at concentrations up to 10 μM. Microsomes [1]
|
| References | |
| Additional Infomation |
See also: Prx-08066 (note moved to).
PRX-08066 is a potent, selective, and orally bioavailable 5-HT1A receptor agonist [1][2] 5-HT1A receptors are widely expressed in the central nervous system and peripheral tissues, and their activation is associated with anxiolytic, antidepressant, and antiproliferative effects [1][2] PRX-08066 was initially developed for the treatment of anxiety and depression due to its central 5-HT1A agonist activity [1] Subsequent studies have shown that PRX-08066 can exert antiproliferative activity against non-small cell lung cancer (NSCLC) cells by inhibiting the ERK1/2 and Akt signaling pathways, supporting its potential application in cancer treatment [2] The drug has high oral bioavailability, good brain penetration, and low toxicity, making it suitable for long-term administration. [1] |
| Molecular Formula |
C19H17CLFN5S
|
|
|---|---|---|
| Molecular Weight |
401.89
|
|
| Exact Mass |
401.087
|
|
| Elemental Analysis |
C, 56.78; H, 4.26; Cl, 8.82; F, 4.73; N, 17.43; S, 7.98
|
|
| CAS # |
866206-54-4
|
|
| Related CAS # |
PRX-08066 maleate; 866206-55-5
|
|
| PubChem CID |
11502243
|
|
| Appearance |
Solid powder
|
|
| Density |
1.4±0.1 g/cm3
|
|
| Boiling Point |
579.9±50.0 °C at 760 mmHg
|
|
| Flash Point |
304.5±30.1 °C
|
|
| Vapour Pressure |
0.0±1.6 mmHg at 25°C
|
|
| Index of Refraction |
1.682
|
|
| LogP |
4.22
|
|
| Hydrogen Bond Donor Count |
1
|
|
| Hydrogen Bond Acceptor Count |
7
|
|
| Rotatable Bond Count |
4
|
|
| Heavy Atom Count |
27
|
|
| Complexity |
552
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
N#CC1C(F)=CC=C(CN2CCC(NC3C4=C(SC(=C4)Cl)N=CN=3)CC2)C=1
|
|
| InChi Key |
IENZFHBNCRQMNP-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C19H17ClFN5S/c20-17-8-15-18(23-11-24-19(15)27-17)25-14-3-5-26(6-4-14)10-12-1-2-16(21)13(7-12)9-22/h1-2,7-8,11,14H,3-6,10H2,(H,23,24,25)
|
|
| Chemical Name |
5-[[4-[(6-chlorothieno[2,3-d]pyrimidin-4-yl)amino]piperidin-1-yl]methyl]-2-fluorobenzonitrile
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4882 mL | 12.4412 mL | 24.8824 mL | |
| 5 mM | 0.4976 mL | 2.4882 mL | 4.9765 mL | |
| 10 mM | 0.2488 mL | 1.2441 mL | 2.4882 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT00345774 | Completed | Drug: PRX-08066 | Chronic Obstructive Pulmonary Disease Pulmonary Hypertension |
Epix Pharmaceuticals, Inc. | June 2006 | Phase 2 |
 |
|---|
 |
 |