| Size | Price | Stock | Qty |
|---|---|---|---|
| 2g |
|
||
| 5g |
|
||
| 10g |
|
||
| Other Sizes |
Purity: ≥98%
α-Lipoic Acid (Thioctic acid; (±)-α-Lipoic acid; DL-α-Lipoic acid) is a novel organosulfur compound which acts as an antioxidant, and is an essential cofactor of mitochondrial enzyme complexes. α-Lipoic Acid prevents NF-κB-dependent HIV-1 LTR activation.
| Targets |
Human Endogenous Metabolite; NF-κB; Mitochondrial bioenergetics; HIV-1
|
|---|---|
| ln Vitro |
The long terminal repeat (LTR) of HIV-1 is the target of cellular transcription factors such as NF-κB, and serves as the promoter-enhancer for the viral genome when integrated in host DNA[1]. The dithiol compound α-Lipoic Acid (Alpha-lipoic acid, ALA), which is a naturally occurring substance, is crucial to mitochondrial bioenergetics. By controlling the transcriptional factors SREBP-1, FoxO1, and Nrf2 and their downstream lipogenic targets through the activation of the SIRT1/LKB1/AMPK pathway, α-Lipoic Acid reduces lipid accumulation in the liver. The NAD+/NADH ratio in HepG2 cells is markedly elevated following treatment with -lipoic acid (250, 500, and 1000 μM) (P<0.05 or P<0.01). In HepG2 cells, treatment with -lipoic acid (50, 125, 250, and 500 μM) increases SIRT1 activity. In HepG2 cells, α-Lipoic Acid (50, 125, 250, 500, and 1000 μM) increases AMPK and acetyl-CoA carboxylase (ACC) phosphorylation in a dose-dependent manner[1].
Hepatocellular carcinoma (HCC) is the most common liver cancer and a major cause of adult death. The current treatments for HCC suffer from drug resistance and poor prognosis; therefore, novel therapeutic agents are urgently needed. Phytochemicals have been proposed to treat a range of cancers. Among them, α-lipoic acid (α-LA), a naturally synthesized antioxidant found in various dietary animal and plant sources, prevents oxidant-mediated cell death in normal cells while inducing apoptosis in several cancer cell lines. Previously, we demonstrated that the treatment of hepatoma cells with α-LA induced apoptosis, which was preceded by the generation of reactive oxygen species (ROS) and activation of the p53 protein, a known inducer of mitochondria-mediated apoptosis. Several studies have shown that ROS-induced apoptosis is associated with endoplasmic reticulum (ER) stress and Unfolded Protein Response (UPR) activation. Herein, we investigated if α-LA-induced apoptosis in hepatoma cell lines was ER stress- and UPR-mediated by gene expression profiling analyses. UPR and ER stress pathways were the most up-regulated after treatment with α-LA. This finding, which has been confirmed by expression analyses of ER- and UPR-associated proteins, provides a better understanding of the molecular mechanisms behind the anti-tumoral action of α-LA on hepatoma cells. [4] Three self-assembled nanoaggregates (CPUL1-LA NAs, CPUL1-DA NAs, and CPUL1-AA NAs) were constructed through lipoic acid (LA), dithiodipropionic acid (DA), and adipic acid (AA) decorated TrxR inhibitor (CPUL1), respectively. Measurements of DLS, TEM, UV-vis, fluorescence, 1H NMR, ITC, and MTT assays verified disulfide-containing CPUL1-LA NAs and CPUL1-DA NAs spontaneously assembled carrier-free nanoparticles in aqueous solution, which possessed high drug contents, excellent stability, improved cytotoxicity against HUH7 hepatoma cells, and potential biosafety because of low cytotoxicity against L02 normal cells. In contrast, disulfide-free CPUL1-AA NAs happened to aggregate and precipitate after 48 h, which showed distinct instability in aqueous solution. Thus, disulfide units seemed to be crucial for constructing controllable and stable nanoaggregates. While measuring the reduction of nanoaggregates by TrxR/NADPH and GSH/GR/NADPH, cyclic disulfide of LA and linear disulfide of DA were verified to endow the nanoaggregates with targeting ability to respond specifically to TrxR over GSH. Furthermore, by tests of flow cytometry, fluorescence images, and CLSM, both CPUL1-LA NAs and CPUL1-DA NAs displayed a faster cellular uptake characteristic to be internalized by cancer cells and could generate more abundant ROS to induce cell apoptosis than that of free CPUL1, resulting in significantly improved antitumor efficacy against HUH7 cells in vitro [5]. |
| ln Vivo |
In order to cause nonalcoholic fatty liver disease (NAFLD), C57BL/6J mice are divided into four groups and fed a high-fat diet (HFD) for 24 weeks. Each group is then given daily administration of α-lipoic acid. The effects of α-Lipoic Acid are then examined in long-term HFD-fed mice with regard to hepatic lipid accumulation. Mice with visceral fat mass are significantly lessened after receiving 100 mg/kg or 200 mg/kg of -lipoic acid. Additionally, α-Lipoic Acid (100 mg/kg or 200 mg/kg) treatment reduces appetite and results in significant weight loss (all P0.05)[1].
Understanding the mechanism by which alpha-lipoic acid supplementation has a protective effect upon nonalcoholic fatty liver disease in vivo and in vitro may lead to targets for preventing hepatic steatosis. Male C57BL/6J mice were fed a normal diet, high-fat diet or high-fat diet supplemented with alpha-lipoic acid for 24 weeks. HepG2 cells were incubated with normal medium, palmitate or alpha-lipoic acid. The lipid-lowering effects were measured. The protein expression and distribution were analyzed by Western blot, immunoprecipitation and immunofluorescence, respectively. We found that alpha-lipoic acid enhanced sirtuin 1 deacetylase activity through liver kinase B1 and stimulated AMP-activated protein kinase. By activating the sirtuin 1/liver kinase B1/AMP-activated protein kinase pathway, the translocation of sterol regulatory element-binding protein-1 into the nucleus and forkhead box O1 into the cytoplasm was prevented. Alpha-lipoic acid increased adipose triacylglycerol lipase expression and decreased fatty acid synthase abundance. In in vivo and in vitro studies, alpha-lipoic acid also increased nuclear NF-E2-related factor 2 levels and downstream target amounts via the sirtuin 1 pathway. Alpha-lipoic acid eventually reduced intrahepatic and serum triglyceride content. The protective effects of alpha-lipoic acid on hepatic steatosis appear to be associated with the transcription factors sterol regulatory element-binding protein-1, forkhead box O1 and NF-E2-related factor 2 [3]. |
| Cell Assay |
The human hepatocellular carcinoma (HepG2) cell line is grown in Dulbecco's modified Eagle's medium at 37°C and 5% CO2 with 10% fetal bovine serum. The following substances are applied to HepG2 cells: AMPK inhibitor (CC, 20 μM, 0.5 h), SIRT1 inhibitor (NA, 10 mM, 12 or 24 h), AMPK activator (AICAR, 2 mM, 1 h), palmitate (PA, 125 μM, 12 h), and -Lipoic Acid (250 μM, 6 or 12 h)[1].
Cell lines [4] The rat hepatoma cell line, FaO, and the hepatocarcinoma cell line, HepG2, were maintained, respectively, in Dulbecco’s medium (DMEM plus Glutamax I) and supplemented with penicillin, streptomycin and 10% heat-inactivated fetal calf-serum (FCS) in a humidified atmosphere of 5% CO2/95% air, at 37 °C. α-lipoic acid (α-LA) and Thapsigargin (TG) were purchased from xxx. α-LA, dissolved in sodium hydroxide NaOH 1 N and neutralized in medium, and TG dissolved in DMSO, were added to the culture media to the final concentrations specified in the text. Morphological assessment of apoptosis [4] Morphological assessment and detection of apoptotic cells was performed using Hoechst 33258 staining. FaO cells (2 × 105 cells/well) were plated in chamber-slides and cultured in the presence or absence of α-LA/lipoic acid. After treatment, the cells were fixed with 2% paraformaldehyde and stained with Hoescht 33258. Stained cells were visualized under a Leica DM2000 fluorescence microscope and images were acquired with a digital camera Leica DCF420C. |
| Animal Protocol |
Mice: Male C57BL/6J mice (6 weeks old; body weight: 22-24 g) are divided into four groups (n=8) and given access to a normal diet and water ad libitum for two weeks. These groups are: normal diet (ND) (10% energy from fat), high-fat diet (HFD) (60% energy from fat), and HFD plus α-Lipoic Acid (100 mg/kg or 200 mg/kg). After the mice's eyes are removed for the preparation of the serum, blood samples are taken 24 weeks after the start of treatment. Centrifugation at 2000×g for 10 min. at 4 °C is used to separate the serum. Harvested in liquid nitrogen and kept at -80°C are the liver tissues.
Male C57BL/6J mice (6-week-old; body weight: 22–24 g) were housed in standard cage conditions at a constant temperature (22±1°C) and a 13:11-h light/dark cycle. All mice were allowed ad libitum access to normal diet and water for 2 weeks before dividing into four groups (n=8): normal diet (ND) (10% energy from fat; D12450B), high-fat diet (HFD) (60% energy from fat) and HFD plus ALA (100 mg/kg or 200 mg/kg). These doses of ALA/lipoic acid were selected to be similar to previous studies. After 24 weeks of treatment, blood samples were collected after the eyeballs of the mice were extracted for serum preparation by centrifugation at 2000×g for 10 min at 4°C. The liver tissues were harvested in liquid nitrogen and stored at −80°C. [3] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
This study aimed to determine the concentration of α-lipoic acid in aqueous humor and to explore whether topical eye drops could increase its content. Methods: Seventy patients scheduled for cataract surgery were randomly divided into two groups. Group 1 served as the control group; Group 2 received a single instillation of 1% α-lipoic acid eye drops. 40-120 μL of aqueous humor was collected immediately before surgery. The aqueous humor samples from each group were mixed to form sample pools representing different dosing intervals. The concentration of α-lipoic acid in the aqueous humor was determined using gas chromatography/mass spectrometry. Aqueous humor samples from Group 1 (control group) patients were mixed to form sample pool 0, with an α-lipoic acid concentration of 27.5 ± 2.6 ng/mL. In the other sample pools, the time intervals from eye drop administration to sampling were 23 minutes, 53 minutes, 72 minutes, 93 minutes, and 114 minutes, with α-lipoic acid concentrations of 33.0 ± 10.8 ng/mL, 52.0 ± 2.5 ng/mL, 86.7 ± 2.5 ng/mL, 91.2 ± 2.5 ng/mL, and 80.3 ± 2.5 ng/mL, respectively. This study indicates the presence of α-lipoic acid in aqueous humor, and its concentration increases after eye drop administration, peaking at approximately 93 minutes. The concentration reached in the anterior chamber allows us to speculate on the potential for utilizing the antioxidant properties of α-lipoic acid. R(+)-α-lipoic acid is a natural compound and an essential cofactor for certain dehydrogenase complexes. The α-lipoic acid/dihydrolipoic acid redox pair exhibits potent antioxidant activity. When exogenous racemic α-lipoic acid is administered orally for symptomatic treatment of diabetic polyneuropathy, its absorption is rapid and almost complete; however, due to high liver extraction rates, its absolute bioavailability is only about 30%. Although the pharmacokinetics of the parent drug in humans are well-studied, little is known about the excretion of α-lipoic acid and the pharmacokinetics of its metabolites. This study evaluated plasma concentration-time curves, urinary excretion, and pharmacokinetic parameters in nine healthy volunteers after a once-daily oral administration of 600 mg of racemic α-lipoic acid. Using a newly established high-performance liquid chromatography-electrochemical analysis method prior to this study, the main metabolic pathways of α-lipoic acid in humans were quantitatively confirmed: S-methylation and β-oxidation. The main circulating metabolites are the S-methylated β-oxidation products 4,6-dimethylthiohexanoic acid and 2,4-dimethylthiobutanoic acid, while their conjugates account for the majority of urinary excretion. There were no statistically significant differences in the pharmacokinetic parameters Cmax, AUC, and tmax between Day 1 and Day 4. Although the half-life of the major metabolites was prolonged compared to the parent drug, no accumulation was observed. Twenty-four hours after administration, the average total amount of α-lipoic acid and its metabolites recovered in urine was 12.4% of the administered dose. These results indicate that urinary excretion of α-lipoic acid and its five major metabolites does not play a major role in the clearance of α-lipoic acid. Therefore, biliary excretion, further electrochemically inert degradation products, and complete utilization of α-lipoic acid as a major substrate for endogenous metabolism should be considered. In an open-label, parallel-group study involving 16 patients (8 with severe renal impairment and 8 with end-stage renal disease requiring hemodialysis), the impact of renal function on the pharmacokinetics, metabolism, and safety of α-lipoic acid (lipoic acid) was assessed by comparing pharmacokinetic parameters with those of 8 healthy controls. α-Lipoic acid was administered orally once daily for four consecutive days, with pharmacokinetic parameters determined on days 1 and 4. The mean percentages of α-lipoic acid excreted in the urine of healthy subjects and subjects with severe renal impairment were 0.2% and 0.05%, respectively. Assuming a bioavailability of 30%, this corresponds to 0.67% and 0.17% of the bioavailable α-lipoic acid, respectively. The percentage of α-lipoic acid and its five metabolites recovered in the urine over both days was 12.0%. The corresponding values for patients with severe renal impairment were 5.2% (day 1) and 6.4% (day 4), respectively. The total percentage of the administered dose cleared by hemodialysis in patients with end-stage renal disease was 4.0%. Compared with the control group, patients with renal impairment showed significantly reduced renal clearance of α-lipoic acid and its major metabolites, 6,8-dimethylthiooctanoic acid, 4,6-dimethylthiohexanoic acid, and 2,4-dimethylthiobutanoic acid. The apparent total clearance of alpha-lipoic acid (ALA) correlated poorly with creatinine clearance. Strong evidence suggests that ALA is primarily excreted via non-renal routes or further degraded into smaller molecules during catabolism. In patients with severely impaired renal function and end-stage renal disease, the area under the curve (AUC) of 4,6-dimethylthiohexanoic acid (4,6-Dimethylthiobutanoic acid) and the half-life of 2,4-dimethylthiobutanoic acid (2,4-Dimethylthiobutanoic acid) were significantly increased on both test days, but these changes were not clinically significant. Although the trough concentrations of both metabolites were slightly elevated in these patients, no accumulation effect was observed. We conclude that the pharmacokinetics of ALA are unaffected by creatinine clearance and remain unaffected in patients with severely impaired renal function or end-stage renal disease. Hemodialysis did not significantly affect the clearance of ALA. Therefore, no dose adjustment of ALA is necessary in patients with renal impairment. After absorption in the small intestine, ALA is distributed to the liver via the portal vein and then to all tissues throughout the body via systemic circulation. The natural R-enantiomer is more readily absorbed and has higher activity than the L-enantiomer. α-Lipoic acid readily crosses the blood-brain barrier. After being distributed in various body tissues, α-lipoic acid exists intracellularly, within mitochondria, and extracellularly. Metabolism/Metabolites α-lipoic acid is metabolized by mitochondrial lipoamide dehydrogenase to reduced dihydrolipoic acid. Dihydrolipoic acid forms a redox pair with lipoic acid. It is also metabolized to lipoamide, which, as a cofactor of lipoic acid, participates in a multi-enzyme complex catalyzing the oxidative decarboxylation of pyruvate and α-ketoglutarate. α-Lipoic acid can be metabolized to dithiol octanoate, which can undergo catabolism. The excretion and biotransformation of racemic alpha-lipoic acid (LA), used to treat symptoms of diabetic polyneuropathy, were investigated by single oral administration of [(14)C]LA to mice (30 mg/kg), rats (30 mg/kg), and dogs (10 mg/kg), and by a single oral administration of unlabeled LA to humans (600 mg). Over 80% of the radioactive material is excreted via the kidneys. Metabolite profiles obtained by radioactive high-performance liquid chromatography (HPLC) showed that LA is extensively metabolized in different species. Based on a novel negative-ion online liquid chromatography/tandem mass spectrometry (LC-MS) method, LA and 12 of its metabolites were identified. Mitochondrial β-oxidation plays a crucial role in the metabolism of LA. Simultaneously, the circulating metabolites underwent reduction of the 1,2-dithionylpentane ring and subsequent S-methylation. Furthermore, it was confirmed for the first time that the generated methyl sulfide portion was oxidized to sulfoxides, primarily in dogs. The disulfoxide of 2,4-dimethyl mercaptobutyric acid is the most polar metabolite identified and is also the major metabolite in dogs. Furthermore, the new data indicate that binding to glycine is an independent metabolic pathway that competes with β-oxidation and primarily occurs in mice. |
| Toxicity/Toxicokinetics |
Toxicity Overview
ALA is generally considered a safe medication. Daily doses of 200 mg to 2400 mg of ALA are considered safe without significant side effects. However, there are currently no reports of safe doses for children. One notable case has been reported in the literature, presenting with status epilepticus (SE), which resolved within a few days. The seizures were treated according to standard criteria for status epilepticus. Reports of ALA poisoning have been rare in the past 20 years. These cases have mostly occurred in children and are treatable. While there is currently no definitive lethal dose of ALA in humans, studies have shown that high doses of 121 mg/kg body weight daily are associated with alterations in liver enzymes and liver function. Therefore, ALA overdose may pose potential harmful side effects, and further research is needed to determine its toxicity. (R)-Lipoic acid is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Cholinesterase inhibitors (or "anticholinesterases") inhibit the activity of acetylcholinesterase. Because acetylcholinesterase plays a crucial role, chemicals that interfere with its activity are potent neurotoxins. Even low doses can cause excessive salivation and tearing, followed by muscle spasms and ultimately death. Neurotoxins and substances in many pesticides have been shown to exert their effects by binding to serine residues at the active site of acetylcholinesterase, thus completely inhibiting the enzyme's activity. Acetylcholinesterase breaks down the neurotransmitter acetylcholine, which is released at the neuromuscular junction, causing muscle or organ relaxation. Inhibition of acetylcholinesterase results in the accumulation and sustained action of acetylcholine, leading to continuous nerve impulse transmission and an inability to stop muscle contractions. The most common acetylcholinesterase inhibitors are phosphorus-containing compounds designed to bind to the enzyme's active site. Their structure requires a phosphorus atom with two lipophilic groups, a leaving group (such as a halide or thiocyanate), and a terminal oxygen atom. Health Effects Acute exposure to cholinesterase inhibitors can cause cholinergic crisis, characterized by severe nausea/vomiting, salivation, sweating, bradycardia, hypotension, collapse, and convulsions. Increased muscle weakness is also possible, and if respiratory muscles are involved, it can lead to death. The accumulation of acetylcholine in motor nerves can lead to overactivation of nicotine receptors at the neuromuscular junction. This results in symptoms such as muscle weakness, fatigue, muscle spasms, fasciculations, and paralysis. When acetylcholine accumulates in autonomic ganglia, it leads to overactivation of nicotine receptors in the sympathetic nervous system. Related symptoms include hypertension and hypoglycemia. Due to the accumulation of acetylcholine (ACh), over-excitation of nicotine acetylcholine receptors in the central nervous system can lead to anxiety, headache, convulsions, ataxia, respiratory and circulatory depression, tremors, general weakness, and even coma. When there is an excess of acetylcholine on muscarinic acetylcholine receptors, leading to over-excitation of these receptors, symptoms such as visual disturbances, chest tightness, wheezing due to bronchoconstriction, increased bronchial secretions, increased salivation, lacrimation, sweating, intestinal peristalsis, and urination may occur. Certain reproductive system effects, such as male and female fertility and growth and development, have been proven to be associated with organophosphate pesticide exposure. Currently, most research on reproductive system effects focuses on farmers in rural areas who use pesticides and insecticides. In women, menstrual cycle disorders, prolonged pregnancy, spontaneous abortion, stillbirth, and some developmental problems in offspring are all associated with organophosphate pesticide exposure. Prenatal exposure is associated with impaired fetal growth and development. Neurotoxicity is also associated with organophosphate pesticide poisoning, leading to four types of neurotoxic effects in humans: cholinergic syndrome, intermediate syndrome, organophosphate-induced delayed polyneuropathy (OPIDP), and chronic organophosphate-induced neuropsychiatric disorder (COPIND). These syndromes are caused by acute or chronic exposure to organophosphate pesticides. Symptoms Symptoms of low-dose exposure include excessive salivation and lacrimation. Symptoms of acute doses include severe nausea/vomiting, salivation, sweating, bradycardia, hypotension, collapse, and convulsions. Worsening muscle weakness is also possible, which can be fatal if respiratory muscles are involved. Hypertension, hypoglycemia, anxiety, headache, tremor, and ataxia may also occur. Treatment If the compound is ingested, rapid gastric lavage with a 5% sodium bicarbonate solution should be performed immediately. For skin contact, wash immediately with soap and water. If the compound gets into the eyes, flush immediately with copious amounts of isotonic saline or water. In severe cases, atropine and/or pralidoxime should be used. Anticholinergic drugs work by counteracting excess acetylcholine and reactivating acetylcholinesterase (AChE). Atropine can be used as an antidote in combination with pralidoxime or other pyridoximes (such as trimedodolite or olbidoxime), but at least two meta-analyses have shown that the use of "-oxime" drugs is not beneficial and may even be harmful. Atropine is a muscarinic receptor antagonist, thus blocking the peripheral effects of acetylcholine. |
| References |
|
| Additional Infomation |
Therapeutic Uses
/Experimental Treatment/ This trial aimed to evaluate the effects of alpha-lipoic acid (ALA) on sensory-positive symptoms and neuropathic deficits in patients with diabetic distal symmetrical polyneuropathy (DSP). In this multicenter, randomized, double-blind, placebo-controlled trial, 181 diabetic patients from Russia and Israel received once-daily oral administration of 600 mg (n = 45) (ALA600), 1200 mg (n = 47) (ALA1200), and 1800 mg (ALA1800) of ALA (n = 46) or placebo (n = 43) after a 1-week placebo introduction period for 5 weeks. The primary efficacy endpoint was the change from baseline in the total symptom score (TSS), including tingling, burning, paresthesia, and numbness in the foot. Secondary endpoints included individual TSS symptoms, neuropathy symptoms and changes (NSC) score, neuropathy impairment score (NIS), and overall patient feedback on efficacy. There were no significant differences in mean TSS at baseline among the treatment groups. Compared with the placebo group, the ALA600 group saw a mean reduction of 4.9 points (51%), the ALA1200 group a reduction of 4.5 points (48%), and the ALA1800 group a reduction of 4.7 points (52%), while the placebo group saw only a reduction of 2.9 points (32%) (all P values <0.05, compared with placebo). The corresponding remission rates (TSS reduction ≥50%) were 62%, 50%, 56%, and 26%, respectively. All three ALA groups showed significant improvements in tingling and burning pain, NSC scores, and overall patient assessment of efficacy. NIS scores were also reduced. Safety analysis showed a dose-dependent increase in the incidence of nausea, vomiting, and dizziness. Conclusion: Oral ALA treatment for 5 weeks improves neuropathic symptoms and functional impairment in patients with DSP. A once-daily oral dose of 600 mg appears to have the best risk-benefit ratio. /Experimental Therapy/ Mitochondria produce reactive oxygen species, which may lead to vascular dysfunction. Alpha-lipoic acid (ALA) and acetyl-L-carnitine can reduce oxidative stress and improve mitochondrial function. In a double-blind crossover study, the authors investigated the effects of ALA/acetyl-L-carnitine combination therapy and placebo (each treatment regimen lasting 8 weeks) on vasodilation and blood pressure in 36 patients with coronary artery disease. Active therapy increased brachial artery diameter by 2.3% (P=0.008), consistent with a decrease in arterial tone. Active therapy trended towards lowering systolic blood pressure in all subjects (P=0.07), and showed significant efficacy in subgroups with blood pressure above the median (151±20 mmHg to 142±18 mmHg; P=0.03) and subgroups with metabolic syndrome (139±21 mmHg to 130±18 mmHg; P=0.03). Therefore, mitochondrial dysfunction may be related to the regulation of blood pressure and vascular tone… /Experimental Therapy/ Lipoic acid is an antioxidant that can inhibit and treat an animal model of multiple sclerosis—experimental autoimmune encephalomyelitis. This study aimed to determine the pharmacokinetics (PK), tolerability, and effects on matrix metalloproteinase-9 (MMP-9) and soluble intercellular adhesion molecule-1 (sICAMP-1) of oral lipoic acid in patients with multiple sclerosis. Thirty-seven patients with multiple sclerosis were randomly assigned to four groups: placebo, lipoic acid 600 mg twice daily, lipoic acid 1200 mg once daily, and lipoic acid 1200 mg twice daily. Subjects took the study capsule for 14 days. The peak serum lipoic acid levels in subjects taking 1200 mg of lipoic acid were significantly higher than those taking 600 mg of lipoic acid, and the peak levels varied considerably among different subjects. We also found a significant negative correlation between peak serum lipoic acid levels and the mean change in serum MMP-9 levels (T = -0.263, P = 0.04). A significant dose-response relationship was also found between lipoic acid and the mean change in serum sICAM-1 levels (P = 0.03). Oral lipoic acid was generally well-tolerated and appeared to reduce serum MMP-9 and sICAM-1 levels. Lipoic acid may play a role in the treatment of multiple sclerosis by inhibiting MMP-9 activity and interfering with T cell migration to the central nervous system. /Experimental Therapies/ Mitochondrial dysfunction and oxidative damage are closely related to the pathogenesis of Parkinson's disease. Several mitochondrial antioxidants/nutrients that can improve mitochondrial function and/or reduce oxidative damage have been used to treat Parkinson's disease. However, few studies have evaluated the preventive effects of combined use of mitochondrial antioxidants/nutrients on Parkinson's disease, and even fewer studies have optimized the dosage of combined medications. This study investigated the preventive effects of two mitochondrial antioxidants/nutrients—R-α-lipoic acid (LA) and acetyl-L-carnitine (ALC)—in a chronic rotenone-induced Parkinson's disease cell model. Results showed that 4 weeks of pretreatment with LA and/or ALC effectively protected SK-N-MC human neuroblastoma cells from rotenone-induced mitochondrial dysfunction, oxidative damage, and the accumulation of α-synuclein and ubiquitin. Most notably, we found that the effective concentration of LA and ALC used in combination was 100 to 1000 times lower than that of either alone. We also found that combined pretreatment with LA and ALC increased mitochondrial biosynthesis and reduced reactive oxygen species production by upregulating peroxisome proliferator-activated receptor γ coactivator 1α (PPARγ 1α), which may be its potential mechanism. This study provides important evidence that combined use of mitochondrial antioxidants/nutrients at optimal doses may be an effective and safe strategy for the prevention of Parkinson's disease. /R-α-Lipoic Acid/ For more complete data on the therapeutic uses of α-lipoic acid (11 in total), please visit the HSDB record page. Drug Warnings Patients with diabetes and those with impaired glucose tolerance should be aware that α-lipoic acid supplementation may lower blood glucose levels.Blood glucose should be monitored, and the dosage of hypoglycemic agents should be adjusted as needed to avoid possible hypoglycemia. Due to a lack of long-term safety data, pregnant and lactating women should avoid using α-lipoic acid. The “long terminal repeat” (LTR) of HIV-1 is a target of cellular transcription factors such as NF-κB. After integration into the host DNA, the LTR acts as a promoter-enhancer of the viral genome. Various LTR reporter gene constructs have been used in vitro to study activators or inhibitors of HIV-1 transcription, for example, to demonstrate that antioxidants such as lipoic acid and selenium inhibit NF-κB-dependent HIV-1 LTR activation. One construct is the pHIVlacZ plasmid, in which HIV-1 LTR drives the expression of the lacZ gene (encoding β-galactosidase, β-gal). Typically, inhibitor screening involves activating cells transfected with pHIVlacZ with tumor necrosis factor-α (TNF-α) and then assessing changes in β-galactosidase activity using a colorimetric o-nitrophenol assay. This article describes a variant of this assay in which LTR activation is induced by pro-fs, a novel HIV-1 gene product encoded by a -1 frameshift of the protease gene. Co-transfection of cells with the pHIVlacZ and pro-fs construct significantly increased β-galactosidase activity. L-ergothionein inhibited the TNF-α and pro-fs-mediated increase in β-galactosidase activity in a dose-dependent manner, with an IC50 of approximately 6 mM. Therefore, an antioxidant strategy utilizing ergothionein, derived from edible plants, may be beneficial for chronic immunodeficiency diseases. [1] Retinal ischemia-reperfusion (RIR) injury leads to neuronal degeneration and triggers various optic nerve diseases. This study aimed to investigate the synergistic neuroprotective effects of rasagiline and idebenone on retinal ischemia-reperfusion injury (RIR). After establishing the RIR model, rasagiline and idebenone were administered intraperitoneally immediately. The combined treatment with the two drugs significantly restored retinal thickness and the number of retinal ganglion cells. Apoptosis in the ganglion cell layer was also alleviated, indicating a synergistic effect between the two drugs, and the expression of brain-derived neurotrophic factor (BDNF) was increased. In addition, idebenone and rasagiline induced the expression of Lin28A and Lin28B, respectively, leading to decreased expression of let-7 family microRNAs and increased expression of Dicer protein. Data from gene overexpression and knockdown experiments indicated that let-7 and Dicer are essential for the synergistic neuroprotective effects of the two drugs. Our results suggest that the combined treatment with rasagiline and idebenone has a synergistic effect, alleviating intraretinal reflex damage and restoring visual function. Furthermore, this combination therapy exerts a neuroprotective effect by enhancing the selective regulation of let-7 by Lin28A/B. These findings suggest that the combination therapy of rasagiline and idebenone is a feasible treatment option for optic nerve diseases. [2] |
| Molecular Formula |
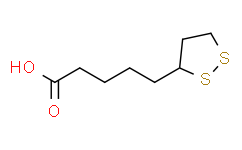
C₈H₁₄O₂S₂
|
|---|---|
| Molecular Weight |
206.3256
|
| Exact Mass |
206.043
|
| Elemental Analysis |
C, 46.57; H, 6.84; O, 15.51; S, 31.08
|
| CAS # |
1077-28-7
|
| Related CAS # |
α-Lipoic Acid;1077-28-7
|
| PubChem CID |
864
|
| Appearance |
Light yellow to yellow solid powder
|
| Density |
1.2±0.1 g/cm3
|
| Boiling Point |
362.5±11.0 °C at 760 mmHg
|
| Melting Point |
60-62ºC
|
| Flash Point |
173.0±19.3 °C
|
| Vapour Pressure |
0.0±1.7 mmHg at 25°C
|
| Index of Refraction |
1.562
|
| LogP |
2.16
|
| Hydrogen Bond Donor Count |
1
|
| Hydrogen Bond Acceptor Count |
4
|
| Rotatable Bond Count |
5
|
| Heavy Atom Count |
12
|
| Complexity |
150
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
S1C([H])(C([H])([H])C([H])([H])S1)C([H])([H])C([H])([H])C([H])([H])C([H])([H])C(=O)O[H]
|
| InChi Key |
AGBQKNBQESQNJD-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C8H14O2S2/c9-8(10)4-2-1-3-7-5-6-11-12-7/h7H,1-6H2,(H,9,10)
|
| Chemical Name |
5-(dithiolan-3-yl)pentanoic acid
|
| Synonyms |
Lipoic Acid; (R)-5-(1,2-Dithiolan-3-yl)pentanoic acid; R-(+)-alpha-Lipoic acid; (+)-alpha-Lipoic acid; Verla-Lipon; Lipoate; Verla Lipon; VerlaLipon; Thioctic Acid; Thioctacide T; Thiogamma Injekt; Thiogamma oral; thioctic acid; dl-Thioctic acid; 1077-28-7; alpha-Lipoic acid; lipoic acid; 5-(1,2-Dithiolan-3-yl)pentanoic acid; DL-alpha-Lipoic acid; 1,2-dithiolane-3-pentanoic acid;
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
DMSO: ~100 mg/mL (~484.7 mM)
H2O: < 0.1 mg/mL |
|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.8466 mL | 24.2330 mL | 48.4660 mL | |
| 5 mM | 0.9693 mL | 4.8466 mL | 9.6932 mL | |
| 10 mM | 0.4847 mL | 2.4233 mL | 4.8466 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT03161028 | Active Recruiting |
Drug: Lipoic acid Drug: Placebo |
Multiple Sclerosis | VA Office of Research and Development |
July 1, 2018 | Phase 2 |
| NCT00765310 | Active Recruiting |
Dietary Supplement: R-alpha lipoic acid Dietary Supplement: Placebo |
Atherosclerosis | Oregon State University | April 2009 | Phase 2 Phase 3 |
| NCT00764270 | Active Recruiting |
Dietary Supplement: R-alpha lipoic acid |
Atherosclerosis | Oregon State University | August 2011 | Phase 2 Phase 3 |
| NCT02910531 | Active Recruiting |
Dietary Supplement: Alpha lipoic acid Drug: Placebo |
Cystinuria | Thomas Chi, MD | June 19, 2017 | Phase 2 |
| NCT02168140 | Active Recruiting |
Drug: bendamustine hydrochloride Drug: 6,8-bis(benzylthio)octanoic acid |
Peripheral T-cell Lymphoma Hepatosplenic T-cell Lymphoma |
Wake Forest University Health Sciences |
September 2014 | Phase 1 |