| Size | Price | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
In laboratory animals, the gastrointestinal absorption of polychlorinated biphenyls (PCBs) is well documented; however, few studies provide quantitative assessments. In rats, all homologues were rapidly absorbed after gavage administration of doses ranging from 5 to 100 mg/kg of monochlorobiphenyl to hexachlorobiphenyl (without reported excipients). Drug retention exceeded 90% of the administered dose within 4 days and appeared to be dose-independent. Due to low excretion, a relationship between substitution patterns and absorption rates could not be established. However, a subsequent study reported that absorption efficiency in rats decreased with increasing chlorine atom count, with 95% absorption for dichlorobiphenyl and only 75% for octachlorobiphenyl. Similar results were reported in monkeys after gavage administration of single doses of 1.5 or 3.0 g/kg of Aroclor 1248, and in ferrets supplemented with 0.05 mg (14)C-labeled Aroclor 1254 in their diet on days 14 and 35 of gestation. Drug retention rates in monkeys and ferrets were estimated to be over 90% and 85.4% of the administered dose, respectively. In minks, absorption exceeded 90% after a single gavage administration of 10 mg PCB 105. In mice, rapid absorption followed by gavage administration of 8 mg/kg PCB 52 (2,2',5,5'-tetrachlorobiphenyl) or 100 mg/kg PCB 77 (3,3',4,4'-tetrachlorobiphenyl) resulted in a 4-7 fold increase in serum concentration within 30-60 minutes, reaching peak serum concentrations 2 hours post-administration. In monkeys, after a single administration of 1.5 or 3.0 g/kg Aroclor 1248 for 4 days, liver concentrations (25 or 53 μg/g) were dose-dependently higher than those in the kidneys (12 or 27 μg/g) and brain tissue (17 or 28 μg/g), and more than twice as high. After 14 days of treatment, this difference increased significantly due to decreased concentrations in kidney and brain tissue and increased concentrations in liver. During the 10-month treatment period in monkeys, blood concentrations of Aroclor 1254 rapidly increased (from approximately 1.2 μg/g initially to approximately 100 μg/g in the high-dose group), ranging from 20 to 80 μg/kg/day, but this increase plateaued during the remaining 27 months of treatment. During the 37-month treatment period, a dose of 5 μg/kg/day caused only a slight increase in blood polychlorinated biphenyl (PCB) levels. When data are expressed as relative concentrations, absorption and bioaccumulation appear to be dose-dependent. However, concentration/dose levels were influenced to some extent by background PCB levels in the control group, with a greater effect on relative concentrations in the low-dose group than in the high-dose group. Four weeks after intraperitoneal injection of 1 g Aroclor 1248 in rats, the residual amount of tetrachlorobiphenyl (TCB) in adipose tissue was 338 μg/g, and chromatographic and spectroscopic analysis of the residues showed an increase in the content of pentachlorobiphenyl and hexachlorobiphenyl (up to 63%). Only 5% of the hyperchlorinated components were present in the TCB-treated group. Chromatographic analysis of excreta showed that over 90% of the dose of Aroclor 1248 (1.5 or 3.0 g/kg body weight) was absorbed by the gastrointestinal tract after a single oral dose. The main route of absorption was excretion into the gastrointestinal tract via bile. By day 14 after PCB administration, 5.6% of the initial dose had been excreted in urine and feces. Metabolites/Metabolites PCBs can be absorbed via inhalation, oral administration, and skin contact. They are transported in the blood and are generally bound to albumin. Due to their lipophilicity, they tend to accumulate in lipid-rich tissues such as the liver, adipose tissue, and skin. PCB metabolism is very slow and depends on the degree and location of chlorination. Polychlorinated biphenyls (PCBs) are metabolized into polar metabolites in the microsomal monooxygenase system catalyzed by cytochrome P-450 enzymes. These polar metabolites can bind to glutathione and glucuronic acid. The main metabolites are hydroxylation products, which are excreted in bile and feces. Due to their slow metabolism, PCBs tend to accumulate in tissues throughout the body. (L4, T6) |
|---|---|
| Toxicity/Toxicokinetics |
Toxicity Summary
Different polychlorinated biphenyls (PCBs) have different mechanisms of action. Dioxin-like PCBs bind to aryl hydrocarbon receptors (AHRs) and disrupt cellular function by altering gene transcription, primarily by inducing the expression of phase I and II enzymes in the liver (especially the cytochrome P450 family). The toxic effects of most PCBs are thought to result from AHR binding. Other PCBs are thought to interfere with calcium channels and/or alter dopamine levels in the brain. PCBs can also cause endocrine disorders by altering thyroid hormone production and binding to estrogen receptors, thereby stimulating the growth of certain cancer cells and producing other estrogen-like effects, such as reproductive dysfunction. They can also accumulate bioaccumulate by binding to receptor proteins such as uteroglobulin. (A3, A4, A30, A66) Toxicity Data LD50: 11 g/kg (oral, rat) (T14) LD50: 880 mg/kg (intraperitoneal, mouse) (T14) Interactions Acute parenteral administration of several commercially available PCB mixtures (Aroclor 1242, 1248, 1254, and 1260) and a synthetic PCB mixture reflecting PCB homologues detected in human milk antagonized 2,3,7,8-TCDD/2,3,7,8-tetrachlorodibenzodioxin/induced mouse immunoreactivity to sheep erythrocytes (SRBCs) at a PCB:TCDD dose ratio >1000:1. Aroclor 1232 had no effect on TCDD-induced immunotoxicity in these mice. Studies have shown that the dose range of Aroclor that can antagonize the acute immunotoxicity of a single injection of 0.0012 or 0.0036 mg/kg 2,3,7,8-TCDD is approximately 1 to 50 mg/kg/day. In studies investigating seven different polychlorinated biphenyl (PCB) homologues (six hexachlorobiphenyls and one pentachlorobiphenyl, with different chlorine substitution patterns) to assess their ability to affect the immunotoxicity of a single intraperitoneal injection of 0.0012 ug/kg 2,3,7,8-TCDD in mice, three were found to have antagonistic effects (2,3,3',4,5,5'-, 2,3,3',4,5'-, and 2,2',4,4',5,5'- homologues), while four had no effect (2,3,3',4,4',5'-, 2,3',4,4',5',6-, 2,2',4,4',5,6'-, and 2,2',4,4',6,6'- homologues). In these studies, the dose range for individual PCB homologues was approximately 1 to 100–300 mg/kg. Oral co-exposure of pregnant mice to 244 mg/kg Aroclor 1254 and 0.020 mg/kg 2,3,7,8-TCDD (Aroclor:TCDD dose ratio of 12,200:1) completely antagonized TCDD-induced cleft palate formation in offspring. The interaction between polychlorinated biphenyls (PCBs) and tetrachlorodibenzo-p-dioxins (TCDD)-induced developmental toxicity is complex. Of the one tetrachlorobiphenyl and two hexachlorobiphenyl homologues studied, one (2,3,3',4,4',5-homologue) enhanced TCDD-induced cleft palate formation, while the other two (2,2',4,4'-homologue and 2,2',4,4',5,5'-homologue) antagonized the effects of TCDD. The antagonistic effects of 2,2',4,4'-tetrachlorobiphenyl and 2,2',4,4',5,5'-hexachlorobiphenyl on TCDD-induced cleft palate formation in mouse offspring exhibit a complex inverted U-shaped relationship with dose. For example, 10-20 mg/kg doses of 2,2',4,4',5,5'-hexadiol (2,2',4,4',5,5'-hexaCB) showed no antagonistic effect against TCDD at a dose of 0.0015 μg/kg, but the antagonistic effect gradually increased with increasing doses of 2,2',4,4',5,5'-hexaCB, reaching a maximum at 500 mg/kg, and then disappeared at 1000 mg/kg. 2,2',4,4',5,5'-hexaCB also antagonized TCDD-induced hydronephrosis in mouse offspring, with its antagonistic effect showing a similar inverted U-shaped relationship with dose. In contrast, simultaneous exposure of pregnant mice to 2,3,3',4,4',5-hexylchlorobenzene and 2,3,4,7,8-pentachlorodibenzofuran appeared to cumulatively lead to hydronephrosis and cleft palate in offspring. Several 13-week oral exposure studies investigated potential binary interactions between three polychlorinated biphenyl (PCB) homologues (at varying dietary concentrations, daily doses ranging from approximately 0.1 to 10 mg/kg/day) and 2,3,7,8-tetrachlorodibenzodioxin (at varying dietary concentrations, daily doses ranging from approximately 0.00003 to 0.3 mg/kg/day), and their effects on various endpoints in rats. The PCB to tetrachlorodibenzodioxin concentration ratios in these studies were selected based on relative concentrations in human milk and fat samples. 2,2',4,4',5,5'-hexachlorobiphenyl (2,2',4,4',5,5'-HexaCB) and 2,3,7,8-tetrachlorodibenzo-p-dioxin (2,3,7,8-TCDD) showed an additive effect in lowering thyroid hormone levels at 4 weeks, but a synergistic effect at 13 weeks; while the combined effect of 2,3,3',4,4',5-hexachlorobiphenyl (2,3,7,8-TCDD) and 2,3,7,8-TCDD in lowering thyroid hormone levels was less than the additive effect. 2,2',4,4',5-Hexachlorobiphenyl did not affect TCDD-induced changes in body weight and thymus weight, but increased relative liver weight when combined with TCDD; while the combined effects of 2,3,3',4,4',5-hexachlorobiphenyl (2,3,7,8-HexaCB) and 3,3',4,4',5-pentachlorobiphenyl (3,3',4,4',5-PentaCB) with TCDD on these endpoints were less additive than additive effects. 2,2',4,4',5,5'-hexamethylenedibenzothiophene (hexaCB) and 2,3,7,8-tetrachlorodibenzodioxin (TCDD) showed a significant synergistic effect in increasing hepatic porphyrin levels, but no such synergistic effect was observed between 2,3,3',4,4',5-hexamethylenedibenzothiophene (hexaCB) and 3,3',4,4',5-pentanedibenzothiophene (pentaCB) and 2,3,7,8-TCDD. All three homologues individually reduced hepatic retinol and retinyl palmitate levels. When used in combination with TCDD, the observed synergistic effect was less than the additive effect, but the TCDD dose used in this study resulted in near-maximum reductions in retinoid levels. Non-human toxicity values Oral LD50 in rats: 11000 mg/kg Dermal LD50 in rabbits: 11.0 g/kg |
| Additional Infomation |
PCB-1248 is a viscous, oily liquid. (NTP, 1992)
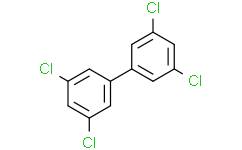
3,3',5,5'-Tetrachlorobiphenyl is a tetrachlorobiphenyl in which the two phenyl groups are replaced by chlorine atoms at the 3 and 5 positions, respectively. It is both a tetrachlorobiphenyl and a dichlorobenzene. Aroclor 1248 is a commercially available mixture of polychlorinated biphenyls (PCBs) with an average chlorine content of 48%. It consists of monochloro to heptachloro homologues. Polychlorinated biphenyls (PCBs) are a class of 209 synthetic organic compounds with 1-10 chlorine atoms attached to their biphenyl molecules. PCBs were once produced as commercial mixtures, but were banned in the 1970s due to their bioaccumulation in the environment and harmful health effects. However, PCBs are not easily decomposed and still exist in the environment today. (L4) See also: Polychlorinated biphenyls (composition). Mechanism of Action A potential molecular target for polychlorinated biphenyls (PCBs) disrupting calcium homeostasis is the renin-mediated Ca²⁺ channel, which may be related to nervous system and neurodevelopmental effects. Mixtures of commercially available PCBs with moderate to high chlorination levels (Aroclor 1248, 1254, and 1260) enhanced the binding of renin to calcium release channels on the sarcoplasmic reticulum of rabbit skeletal or cardiac muscle, while mixtures with lower (Aroclor 1221, 1232) or higher (Aroclor 1268) chlorination levels showed little enhancement. Studies on selected pentachlorobiphenyls showed that ortho-substitution favored activity; 2,2',3,5',6-pentaphenylamine (2,2',3,5',6-pentaCB) exhibited the strongest enhancing effect on renin binding, while the 3,3',4,4',5-isomer showed no enhancement. The enhancing effect produced by the fully ortho-substituted 2,2',4,6,6'-isomer was weaker than that of the 2,2',3,5',6-isomer, suggesting that a certain degree of biphenyl bond rotation may be crucial for complete activity. Studies on freshly dissected rat hippocampal sections showed that perfusion of the tri-ortho-substituted homologue (2,2',3,5',6-pentaCB) enhanced Rynodin binding and inhibited electrophysiological responses induced by electrical impulses, while the mono-ortho-substituted homologue (2,3',4,4'-tetraCB) did not show any enhancing effect on Rynodin binding or inhibition of electrophysiological responses induced by electrical impulses. Rats that were administered 8 or 32 mg/kg 2,2',3,5',6-pentaCB daily by gavage during days 10–16 of gestation exhibited neurobehavioral changes in adulthood (decreased open-field motor activity, accelerated learning speed on working memory tasks, and no change on delayed spatial alternation tasks), as well as altered binding of calcium channels to rhinodine in specific brain regions (e.g., decreased in the hippocampus and increased in the cerebral cortex). While the specific association between these changes in rhinodine binding and the observed neurobehavioral changes remains unclear, the results of this series of studies highlight the potential importance of aryl hydrocarbon receptor-independent mechanisms in the neurological and neurodevelopmental effects induced by PCBs. Drug Warnings Food and Environmental Factors: Signs or symptoms reported by infants or effects on nursing: PCBs and polybrominated biphenyls: sluggishness, hypotonia, depressed mood, apathy. /Excerpt from Table 7/ |
| Molecular Formula |
C12H6CL4
|
|---|---|
| Molecular Weight |
291.988039493561
|
| Exact Mass |
289.92
|
| CAS # |
12737-87-0
|
| PubChem CID |
36400
|
| Appearance |
Colorless mobile oil
Lower chlorinated Aroclors (1221, 1232, 1016, 1242, and 1248) are colorless mobile oils. Increasing chlorine content results in mixtures taking on the consistency of viscous liquids (Aroclor 1254) or sticky resins (Aroclors 1260 and 1262). Arclors 1268 and 1270 are white powders. |
| Melting Point |
340-375 °C
|
| LogP |
6.1
|
| Hydrogen Bond Donor Count |
0
|
| Hydrogen Bond Acceptor Count |
0
|
| Rotatable Bond Count |
1
|
| Heavy Atom Count |
16
|
| Complexity |
189
|
| Defined Atom Stereocenter Count |
0
|
| InChi Key |
UTMWFJSRHLYRPY-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C12H6Cl4/c13-9-1-7(2-10(14)5-9)8-3-11(15)6-12(16)4-8/h1-6H
|
| Chemical Name |
1,3-dichloro-5-(3,5-dichlorophenyl)benzene
|
| Synonyms |
KC-400; KC400; KC 400
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4248 mL | 17.1239 mL | 34.2477 mL | |
| 5 mM | 0.6850 mL | 3.4248 mL | 6.8495 mL | |
| 10 mM | 0.3425 mL | 1.7124 mL | 3.4248 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.