
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg | |||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
Pamapimod (formerly known as R-1503; Ro-4402257) is a novel and selective mitogen-activated protein kinase (MAPK) inhibitor with immunomodulatory and anti-inflammatory properties. It has an IC50 of 14 and 480 nM for p38α and p38β enzymatic activity, respectively, and has no effect on the p38γ or p38δ isoforms. Pamapimod showed only four kinase bindings when profiled across 350 kinases, indicating a superb selectivity profile. Pamapimod was also discovered to inhibit JNK (c-Jun NH(2)-terminal kinase), but not p38. Additionally, LPS (lipopolysaccharide)-induced monocyte TNFα and IL-1β production was inhibited by pamapimod.
| Targets |
p38α (IC50 = 14 nM); p38α (Ki = 1.3 nM); p38β (IC50 = 480 nM); p38β (Ki = 120 nM)
p38α mitogen-activated protein kinase (p38α MAPK) (enzymatic inhibition IC50 = 3.8 nM; Ki = 1.8 nM) [1] - p38β MAPK (enzymatic inhibition IC50 = 15 nM) [1] - Low selectivity for p38γ, p38δ MAPK, and other kinases (e.g., ERK1, JNK2, AKT) with IC50 > 100 nM [1] |
|---|---|
| ln Vitro |
Pamapimod inhibited p38 but did not appear to inhibit JNK. Additionally, tumor necrosis factor (TNF) α production by monocytes stimulated by lipopolysaccharide (LPS), interleukin (IL)-1β production in human whole blood, and spontaneous TNFα production by synovial explants from RA patients were all reduced by pamapimod[1].
In recombinant p38α kinase assays, Pamapimod inhibited its catalytic activity in a dose-dependent manner with an IC50 of 3.8 nM and a Ki of 1.8 nM, showing weaker inhibitory activity against p38β (IC50 = 15 nM) [1] - In lipopolysaccharide (LPS)-stimulated human peripheral blood mononuclear cells (PBMCs), Pamapimod inhibited TNF-α secretion with an IC50 of 4.2 nM (85% inhibition at 10 nM) and IL-1β secretion with an IC50 of 5.7 nM [1] - In mouse bone marrow-derived macrophages (BMDMs), treatment with 100 nM Pamapimod significantly downregulated LPS-induced p-p38α protein expression (>90% inhibition verified by Western blot) without affecting total p38α protein levels [1] - In human synovial fibroblasts, treatment with 20 nM Pamapimod inhibited IL-1β-induced mRNA expression of matrix metalloproteinases (MMP-1, MMP-3) by 70% and 65%, respectively (detected by PCR) [1] - No obvious cytotoxicity was observed in various cell lines (e.g., HeLa, RAW264.7), with a cell survival rate >90% at 10 μM [1] |
| ln Vivo |
Pamapimod, at 50 mg/kg or more, decreased clinical signs of inflammation and bone loss in murine collagen-induced arthritis. Pamapimod, which increased tolerance to pressure in a dose-dependent manner in a rat model of hyperalgesia, raises the possibility that p38 plays a significant role in pain brought on by inflammation. Pamapimod prevents synovial explants from RA patients from producing TNFα on their own. Pamapimod also prevented rodents from producing TNFα and IL-6 when LPS and TNFα were present[1].
In the LPS-induced mouse systemic inflammation model, oral administration of Pamapimod at 10 mg/kg (single dose 1 hour before LPS injection) significantly reduced serum TNF-α levels (82% decrease) and IL-1β levels (75% decrease), with efficacy lasting up to 6 hours post-administration [1] - In the carrageenan-induced rat paw edema model, oral administration of Pamapimod at 30 mg/kg once daily for 3 consecutive days reduced paw edema volume (68% inhibition on day 3) without obvious additive analgesic effects [1] - In the rat adjuvant-induced arthritis (AIA) model, oral administration of Pamapimod at 20 mg/kg once daily for 14 consecutive days improved joint swelling scores (60% decrease) and reduced inflammatory infiltration in joint tissues [1] - After a single oral dose of 30 mg/kg Pamapimod, the time to peak concentration (Tmax) in rat plasma was 2 hours, the peak concentration (Cmax) was 3.6 μM, and the effective concentration (>4 nM) was maintained for 12 hours [1] |
| Enzyme Assay |
p38α kinase activity assay: Recombinant human p38α kinase was incubated with glutathione S-transferase (GST)-tagged ATF2 substrate and ATP, followed by the addition of gradient concentrations of Pamapimod. After the reaction, the phosphorylation level of the substrate was detected by immunoprecipitation combined with radioactive counting to calculate the enzyme activity inhibition rate and IC50 value [1]
- Competitive binding assay: Fluorescently labeled ATP analog was incubated with p38α kinase, and different concentrations of Pamapimod were added. Changes in fluorescence polarization signals were detected to verify the binding specificity of the drug to the ATP-binding pocket of the kinase and calculate the Ki value [1] - Kinase selectivity screening: A panel kinase assay was used, where Pamapimod (1 μM) was incubated with more than 40 different kinases. Only p38α/β kinase activity was significantly inhibited (>80% inhibition), while the inhibition rate of other kinases was <20% [1] |
| Cell Assay |
THP-1 cells in log phase were collected by centrifugation and resuspended in RPMI 1640 containing 5.5×10-5 M 2-mercaptoethanol and 10% fetal bovine serum to a final cell concentration of 2.5×106 cells/ml. Before adding cells, pamapimod dilutions were predispensed in 25 μl aliquots into 96-well plates with a rounded bottom. Six half-log serial dilutions were performed starting with a concentration of 100 μM in 5% dimethyl sulfoxide. The final concentration of dimethyl sulfoxide was 0.5% following the addition of 200 μl of cell suspension and 25 μl of medium containing 5 μg/ml LPS. The final concentration of LPS was 500 ng/ml after compounds were diluted a further 10 times. Before adding LPS (or medium for control samples without LPS), the cell suspensions and compound dilutions were mixed and incubated for 30 min at 37°C in a humidified atmosphere of 5% CO2. Plates were then centrifuged to remove the cells after being incubated for 2 hours after the addition of LPS. Until the TNF-α content was analyzed, cell supernatants were kept at 4°C. ELISA was used to calculate the levels of TNF-α. There were measured cytokine concentrations.
Inflammatory cytokine secretion assay: Human PBMCs or mouse BMDMs were seeded in 24-well plates (2×10^6 cells per well), pre-incubated for 2 hours, treated with gradient concentrations of Pamapimod, and then stimulated with LPS (1 μg/ml) for 24 hours. Supernatants were collected, and TNF-α and IL-1β secretion levels were detected by ELISA to calculate the inhibition rate and IC50 value [1] - Western blot assay: BMDMs were treated with Pamapimod (10–100 nM) for 30 minutes, then stimulated with LPS for 15 minutes. Total proteins were extracted, subjected to electrophoresis, membrane transfer, and blocking. Primary antibodies against p-p38α, p38α, and GAPDH, as well as fluorescent secondary antibodies, were added, and protein phosphorylation and expression levels were detected by chemiluminescence [1] - PCR assay: Total RNA was extracted from human synovial fibroblasts treated with Pamapimod, reverse-transcribed into cDNA, and real-time quantitative PCR was used to detect the mRNA expression levels of MMP-1 and MMP-3 [1] - Cytotoxicity assay: HeLa cells were seeded in 96-well plates (5×10³ cells per well) and treated with Pamapimod at gradient concentrations of 0.1–10 μM. After 72 hours of culture, cell viability was detected by MTT assay to evaluate drug cytotoxicity [1] |
| Animal Protocol |
BALB/c mice
25, 50, 100, 150 mg/kg oral administration Systemic inflammation model establishment: C57BL/6 mice were intraperitoneally injected with LPS (10 mg/kg) to induce inflammation, and the treatment group was orally administered Pamapimod 1 hour before LPS injection [1] - Paw edema model establishment: SD rats were subcutaneously injected with 1% carrageenan solution (0.1 ml/rat) into the right hind paw to induce edema, and the treatment group was orally administered 1 hour before modeling [1] - Arthritis model establishment: Lewis rats were intradermally injected with complete Freund's adjuvant (0.1 ml/rat) into the right hind paw to induce AIA, and oral administration started on day 7 post-modeling [1] - Dosing regimen: A single dose of 10 mg/kg was used for the systemic inflammation model; the paw edema model and arthritis model were administered once daily at doses of 30 mg/kg and 20 mg/kg, respectively, for 3–14 consecutive days. Pamapimod was dissolved in a mixture containing 5% dimethyl sulfoxide, 10% polyethylene glycol 400, and 85% normal saline, and the control group was given an equal volume of vehicle [1] - Detection indicators: Serum TNF-α and IL-1β levels were detected at 2, 4, and 6 hours post-administration in the systemic inflammation model; paw volume was measured every 24 hours in the paw edema model; joint swelling scores were evaluated weekly in the arthritis model, and pathological changes of joint tissues were observed after the administration period [1] |
| ADME/Pharmacokinetics |
After oral administration to rats, Pamapimod was rapidly absorbed, with a peak time (Tmax) of 1.5-2 hours and an oral bioavailability of approximately 40% [1]. The plasma half-life (t1/2) was 6.2 hours, the steady-state volume of distribution (Vdss) was 1.3 L/kg, and the plasma clearance (CL) was 0.14 L/h/kg [1]. In vitro human liver microsomal metabolism experiments showed that Pamapimod was mainly metabolized by CYP2C9 and CYP3A4, with moderate metabolic stability (in vitro half-life = 2.5 hours) [1]. The drug concentration in inflamed joint tissues was approximately 2.8 times that in plasma, demonstrating good tissue targeting [1].
|
| Toxicity/Toxicokinetics |
In a 14-day rat toxicity study, rats taking palapimod orally once daily at doses up to 60 mg/kg showed normal weight gain (growth rate >85%), and no significant abnormalities in liver and kidney function (ALT, AST, creatinine, blood urea nitrogen) or routine blood indicators [1]. The plasma protein binding rate was approximately 95%, mainly bound to albumin, with no significant risk of plasma protein binding displacement [1]. No acute toxic reactions (such as vomiting, diarrhea, or abnormal activity) were observed after a single high-dose (100 mg/kg) oral administration [1].
|
| References | |
| Additional Infomation |
Pamapimod is a pyridopyrimidine compound with the structure 8-methylpyrido[2,3-d]pyrimidine-7(8H)-one, with (1,5-dihydroxypentan-3-yl)amino and 2,4-difluorophenoxy substituents at positions 2 and 6, respectively. It is a potent MAPK inhibitor used to treat rheumatoid arthritis. It is an EC 2.7.11.24 (mitogen-activated protein kinase) inhibitor and an antirheumatic drug. It is a pyridopyrimidine, aromatic ether, difluorobenzene, aromatic amine, secondary amine compound, primary alcohol, and diol. Pamapimod is a selective p38α/β MAPK small molecule inhibitor whose mechanism of action involves binding to the ATP-binding pocket of p38α kinase, inhibiting its phosphorylation activation, thereby blocking the production and release of downstream inflammatory factors (TNF-α, IL-1β)[1].
- It is mainly used in research on inflammatory diseases (including rheumatoid arthritis and sepsis) to reduce tissue damage by inhibiting the inflammatory response [1]. - Compared with other p38 subtypes and kinases, it has higher selectivity for p38α, which can reduce the risk of off-target toxicity [1]. - The drug has good oral absorption properties and targeting of inflamed tissues, and has significant in vivo anti-inflammatory activity, providing a potential option for the treatment of inflammatory diseases. Treatment of Inflammatory Diseases [1] |
| Molecular Formula |
C19H20F2N4O4
|
|
|---|---|---|
| Molecular Weight |
406.38
|
|
| Exact Mass |
406.145
|
|
| Elemental Analysis |
C, 56.15; H, 4.96; F, 9.35; N, 13.79; O, 15.75
|
|
| CAS # |
449811-01-2
|
|
| Related CAS # |
Pamapimod-d4;1246814-57-2
|
|
| PubChem CID |
16220188
|
|
| Appearance |
white solid powder
|
|
| LogP |
1.366
|
|
| Hydrogen Bond Donor Count |
3
|
|
| Hydrogen Bond Acceptor Count |
9
|
|
| Rotatable Bond Count |
8
|
|
| Heavy Atom Count |
29
|
|
| Complexity |
591
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
FC1C([H])=C(C([H])=C([H])C=1OC1C(N(C([H])([H])[H])C2C(C=1[H])=C([H])N=C(N=2)N([H])C([H])(C([H])([H])C([H])([H])O[H])C([H])([H])C([H])([H])O[H])=O)F
|
|
| InChi Key |
JYYLVUFNAHSSFE-UHFFFAOYSA-N
|
|
| InChi Code |
nChI=1S/C19H20F2N4O4/c1-25-17-11(10-22-19(24-17)23-13(4-6-26)5-7-27)8-16(18(25)28)29-15-3-2-12(20)9-14(15)21/h2-3,8-10,13,26-27H,4-7H2,1H3,(H,22,23,24)
|
|
| Chemical Name |
6-(2,4-difluorophenoxy)-2-(1,5-dihydroxypentan-3-ylamino)-8-methylpyrido[2,3-d]pyrimidin-7-one
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4608 mL | 12.3038 mL | 24.6075 mL | |
| 5 mM | 0.4922 mL | 2.4608 mL | 4.9215 mL | |
| 10 mM | 0.2461 mL | 1.2304 mL | 2.4608 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
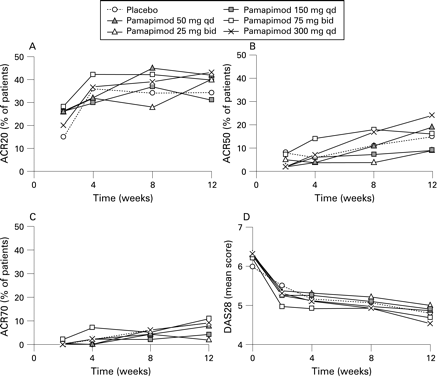 (A–C) Percentages of patients achieving a response according to the American College of Rheumatology criteria with improvements of 20%, 50% and 70% (ACR20, ACR50 and ACR70, respectively).Ann Rheum Dis.2010 Feb;69(2):364-7. |
|---|
Median change from baseline C-reactive protein.Ann Rheum Dis.2010 Feb;69(2):364-7. |