| Size | Price | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
Sulconazole Nitrate, formerly known as Sulconazole and RS-44872, is an imidazole class of antifungal agent used to treat fungal skin infections.
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
A 7-day study of 1% succinazole cream in healthy subjects showed that the total transdermal absorption rate of succinazole after topical application was 8.71%–11.3% of the dose. Another study of 1 gram of 1% succinazole cream in healthy volunteers estimated that approximately 12% of the dose was absorbed through the skin. Typically, the absorption rate of topical imidazole drugs is low (<15%); however, the absorption rate of succinazole may be higher than that of other imidazole drugs. During the 7-day collection period, approximately 6.70% of the dose was recovered in urine and 2.01% in feces. Radioactivity was detected in both urine and feces after 7 days, which may be due to drug storage effects. |
|---|---|
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation
◉ Overview of use during lactation No studies have been conducted on the use of topical suconazole during lactation. Approximately 11% of the dose is absorbed after topical application. It is considered to pose a low risk to nursing infants;[1] however, other antifungal agents with lower absorption may be more appropriate, especially for nursing newborns or preterm infants. Avoid application to the nipple area and ensure that the infant's skin does not come into direct contact with the treated area. Only water-soluble creams or gels should be applied to the breast, as ointments may expose the infant to high concentrations of mineral oil through licking.[2] ◉ Effects on breastfed infants No relevant published information was found as of the revision date. ◉ Effects on breastfeeding and breast milk No relevant published information was found as of the revision date. |
| References | |
| Additional Infomation |
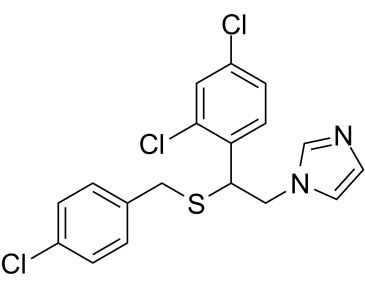
1-{2-[(4-chlorobenzyl)thio]-2-(2,4-dichlorophenyl)ethyl}-1H-imidazolium belongs to the imidazolium class of compounds. Its structure is similar to 1-ethyl-1H-imidazolium, except that one hydrogen atom on the methyl group is replaced by (4-chlorobenzyl)thioalkyl, and the other hydrogen atom is replaced by 2,4-dichlorophenyl. It belongs to the imidazolium, organosulfur, dichlorobenzene, and monochlorobenzene classes of compounds. Shoconazole (trade name: Exelderm) is a broad-spectrum antifungal drug available in both cream and solution formulations. Its active ingredient, shoconazole nitrate, is an imidazolium derivative that inhibits the growth of common pathogenic dermatophytes, thus effectively treating tinea cruris and tinea corporis. Shoconazole appears to be effective and well-tolerated in treating superficial fungal infections. Shoconazole is an azole antifungal drug. Shoconazole is a topical imidazolium derivative with broad-spectrum antifungal and antibacterial activity. Shoconazole is excreted in feces and urine. See also: Shoconazole nitrate (in saline form).
Indications 1.0% shoconazole solution is indicated for the treatment of tinea cruris and tinea corporis caused by Trichophyton rubrum, Trichophyton mentagrophytes, Epidermophyton floccosum, and Microsporum canis; as well as tinea versicolor. Its effectiveness against tinea pedis (athlete's foot) has not been established. Mechanism of Action The mechanism of action of shoconazole is not fully understood; however, it is considered similar to other imidazole derivatives. The mechanism of action of imidazole drugs can be attributed to their structural similarity to metabolically essential purine compounds. Imidazole drugs inhibit lanosterol 14-α demethylase, a cytochrome P-450-dependent enzyme in fungi responsible for converting lanosterol to ergosterol. Since ergosterol is essential for maintaining the integrity of the fungal cell membrane, inhibition of lanosterol 14-α demethylase leads to increased fungal cell permeability. Therefore, the use of imidazole drugs (such as shoconazole) can inhibit fungal growth. Pharmacodynamics Sukonazole is a broad-spectrum antifungal drug that inhibits the growth of dermatophytes, yeasts, and other filamentous and type II fungi. The relative inhibition factor (RIF) represents a fixed portion of the dose-response curve of an antifungal drug and can be used to determine its inhibitory activity. The RIF values of sukonazole against dermatophytes and pathogenic yeasts such as Aspergillus are similar to those of other imidazole drugs. Under in vitro conditions, the bactericidal efficacy of sukonazole depends on its concentration and the growth stage of the inoculated cells. Sukonazole also exhibits antibacterial properties in vitro, with inhibitory concentrations (MICs) below 12.5 mg/L against several Staphylococcus spp., Enterococcus faecalis, and several Gram-positive anaerobes. |
| Molecular Formula |
C18H15N2SCL3
|
|---|---|
| Molecular Weight |
397.7491
|
| Exact Mass |
396.002
|
| CAS # |
61318-90-9
|
| Related CAS # |
Sulconazole mononitrate;61318-91-0
|
| PubChem CID |
5318
|
| Appearance |
Typically exists as solid at room temperature
|
| Density |
1.34 g/cm3
|
| Boiling Point |
558.2ºC at 760 mmHg
|
| Melting Point |
130ºC
|
| Flash Point |
291.4ºC
|
| LogP |
6.518
|
| Hydrogen Bond Donor Count |
0
|
| Hydrogen Bond Acceptor Count |
2
|
| Rotatable Bond Count |
6
|
| Heavy Atom Count |
24
|
| Complexity |
379
|
| Defined Atom Stereocenter Count |
0
|
| InChi Key |
AFNXATANNDIXLG-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C18H15Cl3N2S/c19-14-3-1-13(2-4-14)11-24-18(10-23-8-7-22-12-23)16-6-5-15(20)9-17(16)21/h1-9,12,18H,10-11H2
|
| Chemical Name |
1-[2-[(4-chlorophenyl)methylsulfanyl]-2-(2,4-dichlorophenyl)ethyl]imidazole
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5141 mL | 12.5707 mL | 25.1414 mL | |
| 5 mM | 0.5028 mL | 2.5141 mL | 5.0283 mL | |
| 10 mM | 0.2514 mL | 1.2571 mL | 2.5141 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.