
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
Purity: ≥98%
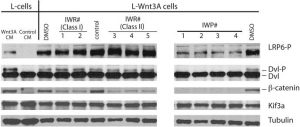
IWP-2 (IWP 2, IWP2) is a novel and potent inhibitor of Wnt processing and secretion (specifically, Porcn-mediated Wnt palmitoylation) with potential antineoplastic activity. It inhibits Wnt with an IC50 of 27 nM in a cell-free assay. IWP-2 showed selective blockage of Porcn-mediated Wnt palmitoylation, and does not affect Wnt/β-catenin in general; also it displays no effect against Wnt-stimulated cellular responses. IWP-2 can decrease MKN28 cell proliferation, migration and invasion, and elevate caspase 3/7 activity. IWP-2 can also downregulate the transcriptional activity of the Wnt/β-catenin signaling pathway and downregulate the expression levels of downstream Wnt/β-catenin target genes in MKN28 cells. IWP-2 may be a potential therapeutic option for gastric cancer.
| Targets |
CK1δ (IC5 = 40 nM)
IWP-2 is a selective ATP-competitive inhibitor of casein kinase 1 (CK1) δ and CK1 ε (CK1δ IC50 = 20 nM; CK1ε IC50 = 18 nM) [2] IWP-2 indirectly inhibits Wnt signaling pathway by targeting CK1δ/ε, with no significant inhibition of other kinases (CK1α, CK1γ, PKA, PKC: IC50 > 10 μM) [1][2] |
|---|---|
| ln Vitro |
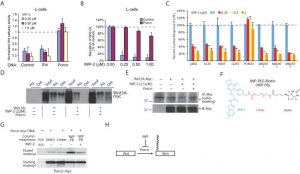
The tested cell lines' growth was decreased by IWP-2 within a single digit μM range. For A818-6, MiaPaCa2, Panc-1, Panc-89, HT29, HEK293, SW620, and Capan cells, IWP-2 reduces cell proliferation with EC50s of 8.96 μM, 1.90 μM, 2.33 μM, and 3.86 μM, 4.67 μM, 2.76, respectively. 1.90 μM, 2.05 μM, and 1.90 μM[2]. Panc-1 cells were either left untreated or given 2.33 μM IWP-2 for a 48-hour period. When compared to untreated cells, the peak activity of CK1δ kinase in IWP-2-treated cells was roughly 66% of residual activity. In Panc1 cells, IWP-2 decreases CK1δ activity [2].
In recombinant CK1δ/ε kinase assays, IWP-2 dose-dependently inhibits kinase activity, with 90% inhibition of CK1δ and 88% inhibition of CK1ε at 100 nM. It blocks ATP binding to CK1δ/ε, suppressing downstream Wnt signaling activation [2] - In HEK293 cells transfected with Wnt-responsive luciferase reporter plasmid, IWP-2 (1 μM) reduces Wnt3a-induced luciferase activity by 82% after 24 hours. It downregulates Wnt target genes (AXIN2, LEF1) at mRNA level by 65% and 58% respectively [1] - In mouse bone marrow-derived macrophages (BMDMs), IWP-2 (5 μM) inhibits Wnt5a-stimulated phagocytosis of fluorescent microspheres by 55% after 1 hour. It does not affect bacterial killing activity (E. coli survival rate unchanged compared to control) [3] - In normal HEK293 cells and BMDMs, IWP-2 shows low toxicity at concentrations up to 25 μM (cell viability > 85% vs. control) [1][3] |
| ln Vivo |
In order to assess the effectiveness of IWP-2 in vivo, C57BL/6 mice were intraperitoneally injected with 200 μL of either free liposomes or IWP-2-liposomes for around two hours, and then a comparable volume of blue dye filling was added. rubber beads or E. Coli DH5α. The uptake of blue beads and E was significantly reduced as a result of IWP-2. Coli as measured over a 2-hour period by CFU in peritoneal lavage cells. Moreover, the related mice's lavage fluid had 2-4 times lower levels of TNF-α and IL-6 than the control values. Even the release of the anti-inflammatory cytokine IL-10 is markedly increased by IWP-2 [3].
|
| Enzyme Assay |
In Vitro Kinase Assays[2]
In vitro kinase assays were performed with different CK1 isoforms and IWP derivatives (such as IWP-2) at an ATP concentration of 10 μM and using DMSO controls as described previously. Where indicated, higher ATP concentrations (50, 100, 250, and 500 μM) were used. Bovine GST-CK1α (FP296), rat recombinant CK1δ kinase domain (CK1δKD), rat GST-wtCK1δ (FP449), rat GST-M82FCK1δ (FP1153), recombinant human CK1ε, TLK2, and ZAP70 were used as sources of enzyme. Phosphorylated proteins were separated by SDS-PAGE and stained with Coomassie. α-Casein served as a substrate for most kinase assay reactions. Kinase assays performed with ZAP70 were done using poly(l-glutamic acid-l-tyrosine) as substrate. Phosphate incorporation was detected by autoradiography of dried gels. The phosphorylated protein bands were cut out and quantified by Cherenkov counting. Dose–response analyses were carried out using GraphPad Prism 6 statistical software. High-Throughput Kinase Profiling[2] The residual activity of 320 eukaryotic kinases was measured by ProQinase GmbH in the presence of compounds IWP-2 and 19 (1 μM). Dendrograms illustrating the phylogenetic relations of the kinases were generated using TREEspotTM Software Tool Image and reprinted with permission from KINOMEscan. CK1δ/ε kinase activity assay: Purified recombinant human CK1δ or CK1ε was incubated with synthetic peptide substrate (CK1-specific) and IWP-2 (0.1 nM-1 μM) in assay buffer (50 mM Tris-HCl, pH 7.5, 10 mM MgCl₂, 1 mM DTT, 0.1 mM ATP) at 30°C for 60 minutes. Phosphorylated substrate was detected by radiolabeled ATP counting, and IC50 values were calculated from dose-response curves [2] - ATP competition assay: CK1ε was incubated with increasing concentrations of ATP (0.05-1 mM) and fixed IWP-2 (18 nM). Kinase activity was measured to confirm competitive binding to the ATP-binding pocket of CK1ε [2] - Kinase selectivity assay: IWP-2 (10 μM) was screened against a panel of 40+ kinases (including CK1α, CK1γ, PKA, PKC, ERK1/2) using respective substrate peptides and assay buffers. Kinase activity was quantified by colorimetric method, with no significant inhibition (>50% activity reduction) observed for off-target kinases [2] |
| Cell Assay |
Cell Viability Assay[1]
Cells were seeded at a concentration of 5 × 104 cells/mL in 96-well cell culture plates and allowed to attach overnight at 37 °C and 5% CO2. To investigate the effects of compounds on cancer cell proliferation, cells were treated with various concentrations (ranging from 0.313 μM to 10 μM) of inhibitor, with untreated and DMSO-treated cells serving as a control. After an incubation period of 48 h at 37 °C, 10 μL of a MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) 12 mM solution in PBS were added, followed by further incubation for 4 h at 37 °C. Media containing MTT was then removed carefully, and 100 μL of 0.04 N HCl in isopropanol was added. To dissolve the formazan crystals, the plates were placed for 30 min on an orbital shaker. The resulting purple solution was spectrophotometrically measured at 570 nm. Experiments were repeated at least three times with four replicates per assay. Fractionation of Cell Extracts by FPLC.[1] IWP-2-treated (EC50 = 2,33 μM) and DMSO-treated Panc1 cells were lysed in sucrose lysis buffer. Total protein extract (1.4 mg) was diluted in prefiltered FPLC buffer A (50 mM Tris–HCl pH 7.5, 1 mM EDTA, 1 mM EGTA, 5% (v/v) glycerol, 0.03% (v/v) Brij-35, 1 mM benzamidine, 25 μg/mL aprotinin, 0.1% (v/v) β-mercaptoethanol). Cell lysates were then passed through a 0.45 μm filter and injected into an anion exchange column attached to an EttanLC FPLC system. Proteins bound to the cationic surfaces of the column were eluted with a linear ascending NaCl gradient by gradually increasing percentage of FPLC buffer B (equal to buffer A plus 1 M NaCl). Fractions of 250 μL volume were collected. Three μL from selected protein fractions were used for in vitro kinase assays, as described above, to determine CK1-specific kinase activity. To confirm CK1 in the peak fraction, kinase assays were repeated in the presence of given concentrations of CK1δ-specific inhibitors IC261 and PF670462. DMSO was used as a control and GST-p531–64 as substrate. Dual Luciferase Reporter Gene Assay[1] The assay was performed as described previously. Briefly, compounds were tested in HEK293T cells in a 96-well plate format. For transient transfection, Lipofectamine2000 and plasmids were pre-incubated in Opti-MEM medium for 15 min at room temperature. For the autocrine/paracrine assay setup, 3 × 106 cells (for one 96-well plate) were transfected with the Wnt3A-expressing vector and the Super(8x)TOPflash reporter vector together with the TK-Renilla luciferase control vector for internal luminescence normalization purposes, followed by incubation for 8 h. Transfected cells were harvested and seeded on 96-well plates in 110 μL media at 25,000 cells per well and allowed to adhere for 1 h. Cells were treated either with 10 μL of compound dilution (final concentrations 5, 2.5, 1, 0.5, 0.1, 0.05, 0.01, 0.001, 0.001 μM with 0.5% DMSO) or DMSO (0.5%) as the vehicle controls. For paracrine pathway activation, cells were stimulated by addition of Wnt3A-conditioned medium that was freshly harvested from mouse L-cells overexpressing the Wnt protein (L-Wnt3A). Control cells were treated with L-cell medium. Transfection of cells was done with the same vectors as described above, except for the Wnt3A-expressing vector. Cells were seeded in 80 μL and 25,000 cells/well 12 h post-transfection before stimulation with 30 μL of Wnt3A-conditioned medium. Compound treatment was conducted as described above. After 22 h incubation, the medium was carefully aspirated and both luciferase activities measured using the Dual-GloLuciferase Assay System according to the manufacturer’s protocol on a Tecan-Infinite 200 Plate Reader. Data were processed by normalizing Firefly with Renilla luciferase signals from each well. Each condition was repeated in technical triplicates with at least two independent biological replicates. EC50 and IC50 values were calculated by nonlinear regression analysis using the GraphPad Prism 5 software (version 5.03). Wnt reporter cell assay: HEK293 cells were seeded in 96-well plates at 5×10³ cells/well and transfected with β-catenin-responsive luciferase plasmid and Renilla luciferase plasmid (internal control). After 24 hours, cells were pretreated with IWP-2 (0.1-10 μM) for 1 hour, then stimulated with Wnt3a (50 ng/mL) for 24 hours. Dual-luciferase assay system was used to measure luciferase activity [1] - Macrophage phagocytosis assay: Mouse BMDMs were isolated and seeded in 24-well plates at 1×10⁵ cells/well. Cells were pretreated with IWP-2 (1-10 μM) for 1 hour, then incubated with fluorescently labeled microspheres (1 μm) and Wnt5a (100 ng/mL) for 1 hour. Phagocytosis was quantified by flow cytometry (fluorescence-positive cells percentage) [3] - Wnt target gene expression assay: HEK293 cells were seeded in 6-well plates at 2×10⁵ cells/well, pretreated with IWP-2 (1 μM) for 1 hour, then stimulated with Wnt3a (50 ng/mL) for 24 hours. Total RNA was extracted for qPCR analysis of AXIN2 and LEF1 mRNA levels [1] |
| Animal Protocol |
Mice: About 3-mo-old C57BL/6 mice are housed four to five in a cage at 23°C in a 12-h light/dark cycle. Mice are injected intraperitoneally (i.p.) first with either 200 μL of liposome-IWP-2 (LI) or liposome (L) and then after 2 h with 1×108 or 2×108 CFU E. coli in 200 μL of sterile PBS. After 2 h or 24 h mice are killed, and the peritoneal cavity is washed with 5 mL of sterile ice-cold PBS. The peritoneal lavage fluid is centrifuged at 300× g for 5 min, the cell pellet is resuspended in RPMI 1640 complete medium, and the supernatant is used for cytokine assay. For ex vivo experiments, peritoneal phagocytes are isolated as above from normal mice, and equal numbers of cells are plated in medium overnight at 37°C in 5% CO2 before performing further experiments.
Mice and rats Mice Infection. [3] About 3-mo-old C57BL/6 mice were were housed four to five in a cage at 23 °C in a 12-h light/dark cycle. Mice were injected intraperitoneally (i.p.) first with either 200 µL of liposome-IWP-2 (LI) or liposome (L) and then after 2 h with 1 × 108 or 2 × 108 CFU E. coli in 200 µL of sterile PBS. After 2 h or 24 h mice were killed, and the peritoneal cavity was washed with 5 mL of sterile ice-cold PBS. The peritoneal lavage fluid was centrifuged at 300 × g for 5 min, the cell pellet was resuspended in RPMI 1640 complete medium, and the supernatant was used for cytokine assay. For ex vivo experiments, peritoneal phagocytes were isolated as above from normal mice, and equal numbers of cells were plated in medium overnight at 37 °C in 5% CO2 before performing further experiments. Preparation of Liposome-IWP-2. [3] Liposome-IWP-2 was prepared with L-α-phosphatidylcholine, octadecylamine, and IWP-2 in a 20:2:0.1 ratio (100 µg of IWP-2 was used). The lipid mixture was dissolved in 1 mL of chloroform, and the solvent was evaporated under low pressure by a rotatory evaporator. The thin dry film was dispersed in 1 mL PBS, and the suspension was sonicated for 30 s twice in an ultrasonicator. Liposome with entrapped IWP-2 was separated from excess free drug by two successive washings in PBS with ultracentrifugation (100,000 × g, 30 min, and 4 °C). Control liposome was prepared similarly without adding IWP-2. [3] |
| References |
[1]. Chen B, et al. Small molecule-mediated disruption of Wnt-dependent signaling in tissue regeneration and cancer. Nat Chem Biol. 2009 Feb;5(2):100-7.
[2]. García-Reyes B, et al. Discovery of Inhibitor of Wnt Production 2 (IWP-2) and Related Compounds As Selective ATP-Competitive Inhibitors of Casein Kinase 1 (CK1) δ/ε. J Med Chem. 2018 May 10;61(9):4087-4102. [3]. Maiti G, et al. The Wingless homolog Wnt5a stimulates phagocytosis but not bacterial killing. Proc Natl Acad Sci U S A. 2012 Oct 9;109(41):16600-5 |
| Additional Infomation |
N-(6-methyl-1,3-benzothiazol-2-yl)-2-[(4-oxo-3-phenyl-6,7-dihydrothieno[3,2-d]pyrimidin-2-yl)thio]acetamide is an organic nitrogen heterocyclic compound, an organic heterobicyclic compound, and an organic sulfur heterocyclic compound. The extensive influence of secreted Wnt signaling proteins in tissue homeostasis and tumorigenesis has spurred efforts to identify small molecules that target Wnt-mediated cellular responses. By screening a diverse library of synthetic compounds, we discovered two classes of novel small molecules that interfere with Wnt pathway responses; one class inhibits the activity of porcupine, a membrane-bound acyltransferase crucial for Wnt protein synthesis; the other class prevents the degradation of axin protein, an inhibitor of Wnt/β-catenin pathway activity. Using these small molecules, we established a chemogenetic approach to study Wnt pathway responses and stem cell function in adult tissues. We achieved transient reversible inhibition of the Wnt/β-catenin signaling pathway response in vivo and established a mechanism-based method for targeting cancer cell growth. The signal transduction mechanism shown in this paper can not only be regulated by chemical methods, but also participates in the Wnt-independent signal transduction pathway, and therefore can be widely applied in the fields of chemogenetics and therapy. [1] Wnt signaling pathway inhibitors (IWPs) are known Wnt signaling pathway antagonists that target the membrane-bound O-acyltransferase porcupine protein (Porcn) to prevent palmitoylation of the key Wnt ligand. Since IWPs have structural similarity to benzimidazole CK1 inhibitors, we hypothesized that IWPs could also inhibit CK1 subtypes. Molecular modeling revealed a possible binding mode of IWP-2 in the ATP-binding pocket of CK1δ, which was confirmed by X-ray diffraction analysis. In vitro kinase activity assays showed that IWP is an ATP-competitive inhibitor of wtCK1δ. IWP also strongly inhibited the gated mutant M82FCK1δ. In an analysis of 320 kinases, IWP-2 specifically inhibited CK1δ. IWP-2 and IWP-4 also inhibited the viability of various cancer cell lines. We developed a modified IWP-derived CK1 inhibitor using medicinal chemistry. Our results suggest that the role of IWP is not limited to Porcn and may also affect the CK1δ/ε-related pathway. [2] Phagocytosis is a major defense mechanism coordinated by monocytes/macrophages. Dysregulation of phagocytosis can lead to pathological conditions. In this study, we demonstrated that Wnt5a stimulates phagocytosis through PI3 kinase-Rac1 and lipid raft-dependent processes. Blocking the expression of the putative Wnt5a receptor Frizzled 5 inhibited Wnt5a-mediated enhancement of phagocytosis. Enhanced bacterial phagocytosis by Wnt5a-Fz5 signaling increases the secretion of pro-inflammatory cytokines but does not increase bacterial kill rate. In addition, the small molecule inhibitor IWP-2 of the Wnt signaling pathway can reduce the secretion of functional Wnt5a, which not only inhibits phagocytosis and the secretion of pro-inflammatory cytokines, but also accelerates bacterial killing. [3]
IWP-2 is a selective CK1δ/ε small molecule inhibitor and a key regulator of the Wnt signaling pathway.[1][2] - Its mechanism of action involves competitive binding to the ATP-binding pocket of CK1δ/ε, inhibiting its kinase activity, thereby blocking the secretion of Wnt ligands and downstream signal transduction.[1][2] - IWP-2 has shown efficacy in inhibiting Wnt-dependent signaling in cancer cell and tissue regeneration models in vitro, and regulating Wnt5a-mediated phagocytosis in macrophages.[1][3] - It is widely used as a tool compound for studying the function of the Wnt signaling pathway. Development, tissue homeostasis, cancer and immune cell biology [1][2][3] - The high selectivity of IWP-2 for CK1δ/ε minimizes off-target effects, making it valuable for elucidating CK1-dependent Wnt signaling cascades [2] |
| Molecular Formula |
C22H18N4O2S3
|
|
|---|---|---|
| Molecular Weight |
466.6
|
|
| Exact Mass |
466.059
|
|
| Elemental Analysis |
C, 56.63; H, 3.89; N, 12.01; O, 6.86; S, 20.62
|
|
| CAS # |
686770-61-6
|
|
| Related CAS # |
IWP-2;686770-61-6
|
|
| PubChem CID |
2155128
|
|
| Appearance |
White to off-white solid powder
|
|
| Density |
1.5±0.1 g/cm3
|
|
| Melting Point |
257 °C(dec.)
|
|
| Index of Refraction |
1.787
|
|
| LogP |
5.25
|
|
| Hydrogen Bond Donor Count |
1
|
|
| Hydrogen Bond Acceptor Count |
7
|
|
| Rotatable Bond Count |
5
|
|
| Heavy Atom Count |
31
|
|
| Complexity |
796
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
S1C([H])([H])C([H])([H])C2=C1C(N(C1C([H])=C([H])C([H])=C([H])C=1[H])C(=N2)SC([H])([H])C(N([H])C1=NC2C([H])=C([H])C(C([H])([H])[H])=C([H])C=2S1)=O)=O
|
|
| InChi Key |
WRKPZSMRWPJJDH-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C22H18N4O2S3/c1-13-7-8-15-17(11-13)31-21(23-15)25-18(27)12-30-22-24-16-9-10-29-19(16)20(28)26(22)14-5-3-2-4-6-14/h2-8,11H,9-10,12H2,1H3,(H,23,25,27)
|
|
| Chemical Name |
N -(6-Methyl-2-benzothiazolyl)-2-[(3,4,6,7-tetrahydro-4-oxo-3-phenylthieno[3,2- d ]pyrimidin-2-yl)thio]-acetamide
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1432 mL | 10.7158 mL | 21.4316 mL | |
| 5 mM | 0.4286 mL | 2.1432 mL | 4.2863 mL | |
| 10 mM | 0.2143 mL | 1.0716 mL | 2.1432 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
 |
|---|
 |