
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg | |||
| 100mg | |||
| 250mg | |||
| Other Sizes |
Purity: ≥98%
Nebivolol (R065824; R-065824; Nobiten; Vasoxen) is a potent and selective beta1/β1-adrenoceptor antagonist with antihypertensive effects. It suppresses the β1-adrenoceptor with an IC50 of 0.8 nM. Nebivolol is a beta blocker that has been used to treat heart failure and high blood pressure.
| ln Vitro |
Nebivolol has antioxidative and potent stimulatory effects on endothelial nitric oxide synthase activity, which improve endothelial dysfunction[1].
Nebivolol (0.1 μM–10 μM, 1, 2, 4, 7 or 14 days) inhibits the proliferation of haCSMC or haEC, with IC50 values of approximately 6.0 μM[2]. Nebivolol (0.1 μM-10 μM, 24 h) causes apoptosis at a moderate rate[2]. Nebivolol (0.1, 1, 5, 10 μM, 4 days) causes HaCEs to secrete less endothelin-1 and to produce more NO[2]. |
||
|---|---|---|---|
| ln Vivo |
|
||
| Cell Assay |
Nebivolol (10-7~10-5 M) is added to human coronary smooth muscle cells (haCSMCs) and endothelial cells (haECs) at varying concentrations for a period of 1, 2, 4, 7, and 14 days. Bromodeoxyuridine (BrdU) incorporation is used to analyze cell proliferation, while PI or annexin V staining is used to identify cell apoptosis.
|
||
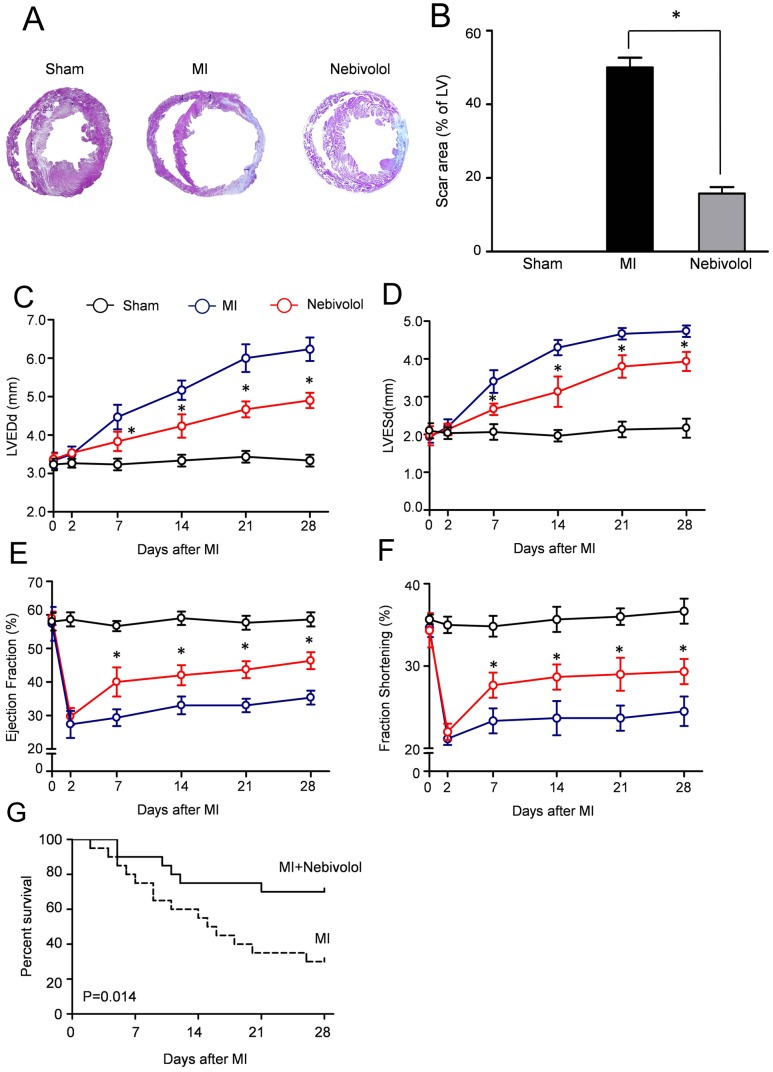
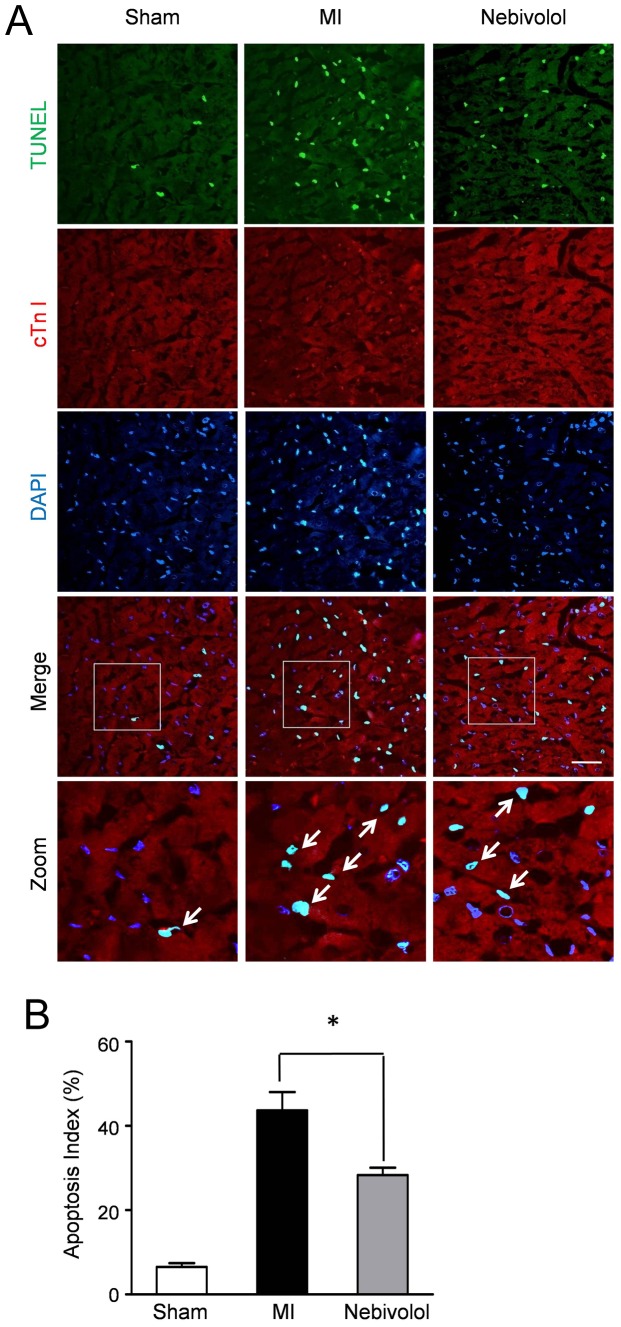
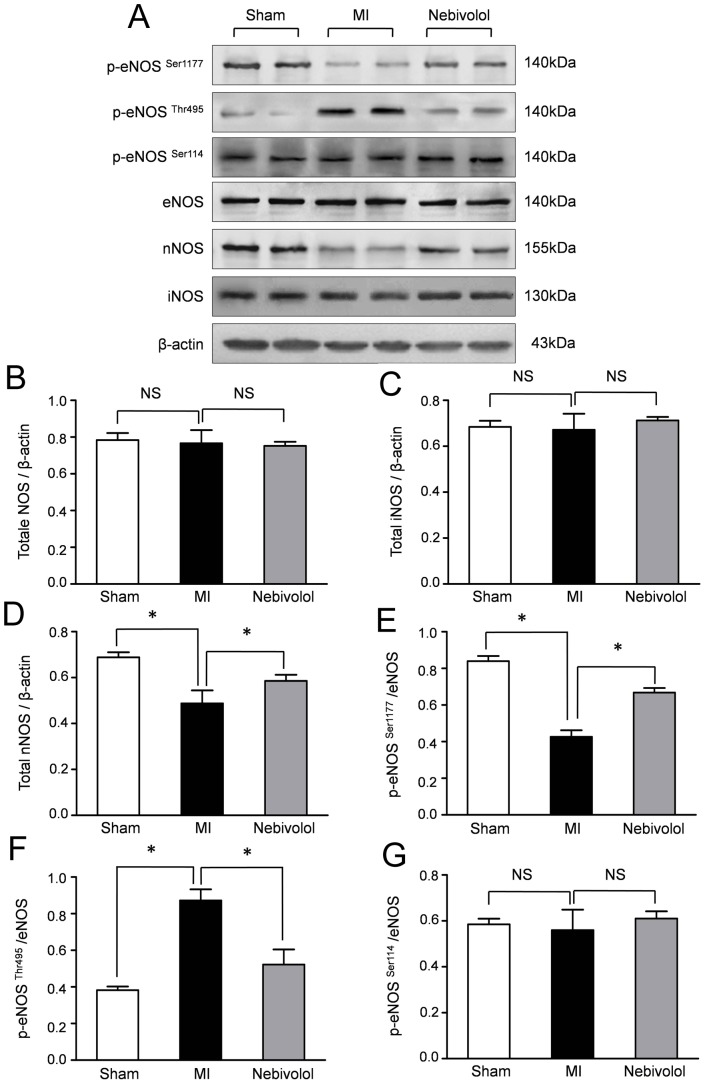
| Animal Protocol |
|
||
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Nebivolol absorption is not affected by food. The time to peak concentration (Tmax) of nebivolol is 1.5–4 hours. Its bioavailability ranges from 12–96% for individuals with varying CYP2D6 metabolic capacity. At a 20 mg dose, the Cmax of d-nebivolol is 2.75 ± 1.55 ng/mL, the Cmax of l-nebivolol is 5.29 ± 2.06 ng/mL, the Cmax of both enantiomers is 8.02 ± 3.47 ng/mL, and the Cmax of nebivolol glucuronide is 68.34 ± 44.68 ng/mL. For a 20 mg dose, the AUC of d-nebivolol was 13.78 ± 15.27 ngh/mL, the AUC of l-nebivolol was 27.72 ± 15.32 ngh/mL, the AUC of both enantiomers was 41.50 ± 29.76 ngh/mL, and the AUC of nebivolol glucuronide was 396.78 ± 297.94 ngh/mL. In individuals with high CYP2D6 metabolism, 38% of the drug was excreted in the urine and 44% in the feces. In individuals with low CYP2D6 metabolism, 67% of the drug was excreted in the urine and 13% in the feces. <1% of the dose was excreted as unmetabolized drug. At a dose of 20 mg, the apparent volume of distribution (VOD) of d-nebivolol was 10,290.81 ± 3911.72 L, that of l-nebivolol was 8,066.66 ± 4,055.50 L, and the total VOD of the two enantiomers was 10,423.42 ± 6796.50 L. At a dose of 20 mg, the clearance of d-nebivolol was 1241.63 ± 749.77 L/h, that of l-nebivolol was 435.53 ± 180.93 L/h, and the clearance of the two enantiomers was 635.31 ± 300.25 L/h. Metabolism/Metabolites Nebivolol is primarily metabolized via glucuronidation and CYP2D6-mediated hydroxylation. Metabolic processes include N-dealkylation, hydroxylation, oxidation, and glucuronidation. Aromatic hydroxylated and acyclic oxide metabolites are active, while N-dealkylated metabolites and glucuronides are inactive. Biological Half-Life D-Nebivolol has a half-life of 12 hours in CYP2D6 fast metabolizers and 19 hours in slow metabolizers. |
||
| Toxicity/Toxicokinetics |
Hepatotoxicity
In patients taking beta-blockers, the incidence of mild to moderate elevations in serum transaminase levels is less than 2%, usually transient and asymptomatic, and resolves with continued treatment. Information regarding the incidence of ALT elevations during nebivolol treatment is currently unavailable. Although nebivolol has been used in several large clinical trials, it has not been found to be associated with clinically significant liver injury. Probability score: E (Unlikely a cause of clinically significant liver injury). Pregnancy and Lactation Effects ◉ Overview of use during lactation Since there is currently no information regarding the use of nebivolol during lactation, alternative medications are recommended, especially for breastfeeding newborns or premature infants. ◉ Effects on breastfed infants No published information found as of the revision date. ◉ Effects on lactation and breast milk No published information found as of the revision date. Protein Binding Nebivolol has a 98% binding rate to plasma proteins, mainly binding to serum albumin. |
||
| References | |||
| Additional Infomation |
2,2'-Iminobis[1-(6-fluoro-3,4-dihydro-2H-chromen-2-yl)ethanol] belongs to the chromanoid class of compounds, with the structure 2,2'-iminodiethanol, in which one hydrogen atom on each hydroxyl carbon atom is replaced by a 6-fluorochroman-2-yl group. It is an organofluorine compound, a secondary amino compound, a secondary alcohol, a diol, and also a chromanoid compound. Nebivolol is a racemic mixture of two enantiomers, one a β-adrenergic antagonist and the other a cardiac stimulant without β-adrenergic activity. Compared to atenolol, propranolol, or indolol, nebivolol treatment significantly reduces systolic and diastolic blood pressure. Nebivolol and other β-blockers are often not first-line treatments because many patients first receive thiazide diuretics. Nebivolol was approved by the U.S. Food and Drug Administration (FDA) on December 17, 2007. Nebivolol is a beta-blocker and antihypertensive drug that exerts its additional vasodilatory effect by releasing nitric oxide. No clinically significant liver injury has been found associated with nebivolol. Nebivolol is a beta-adrenergic receptor antagonist with both antihypertensive and vasodilatory effects. Nebivolol binds to and blocks beta-adrenergic receptors in the heart, thereby reducing myocardial contractility and heart rate. This leads to a decrease in cardiac output, which in turn lowers blood pressure. Furthermore, nebivolol enhances the effects of nitric oxide (NO), thereby relaxing vascular smooth muscle and exerting a vasodilatory effect. Nebivolol is a cardiac-selective beta-adrenergic receptor antagonist (beta-blocker) that exerts its vasodilatory effect through the endothelial L-arginine/nitric oxide system. It is used to treat hypertension and chronic heart failure in elderly patients. See also: Nebivolol hydrochloride (salt form).
Indications Nebivolol is indicated for the treatment of hypertension. Mechanism of Action Nebivolol is a highly selective β1-adrenergic receptor antagonist with a weaker antagonistic effect on β2-adrenergic receptors. Denebivolol reduces resting heart rate, exercise heart rate, myocardial contractility, systolic blood pressure, and diastolic blood pressure by blocking β1-adrenergic receptors. The selectivity of denebivolol limits the adverse effects of β-blockers on airway or insulin sensitivity. Nebivolol also inhibits aldosterone, and its β1-receptor antagonism in the juxtaglomerular apparatus also inhibits renin release. Decreased aldosterone leads to reduced blood volume, and decreased renin leads to weakened vasoconstriction. Levonorbivolol, on the other hand, has β3-adrenergic receptor agonist activity, stimulating endothelial nitric oxide synthase, increasing nitric oxide levels, thereby leading to vasodilation, decreased peripheral vascular resistance, increased stroke volume, ejection fraction, and cardiac output. Nebivolol's vasodilatory, oxidative stress-reducing, and platelet-volume- and aggregation-reducing effects may benefit patients with heart failure. Pharmacodynamics Nebivolol is a selective β1-adrenergic receptor antagonist that reduces vascular resistance, increases stroke volume and cardiac output, and does not affect left ventricular function. It has a long duration of action, with efficacy still observable 48 hours after discontinuation; it has a wide therapeutic window, and patients typically take 5-40 mg daily. Patients should not abruptly discontinue the drug, as this may worsen coronary artery disease. Diabetic patients should monitor their blood glucose levels, as β-blockers may mask symptoms of hypoglycemia. |
| Molecular Formula |
C22H26CLF2NO4
|
|
|---|---|---|
| Molecular Weight |
441.9
|
|
| Exact Mass |
405.175
|
|
| Elemental Analysis |
C, 65.17; H, 6.22; F, 9.37; N, 3.45; O, 15.78
|
|
| CAS # |
118457-14-0
|
|
| Related CAS # |
Nebivolol hydrochloride; 152520-56-4; (Rac)-Nebivolol; 99200-09-6; (rac)-Nebivolol-d4; 1219407-55-2
|
|
| PubChem CID |
71301
|
|
| Appearance |
Solid powder
|
|
| Density |
1.3±0.1 g/cm3
|
|
| Boiling Point |
600.5±55.0 °C at 760 mmHg
|
|
| Melting Point |
223.0-228.0
|
|
| Flash Point |
316.9±31.5 °C
|
|
| Vapour Pressure |
0.0±1.8 mmHg at 25°C
|
|
| Index of Refraction |
1.581
|
|
| LogP |
3.67
|
|
| Hydrogen Bond Donor Count |
3
|
|
| Hydrogen Bond Acceptor Count |
7
|
|
| Rotatable Bond Count |
6
|
|
| Heavy Atom Count |
29
|
|
| Complexity |
483
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
FC1=CC=C2C(CC[C@]([C@@H](O)CNC[C@H](O)[C@@]3([H])CCC(C=C(F)C=C4)=C4O3)([H])O2)=C1
|
|
| InChi Key |
KOHIRBRYDXPAMZ-YHDSQAASSA-N
|
|
| InChi Code |
InChI=1S/C22H25F2NO4/c23-15-3-7-19-13(9-15)1-5-21(28-19)17(26)11-25-12-18(27)22-6-2-14-10-16(24)4-8-20(14)29-22/h3-4,7-10,17-18,21-22,25-27H,1-2,5-6,11-12H2/t17-,18-,21-,22+/m0/s1
|
|
| Chemical Name |
(1S)-1-[(2S)-6-fluoro-3,4-dihydro-2H-chromen-2-yl]-2-[[(2S)-2-[(2R)-6-fluoro-3,4-dihydro-2H-chromen-2-yl]-2-hydroxyethyl]amino]ethanol
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2630 mL | 11.3148 mL | 22.6296 mL | |
| 5 mM | 0.4526 mL | 2.2630 mL | 4.5259 mL | |
| 10 mM | 0.2263 mL | 1.1315 mL | 2.2630 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Determination of Drug Levels for Pharmacotherapy of Heart Failure
CTID: NCT06035978
Phase: Phase 4 Status: Not yet recruiting
Date: 2024-01-18
 |
|---|
 |
 |