| Size | Price | |
|---|---|---|
| 500mg | ||
| 1g | ||
| Other Sizes |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
The plasma concentration profile of bromitezolam can be described using a one-compartment open model of first-order absorption. 0.63 L/kg Total clearance: 109 mL/min. Metabolisms/Metabolites There are two main metabolites: a 1-methylhydroxy derivative and a 4-hydroxy derivative (Eberts et al., 1981; Boehringer Ingelheim, Product Information). The 4-hydroxy metabolite has significantly lower pharmacological activity than the parent drug, but the 1-methylhydroxy metabolite may have comparable activity (Gall et al., 1978; Jochemsen et al., 1982; Sethy and Harris, 1982; Jochemsen et al., Unpublished Results). However, in young, healthy subjects, these active compounds did not reach measurable concentrations in plasma after a single dose of bromitezolam (Jochemsen et al., 1982; Jochemsen et al., Unpublished Results). It is known that the human metabolites of bromitezolam include α-hydroxy-BRT and 6-hydroxy-BRT. Biological half-life 4.4 hours. |
|---|---|
| Toxicity/Toxicokinetics |
Toxicity Summary
Benzodiazepines bind nonspecifically to benzodiazepine receptors BNZ1 (mediates sleep) and BNZ2 (affects muscle relaxation, anticonvulsant activity, motor coordination, and memory). Since benzodiazepine receptors are thought to couple to γ-aminobutyric acid A (GABAA) receptors, they enhance the effects of GABA by increasing the affinity of GABA for GABA receptors. The binding of the inhibitory neurotransmitter GABA to this site opens chloride ion channels, leading to cell membrane hyperpolarization and preventing further cell excitation. Pregnancy and Lactation Effects ◉ Overview of Use During Lactation Bromotezolam has not been approved for marketing by the U.S. Food and Drug Administration (FDA). Information on bromotezolam entering breast milk is limited, although the levels appear to be very low. Especially in breastfed newborns or premature infants, alternative medications may be necessary. If bromotezolam is used, the infant should be monitored for sedation, feeding difficulties, and poor weight gain. ◉ Effects on Breastfed Infants A woman with bipolar disorder took bromitezolam 0.25 mg, alprazolam 0.4 mg, and trazodone 50 mg daily during pregnancy and postpartum. She also took sertraline 25 mg daily, gradually increasing to 100 mg twice daily. The infant was admitted to the neonatal intensive care unit due to neonatal withdrawal syndrome and discharged on day 8 after birth. The mother exclusively breastfed the infant for 3 months while taking bromitezolam 0.25 mg (once daily), sertraline 50 mg (twice daily), alprazolam 0.4 mg (once daily), and zolpidem 10 mg (once daily). No drug-related adverse reactions were observed at follow-up examinations at 1, 3, or 6 months. ◉ Effects on Lactation and Breast Milk No relevant published information was found as of the revision date. The mean value of protein-bound free fraction (%) is 8.4 ± 0.7. |
| Additional Infomation |
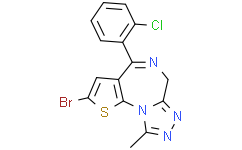
Brotezolam is an organic molecular entity. It is a sedative-hypnotic thiophene benzodiazepine, belonging to the benzodiazepine analogue class. It has anti-anxiety, anticonvulsant, hypnotic, sedative, and skeletal muscle relaxant effects. Brotezolam's effects are similar to those of short-acting benzodiazepines (such as triazolam). Brotezolam is indicated for the treatment of severe or asthenic insomnia for 2-4 weeks. Brotezolam is a highly potent drug with rapid elimination and an average half-life of 4.4 hours (range 3.6-7.9 hours). Brotezolam is not approved for sale in the UK, US, or Canada, but is available in the Netherlands, Germany, Spain, Belgium, Austria, Portugal, Israel, Italy, and Japan. Brotezolam is a triazolam benzodiazepine derivative with sedative, hypnotic, anti-anxiety, and anticonvulsant effects. Brotezolam binds to benzodiazepine binding sites on γ-aminobutyric acid (GABA)-A receptors in the central nervous system (CNS). This leads to increased chloride channel opening, membrane hyperpolarization, and enhanced GABA's inhibitory effect on the CNS. Brotezolam is a sedative-hypnotic thienodiazepine, belonging to the benzodiazepine analogue class. It has anxiolytic, anticonvulsant, hypnotic, sedative, and skeletal muscle relaxant effects, and its efficacy is considered similar to short-acting benzodiazepines such as triazolam. It is used for short-term treatment of severe or serious insomnia. Brotezolam is a highly potent drug, rapidly metabolized, with an average half-life of 4.4 hours (range 3.6–7.9 hours). Brotezolam is not approved for sale in the United Kingdom, the United States, or Canada. It is approved for sale in the Netherlands, Germany, Spain, Belgium, Austria, Portugal, Israel, Italy, and Japan.
Drug Indications Bromotezolam is indicated for the treatment of severe or serious insomnia, with a course of treatment lasting 2-4 weeks. |
| Molecular Formula |
C15H10N4SCLBR
|
|---|---|
| Molecular Weight |
393.6887
|
| Exact Mass |
391.949
|
| CAS # |
57801-81-7
|
| PubChem CID |
2451
|
| Appearance |
Typically exists as solid at room temperature
|
| Melting Point |
211-213
|
| LogP |
2.8
|
| Hydrogen Bond Donor Count |
0
|
| Hydrogen Bond Acceptor Count |
4
|
| Rotatable Bond Count |
1
|
| Heavy Atom Count |
22
|
| Complexity |
465
|
| Defined Atom Stereocenter Count |
0
|
| InChi Key |
UMSGKTJDUHERQW-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C15H10BrClN4S/c1-8-19-20-13-7-18-14(9-4-2-3-5-11(9)17)10-6-12(16)22-15(10)21(8)13/h2-6H,7H2,1H3
|
| Chemical Name |
4-bromo-7-(2-chlorophenyl)-13-methyl-3-thia-1,8,11,12-tetrazatricyclo[8.3.0.02,6]trideca-2(6),4,7,10,12-pentaene
|
| Synonyms |
We-941-BS; We-941; Brotizolam
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5401 mL | 12.7003 mL | 25.4007 mL | |
| 5 mM | 0.5080 mL | 2.5401 mL | 5.0801 mL | |
| 10 mM | 0.2540 mL | 1.2700 mL | 2.5401 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.