| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| Other Sizes |
Purity: ≥98%
Mavorixafor HCl (also known as AMD-1107, AMD070, X4P-001) is a potent, selective and orally bioavailable antagonist of CXCR4 with anti-HIV activity. It inhibits CXCR4 with an IC50 value of 13 nM in a CXCR4 125I-SDF inhibition binding assay, also inhibits the replication of T-tropic HIV-1 (NL4.3 strain) in MT-4 cells and PBMCs. The CXC chemokine receptor CXCR2 is upregulated in a variety of different tumor cell types and involved in tumor cell proliferation and progression. Inhibition of CXCR2 led to reduced metastasis and decreased tumorigenesis. As a CXCR2 antagonist, SX-682 has the potential to treat cancer. AMD-070 has been evaluated in phase I/II studies in different solid tumors by X4 Pharmaceuticals. This compound also has the potential use for the treatment of HIV-1 infection.
On April 29, 2024, the U.S. Food and Drug Administration (FDA) approved mavorixafor (trademarked as Xolremdi by X4 Pharmaceuticals) for the treatment of warts, hypogammaglobulinemia, immunodeficiency, myelokathexis (WHIM) syndrome.| Targets |
125I-SDF-CXCR4 ( IC50 = 13 nM ); HIV-1 (NL4.3 strain) ( IC50 = 1 nM ); HIV-1 (NL4.3 strain) ( IC50 = 9 nM ); HIV-1 (NL4.3 strain) ( IC50 = 3 nM ); HIV-1 (NL4.3 strain) ( IC50 = 26 nM )
|
||
|---|---|---|---|
| ln Vitro |
|
||
| ln Vivo |
|
||
| Animal Protocol |
|
||
| ADME/Pharmacokinetics |
Absorption
In adults with WHIM syndrome, after a once-daily dose of 400 mg, the steady-state mean (CV%) Cmax was 3304 (58.6%) ng/mL, and the 0-24 hour AUC (AUC0-24h) was 13970 (58.4%) ng·h/mL. The pharmacokinetics of Mavorixafor are non-linear; the increases in Cmax and AUC0-24h are greater than dose-proportional across the dose range of 50 mg (0.125 times the recommended dose) to 400 mg. In healthy subjects, steady-state plasma concentrations of Mavorixafor are reached approximately after 9 to 12 days following administration of the highest approved recommended dose. At the highest approved recommended dose, the median (range) Tmax of Mavorixafor is 2.8 hours (1.9 to 4 hours). Food decreases Cmax and AUC. Excretion Route In healthy subjects, following a single oral dose of radiolabeled mavolixafor, 74.2% of the administered dose was recovered within a 240-hour collection period, with 61.0% of the radioactive material recovered in feces and 13.2% (3% unchanged) in urine. Volume of Distribution In adults with WHIM syndrome, the volume of distribution of mavolixafor is 768 L. Clearance In healthy subjects, following a single dose of 400 mg mavolixafor, the mean (coefficient of variation) apparent clearance was 62 L/h (40%). Mavolixafor exhibits at least partially nonlinear apparent clearance; however, this nonlinear clearance is not clinically significant at the approved recommended dose. Protein Binding In vitro studies have shown that mavolixafor binds to human plasma proteins >93%. Metabolism/Metabolites Mavolixafor is primarily metabolized via CYP3A4, and secondarily via CYP2D6. Biological Half-Life In healthy subjects, the mean (CV%) terminal half-life after a single dose of 400 mg mavolixafor is 82 hours (34%). Mavorixafor (AMD070) is a small molecule drug with high oral bioavailability; good oral bioavailability was observed in both rats and dogs [1] - In CD-1 mice, after oral administration of 400 μg/mouse of Mavorixafor (AMD070), the drug was detected in lung tissue, and its concentration varied over time. EC₉₀ (44 ng/mL) was used as the reference threshold for pharmacodynamic activity [2] - In C57BL/6 mice, after intraperitoneal injection of Mavorixafor (AMD070), the drug was distributed in plasma, liver, and lung tissue, and its concentration was detected at different time points [2] |
||
| Toxicity/Toxicokinetics |
Mavorixafor (AMD070) showed no cytotoxicity to MT-4 cells and PBMCs at in vitro concentrations exceeding 23 μM[1]
Effects during pregnancy and lactation ◉ Overview of use during lactation There is currently no information regarding the use of mavorixafor during lactation. The manufacturer recommends against breastfeeding during treatment and for three weeks after the last dose. ◉ Effects on breastfed infants No published information was found as of the revision date. ◉ Effects on lactation and breast milk No published information was found as of the revision date. |
||
| References |
PLoS One.2016 Mar 21;11(3):e0151765;J Med Chem.2010 Apr 22;53(8):3376-88.
|
||
| Additional Infomation |
Mavorixafor is a CXC chemokine receptor 4 (CXCR4) antagonist. It was first approved by the FDA on April 30, 2024, for the treatment of warts, hypogammaglobulinemia, infection, and bone marrow retention (WHIM) syndrome. WHIM syndrome is a inherited immunodeficiency disorder characterized by a decrease in the number of mature neutrophils and lymphocytes. WHIM syndrome is caused by mutations in the CXCR4 gene, leading to overactivation of the CXCR4 signaling pathway. Mavorixafor inhibits CXCR4 activation. Because CXCR4 mutations are also associated with human immunodeficiency virus (HIV), Waldenström macroglobulinemia (WM), B-cell non-Hodgkin lymphoma, and solid tumors including melanoma, Mavorixafor is being investigated in these diseases. Mavorixafor's mechanism of action is as a CXC chemokine receptor 4 antagonist, cytochrome P450 2D6 inhibitor, cytochrome P450 3A4 inhibitor, and P-glycoprotein inhibitor. Mavorixafor is an orally bioavailable CXC chemokine receptor 4 (CXCR4) inhibitor with potential antitumor and immune checkpoint inhibitory activities. After administration, Mavorixafor selectively binds to CXCR4, preventing CXCR4 from binding to its ligand stromal cell-derived factor 1 (SDF-1 or CXCL12). This inhibits receptor activation, leading to reduced proliferation and migration of CXCR4-overexpressing tumor cells. Furthermore, inhibition of CXCR4 prevents the recruitment of regulatory T cells and myeloid-derived suppressor cells (MDSCs) to the tumor microenvironment, thereby eliminating CXCR4-mediated immunosuppression and activating cytotoxic T lymphocyte-mediated immune responses against cancer cells. The G protein-coupled receptor CXCR4 is upregulated in various tumor cell types, inducing the recruitment of immunosuppressive cells to the tumor microenvironment, inhibiting immune surveillance, and promoting tumor angiogenesis and tumor cell proliferation. It is also a co-receptor for HIV entry into T cells. Mavorixafor is a small molecule drug, currently in Phase IV clinical trials (covering all indications), and was first approved in 2024 for the treatment of cancer, with six investigational indications. It is a derivative of AMD3100, a CXCR4 blocker.
|
| Molecular Formula |
C21H28CLN5
|
|
|---|---|---|
| Molecular Weight |
385.933523178101
|
|
| Exact Mass |
385.203
|
|
| CAS # |
880549-30-4
|
|
| Related CAS # |
558447-26-0;880549-30-4 (HCl); 2309699-17-8
|
|
| PubChem CID |
71576687
|
|
| Appearance |
Light yellow to brown solid at room temperature
|
|
| LogP |
5.078
|
|
| Hydrogen Bond Donor Count |
3
|
|
| Hydrogen Bond Acceptor Count |
4
|
|
| Rotatable Bond Count |
7
|
|
| Heavy Atom Count |
27
|
|
| Complexity |
431
|
|
| Defined Atom Stereocenter Count |
1
|
|
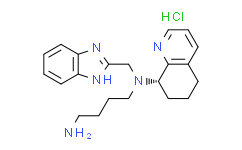
| SMILES |
N([C@H]1CCCC2C=CC=NC1=2)(CCCCN)CC1=NC2C=CC=CC=2N1.Cl
|
|
| InChi Key |
DBNMEMJSDAAGNZ-FYZYNONXSA-N
|
|
| InChi Code |
InChI=1S/C21H27N5.ClH/c22-12-3-4-14-26(15-20-24-17-9-1-2-10-18(17)25-20)19-11-5-7-16-8-6-13-23-21(16)19;/h1-2,6,8-10,13,19H,3-5,7,11-12,14-15,22H2,(H,24,25);1H/t19-;/m0./s1
|
|
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples.
Injection Formulations
Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline)(e.g. IP/IV/IM/SC) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). View More
Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] Oral Formulations
Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). View More
Oral Formulation 3: Dissolved in PEG400 (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5911 mL | 12.9557 mL | 25.9114 mL | |
| 5 mM | 0.5182 mL | 2.5911 mL | 5.1823 mL | |
| 10 mM | 0.2591 mL | 1.2956 mL | 2.5911 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.