| Size | Price | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Chlordiazepoxide is primarily excreted in the urine, with 1%–2% in its unchanged form and 3%–6% in conjugates. Increased clearance of chlordiazepoxide and its metabolites was observed in mice after a single pretreatment. This increase in clearance was insufficient to explain the observed tolerance and could not rule out the possibility of altered distribution of the drug between blood and brain tissue. In cynomolgus monkeys, 14C levels in tissues peaked 2–6 hours after oral administration of chlordiazepoxide. The concentration ratio of (14)C in brain tissue to blood was greater than 1. The highest concentrations of (14)C were found in the liver and kidneys, and lower concentrations in the heart, lungs, spleen, brain, adrenal glands, pancreas, and fat. After 24 hours, chlordiazepoxide levels were 15% in the gastrointestinal tract, 33% in tissues, 34% in urine, and 1% in feces. Oral administration of chlordiazepoxide is absorbed faster and more predictably than intramuscular injection, but there is significant inter-individual variability in plasma concentrations. In healthy male volunteers, the plasma concentration-time curve of chlordiazepoxide following intravenous injection can be well described by a bi-exponential expression conforming to a two-compartment model system. The mean half-lives for the distribution and elimination phases were 0.25 hours and 9.4 hours, respectively, while the mean central compartment volume (V1) and total volume of distribution (VDβ) were 18% and 31% of body weight, respectively. Drug absorption after intramuscular administration is relatively slow, thus requiring a two-compartment “muscle model” that includes the precipitation and dissolution of the drug in muscle tissue to adequately describe the plasma concentration of chlordiazepoxide. The volume of distribution of chlordiazepoxide was significantly larger in female subjects than in males, indicating a more extensive distribution of the drug in women. Chlordiazepoxide is absorbed only to a limited extent by erythrocytes. For more complete data on absorption, distribution, and excretion of chlordiazepoxide (9 types), please visit the HSDB record page. Metabolism/MetabolitesHepatic metabolism. In humans and dogs, biotransformation results in the successive hydrolysis of the 2-methylamino substituent and the hydrolytic cleavage of the resulting lactam; 7-chloro-1,3-dihydro-5-phenyl-2H-1,4-benzodiazepine-2-one-4-oxide and N-(2-amino-5-chloro-α-phenyl-benzyl)glycine-N-oxide are excreted in the urine. In dogs, 1% of the ritapride dose is present in the urine as oxazepam glucuronide, and another 1% is present in the feces as free oxazepam glucuronide. These minor metabolites are presumed to be produced via a lactamation reaction, through a step involving the reduction of the N-oxide functional group. In humans and monkeys, 2-amino-7-chloro-5-phenyl-3H-1,4-benzodiazepine-4-oxide is generated. /Excerpt from Table/ In Pseudomonas and Clostridium, N-(2-amino-5-chloro-α-phenylbenzyl)-glycine-N-oxide is produced. /Excerpt from Table/ ...The main active metabolite of chlordiazepoxide is desmethylchlordiazepoxide... Hepatic metabolism. Elimination pathway: Chlordiazepoxide is excreted in the urine, of which 1%–2% is unchanged and 3%–6% is conjugates. Half-life: 24–48 hours Biological half-life 24–48 hours The elimination half-life of the main active metabolite, desmethylchlordiazepoxide, is 1–4 days... After intravenous injection, the plasma concentration-time curve of chlordiazepoxide can be well described by a bi-exponential expression conforming to a two-compartment model system. Dosage in healthy male volunteers. The mean half-lives for the distribution and elimination phases were 0.25 hours and 9.4 hours, respectively… This study investigated the in vivo distribution of chlordiazepoxide (CDP) 50 mg 10 minutes after intravenous infusion. Subjects included healthy individuals and patients with biopsy-confirmed cirrhosis. The mean kinetic parameter, half-life (β), for healthy subjects was 10.0 hours. The mean half-life (β) for patients with cirrhosis was 34.9 hours. Half-life: 5–30 hours / From table / |
|---|---|
| Toxicity/Toxicokinetics |
Toxicity Summary
Chlordiazepoxide binds to stereospecific benzodiazepine (BZD) binding sites on GABAsub>A receptor complexes in multiple sites of the central nervous system, including the limbic system and reticular formation. This leads to increased binding of the inhibitory neurotransmitter GABA to GABAsub>A receptors. Consequently, BZDs enhance GABA-mediated influx of chloride ions through GABA receptor channels, resulting in membrane hyperpolarization. Net neurodepressant effects lead to the observed sedative, hypnotic, anxiolytic, and muscle relaxant properties. Toxicity Data LD50: 537 mg/kg (oral, rat) (A308) Interactions Chlordiazepoxide has been reported to have an additive sedative effect or enhanced atropine-like effect when used in combination with tricyclic antidepressants. Several case reports describe this interaction. The elimination and plasma clearance of chlordiazepoxide in women taking oral contraceptives may be reduced. The effects of co-administration of ethanol (4 g/kg) and clozapine (CDP, 12.5 mg/kg) on C-adenosine monophosphate (C-GMP) and C-guanosine monophosphate (C-GMP) levels in the mouse brain were investigated. Results showed that the co-administration did not produce an additive effect on C-Adenosine monophosphate levels. The combined action of ethanol and clozapine (CDP) resulted in an additive decrease in C-GMP concentration in the cerebellum at 2 and 4 hours. This led to a prolonged period (approximately 2.5 hours) during which cerebellar C-GMP levels were below 30% of the control, coinciding with an increase in sleep duration. Compared to mice injected with clozapine alone, mice injected with clozapine (CDP)/ethanol showed significantly higher levels of clozapine in both blood and brain tissue. The increased CDP concentration may be partly responsible for the prolonged ethanol-induced sleep overload. N-demethylated metabolites and/or their metabolites were the main contributors to the additive effect. For more complete data on interactions of chlordiazepoxide (9 types in total), please visit the HSDB record page. Non-human toxicity values Oral LD50 in rats: 548 mg/kg Intraperitoneal LD50 in rats: 143 mg/kg Intravenous LD50 in rats: 165 mg/kg Oral LD50 in mice: 260 mg/kg For more complete data on non-human toxicity of chlordiazepoxide (7 types in total), please visit the HSDB record page. |
| Additional Infomation |
Therapeutic Uses
Adjuvant, anesthetic; anti-anxiety drug, benzodiazepine; GABA modulator; non-barbiturate sedative. Veterinary Use: Adding 15-30% to feed can induce central nervous system depression in orioles; quail (5-10% dose) will experience an excitatory phase before central nervous system depression. Intraperitoneal injection of 3-10 mg/kg is used to treat aggressive behavior in rats. As a central nervous system depressant in rats and mice (80 mg/kg). As an adrenergic blocker in mice (20 mg/kg). In zoo animals, administration of 4-25 mg/kg via an appropriate route can tame aggressive animals; barbiturates can be used as adjunctive therapy. In rhesus monkeys, chlordiazepoxide can be used as an anticonvulsant (4.4 mg/kg, intravenously). /Excerpt from Table/ Chlordiazepoxide…is used…to treat agitation caused by acute alcohol withdrawal. ...Can be used as a pre-anesthetic medication and for obstetric delivery. /Benzodiazepines/ For more complete data on the therapeutic uses of chlordiazepoxide (7 types), please visit the HSDB record page. Drug Warnings The following precautions should be taken when using chlordiazepoxide hydrochloride: patients with known hypersensitivity to this product, elderly and severely depressed individuals, pregnant and lactating women, patients with known renal and hepatic impairment, patients taking other central nervous system depressants, and patients with a history of drug addiction or who have arbitrarily changed their drug dosage. Chlordiazepoxide Hydrochloride The safety and efficacy of oral chlordiazepoxide in children under 6 years of age have not been established. Intravenous preparations prepared with 0.9% sodium chloride injection or water for injection should not be administered intramuscularly (SRP: absorption at this site is very slow and varies greatly from person to person). A double-blind crossover trial investigated the efficacy of a normal oral dose (10 mg three times daily) of chlordiazepoxide (Ripril) in 7 patients with respiratory failure primarily caused by chronic bronchitis. In 6 patients, the drug caused a significant increase in mixed venous blood carbon dioxide partial pressure and a significant decrease in forced expiratory volume in one second. The conclusion is that clozapine is contraindicated in patients with respiratory failure due to chronic bronchitis. For more complete data on clozapine (13 total), please visit the HSDB record page. Pharmacodynamics Clozapine has anxiolytic, sedative, appetite-stimulating, and mild analgesic effects. The drug appears to block EEG arousal induced by stimulation of the brainstem reticular formation. Extensive studies in various animals have shown its action on the limbic system, and recent evidence suggests the limbic system is involved in emotional responses. Oral doses that do not induce sedation tamed aggressive monkeys. Clozapine exhibits a “taming” effect, eliminating fear and aggression. The taming effect of clozapine was further confirmed in rats that had become aggressive due to damage to the septum. Effective doses inhibiting aggressive responses were far lower than those inducing sedation in these animals. |
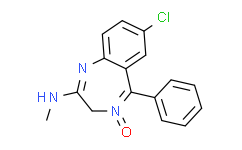
| Molecular Formula |
C16H14CLN3O
|
|---|---|
| Molecular Weight |
299.76
|
| Exact Mass |
299.082
|
| CAS # |
58-25-3
|
| Related CAS # |
438-41-5
|
| PubChem CID |
2712
|
| Appearance |
Yellow crystalline powder
Light yellow plates from ethanol |
| Melting Point |
236-236.5
236-236.5 °C |
| LogP |
2.4
|
| Hydrogen Bond Donor Count |
1
|
| Hydrogen Bond Acceptor Count |
3
|
| Rotatable Bond Count |
1
|
| Heavy Atom Count |
21
|
| Complexity |
580
|
| Defined Atom Stereocenter Count |
0
|
| InChi Key |
BUCORZSTKDOEKQ-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C16H14ClN3O/c1-18-15-10-20(21)16(11-5-3-2-4-6-11)13-9-12(17)7-8-14(13)19-15/h2-9,21H,10H2,1H3
|
| Chemical Name |
7-chloro-4-hydroxy-N-methyl-5-phenyl-3H-1,4-benzodiazepin-2-imine
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3360 mL | 16.6800 mL | 33.3600 mL | |
| 5 mM | 0.6672 mL | 3.3360 mL | 6.6720 mL | |
| 10 mM | 0.3336 mL | 1.6680 mL | 3.3360 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.