
| Size | Price | Stock | Qty |
|---|---|---|---|
| 100mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| 5g |
|
||
| 10g |
|
||
| 25g |
|
||
| 50g | |||
| Other Sizes |
Purity: ≥98%
| Targets |
VEGFR3 (IC50 = 20 nM); Braf (IC50 = 22 nM); Raf-1 (IC50 = 6 nM); VEGFR2 (IC50 = 90 nM); PDGFRβ (IC50 = 57 nM); BrafV599E (IC50 = 38 nM); c-Kit (IC50 = 68 nM); Flt3 (IC50 = 58 nM)
VEGFR-2 (IC50 = 90 nM) [2] - VEGFR-3 (IC50 = 20 nM) [2] - PDGFR-β (IC50 = 580 nM) [2] - RAF1 (IC50 = 6 nM) [2] - B-RAF (IC50 = 22 nM) [2] - c-KIT (IC50 = 570 nM) [2] - FLT3 (IC50 = 590 nM) [2] |
|---|---|
| ln Vitro |
Sorafenib's IC50 values of 22 nM and 38 nM, respectively, inhibit both wild-type and V599E mutant B-Raf activity. Additionally, mVEGFR2 (Flk-1) as well as mVEGFR3, mPDGFRβ, Flt3, and c-Kit are all potently inhibited by sorafenib, with respective IC50 values of 15 nM, 20 nM, 57 nM, 58 nM, and 68 nM. Sorafenib has a 580 nM IC50 and only moderately inhibits FGFR-1. Sorafenib tosylate is ineffective against the following targets: ERK-1, MEK-1, EGFR, HER-2, IGFR-1, c-Met, PKB, PKA, cdk1/cyclinB, PKCα, PKCγ, and pim-1. In NIH 3T3 cells, sorafenib significantly reduces VEGFR2 phosphorylation with an IC50 of 30 nM, and Flt-3 phosphorylation in HEK-293 cells with an IC50 of 20 nM. In most cell lines, sorafenib potently inhibits MEK 1/2 and ERK 1/2 phosphorylation, but not in A549 or H460 cells. It has no impact on the PKB pathway's inhibition. Sorafenib has an IC50 of 0.28 μM and 2.6 μM, respectively, and prevents HAoSMC and MDA-MB-231 cells from proliferating. [1] Sorafenib also significantly inhibits the phosphorylation of eIF4E and down-regulates Mcl-1 levels in hepatocellular carcinoma (HCC) cells in a MEK/ERK-independent manner, in addition to inhibiting the RAF/MEK/ERK signaling pathway. With IC50 values of 6.3 μM and 4.5 μM, respectively, sorafenib inhibits the proliferation of PLC/PRF/5 and HepG2 cells and significantly induces apoptosis.[2]
Sorafenib (Bay 43-9006) exhibited potent inhibitory activity against multiple kinases involved in angiogenesis and tumor cell proliferation. It inhibited VEGFR-2, VEGFR-3, PDGFR-β, RAF1, B-RAF, c-KIT, and FLT3 with IC50 values ranging from 6 nM to 590 nM [2] - In human umbilical vein endothelial cells (HUVECs), Sorafenib (0.1–10 μM) dose-dependently inhibited VEGF-induced proliferation, with an IC50 of 0.24 μM. It also blocked VEGF-induced migration and tube formation of HUVECs, with maximum inhibition rates of 85% and 90% at 10 μM, respectively [2] - In various human tumor cell lines (HT29, SW620, A375, SK-MEL-28, HepG2), Sorafenib inhibited cell proliferation with IC50 values ranging from 2.6 μM to 10.1 μM. The highest potency was observed in SK-MEL-28 melanoma cells (IC50 = 2.6 μM) [2] - Sorafenib (1–10 μM) induced apoptosis in A375 melanoma cells, as evidenced by increased caspase-3/7 activity (2.3-fold at 10 μM) and Annexin V-positive cells (35% at 10 μM) compared to control [3] - The compound (5 μM) suppressed the RAF-MEK-ERK signaling pathway in A375 cells, reducing phosphorylated ERK (p-ERK) levels by 70% as detected by Western blot [3] - In HT29 colon cancer cells, Sorafenib (10 μM) inhibited VEGF secretion by 45% and bFGF secretion by 38% compared to untreated cells [1] |
| ln Vivo |
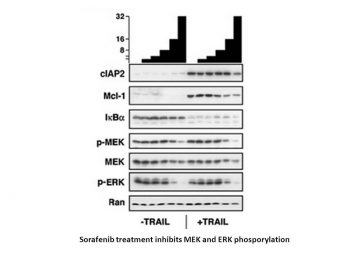
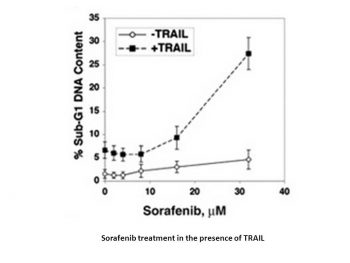
Sorafenib (60 mg/kg) administered orally exhibits no toxicity and broad spectrum, dose-dependent anti-tumor activity against a number of human tumor xenograft models, including MDA-MB-231, Colo-205, HT-29, DLD-1, NCI-H460, and A549. Sorafenib treatment significantly reduces tumor microvessel area (MVA) and microvessel density (MVD) in MDA MB-231, HT-29, and Colo-205 tumor xenografts, which is associated with its anti-tumor efficacy. However, it has no effect on MEK 1/2 phosphorylation or pERK 1/2 levels in HT-29 or MDA-MB-231 xenografts.[1] In SCID mice, sorafenib treatment results in a dose-dependent growth inhibition of PLC/PRF/5 tumor xenografts with TGIs of 49% and 78% at 10 mg/kg and 30 mg/kg, respectively. This is consistent with the inhibition of ERK and eIF4E phosphorylation, reduction of the microvessel area, and induction of tumor cell apoptosis. [2] By inhibiting NF-B mediated Mcl-1 and cIAP2 expression, sorafenib sensitizes bax-/- cells to TRAIL in a dose-dependent manner. In TRAIL-resistant HCT116 bax-/- and HT29 tumor xenografts, sorafenib (30–60 mg/kg) and TRAIL (5 mg/kg) showed dramatic efficacy. [3]
In nude mice bearing HT29 colon cancer xenografts, oral administration of Sorafenib (30 mg/kg, twice daily for 21 days) significantly inhibited tumor growth, with a tumor growth inhibition rate (TGI) of 68% compared to vehicle control. Tumor weight was reduced from 1.2 g (vehicle) to 0.38 g (treatment) [1] - Sorafenib (30 mg/kg, p.o., twice daily) reduced microvessel density (MVD) in HT29 xenografts by 52% as measured by CD31 immunohistochemistry, indicating anti-angiogenic activity [1] - In nude mice with A375 melanoma xenografts, Sorafenib (50 mg/kg, p.o., once daily for 28 days) achieved a TGI of 75%, with tumor volume reduced from 1200 mm³ (vehicle) to 300 mm³ (treatment) [3] - The drug (50 mg/kg, p.o.) inhibited the RAF-MEK-ERK pathway in A375 xenografts, with p-ERK levels reduced by 65% compared to vehicle-treated tumors [3] - In SCID mice bearing HepG2 hepatocellular carcinoma xenografts, Sorafenib (40 mg/kg, p.o., twice daily for 21 days) exhibited a TGI of 62% and reduced MVD by 48% [2] |
| Enzyme Assay |
Raf-1 (80 ng), wt BRAF (80 ng), or V599E BRAF (80 ng) are mixed with MEK-1 (1 μg) in the assay buffer (20 mM Tris (pH 8.2), 100 mM NaCl, 5 mM MgCl2, and 0.15% β-mercaptoethanol) to test the effects of the compound on different RAF kinase isoforms. Adding 25 μL of 10 μM γ-[33P]ATP (400 Ci/mol) and incubating the mixture at 32°C for 25 minutes kickstarts the RAF kinase assay (final volume of 50 μL). By filtering phosphorylated MEK-1 onto a phosphocellulose mat, radioactivity that is not bound to the protein is removed. Phosphorylated MEK-1 is then harvested. Using a β-plate counter, filter-bound radioactivity is measured after drying by microwave heating.
Kinase activity assay: Recombinant human kinases (VEGFR-2, VEGFR-3, PDGFR-β, RAF1, B-RAF, c-KIT, FLT3) were incubated with their respective peptide substrates, ATP, and various concentrations of Sorafenib (0.1 nM–10 μM) in assay buffer. The reaction was carried out at 30°C for 60 minutes and terminated by adding a stop reagent. Phosphorylated substrates were detected using a scintillation proximity assay, and IC50 values were calculated by nonlinear regression [2] |
| Cell Assay |
For 72 hours, Sorafenib tosylate is infused into cells at progressively higher concentrations. The Cell TiterGlo ATP Luminescent assay kit is used to calculate the number of cells. This assay counts the number of live cells in each well by measuring the luminescent signal, which is dependent on the amount of cellular ATP.
HUVEC proliferation assay: HUVECs were seeded in 96-well plates at 5×103 cells per well and cultured overnight. Cells were pretreated with Sorafenib (0.1–10 μM) for 1 hour, then stimulated with VEGF (50 ng/mL) for 72 hours. Cell viability was measured by MTT assay, and IC50 values for proliferation inhibition were determined [2] - HUVEC migration and tube formation assay: For migration, HUVECs were seeded in the upper chamber of transwell inserts, and Sorafenib (0.1–10 μM) plus VEGF (50 ng/mL) was added to the lower chamber. After 24 hours, migrated cells were stained and counted. For tube formation, HUVECs were seeded on Matrigel-coated plates with Sorafenib (0.1–10 μM) and VEGF (50 ng/mL), and tube structures were quantified after 6 hours [2] - Tumor cell proliferation assay: Human tumor cell lines (HT29, SW620, A375, etc.) were seeded in 96-well plates at 3×103 cells per well. After overnight culture, cells were treated with Sorafenib (0.1–30 μM) for 72 hours. Cell viability was assessed by MTT assay, and IC50 values were calculated [2] - Apoptosis assay: A375 cells were seeded in 6-well plates at 2×105 cells per well and treated with Sorafenib (1–10 μM) for 24 hours. Caspase-3/7 activity was measured using a luminescent assay kit, and Annexin V staining was performed followed by flow cytometry to detect apoptotic cells [3] - Western blot for signaling pathways: A375 cells or HT29 cells were treated with Sorafenib (1–10 μM) for 24 hours, then lysed. Protein extracts were separated by SDS-PAGE, transferred to membranes, and probed with antibodies against p-ERK, ERK, and β-actin. Band intensities were quantified using image analysis software [3] - Cytokine secretion assay: HT29 cells were seeded in 24-well plates and treated with Sorafenib (1–10 μM) for 48 hours. Culture supernatants were collected, and VEGF/bFGF concentrations were measured by ELISA [1] |
| Animal Protocol |
Mice: Female NCr-nu/nu mice are used. Mice bearing 75 to 150 mg tumors are treated orally with Sorafenib (7.5 to 60 mg/kg), administered daily for 9 days. In each model, Sorafenib produces dose-dependent tumor growth inhibition with no evidence of toxicity, as measured by increased weight loss relative to control animals or drug-related lethality. In parallel to the antitumor efficacy studies, additional groups of four mice bearing 100 to 200 mg tumors are treated orally with vehicle or Sorafenib (30 to 60 mg/kg), administered daily for 5 days, which is the shortest treatment duration producing complete tumor stasis in the treated groups.
Rats: Male albino rats weighing 100 to 120 g are used for the study. Rats are weighed and randomly split into three groups following an acclimatization period. For 8 weeks, the car is given daily to Group 1 (the healthy control group; n=10). An i.p. single dose of 200 mg/kg DENA is administered to Group 2 (the DENA group; n=15). Six weeks after receiving a DENA intravenously in Group 3 (the Sorafenib group; n=12), Sorafenib is administered orally at a dose of 10 mg/kg daily for two weeks. Rats are weighed, put to sleep with ether, killed at the conclusion of the experiment (8 weeks), and their livers are removed. Fresh liver is weighed after being dried on a clean paper towel and going through two ice-cold saline washes. The liver index is calculated using the formula liver weight (g)/final body weight (g)×100. HT29 colon cancer xenograft model: Nude mice (6–8 weeks old) were subcutaneously implanted with 5×106 HT29 cells. When tumors reached 100–150 mm³, mice were randomly divided into vehicle control and Sorafenib treatment groups (n=8 per group). The drug was dissolved in 10% DMSO + 90% Cremophor EL, diluted with saline (1:1), and administered orally at 30 mg/kg twice daily for 21 days. Tumor volume and body weight were measured every 3 days. At the end of treatment, tumors were excised for MVD analysis by CD31 immunohistochemistry [1] - A375 melanoma xenograft model: Nude mice were subcutaneously injected with 2×106 A375 cells. When tumors reached 150–200 mm³, mice were assigned to vehicle or Sorafenib groups (n=8 per group). The drug was prepared as above and administered orally at 50 mg/kg once daily for 28 days. Tumor volume was measured twice weekly. After sacrifice, tumors were collected for Western blot analysis of p-ERK [3] - HepG2 hepatocellular carcinoma xenograft model: SCID mice were subcutaneously implanted with 3×106 HepG2 cells. Once tumors reached 100–120 mm³, mice were treated with Sorafenib (40 mg/kg, p.o., twice daily) or vehicle for 21 days. Tumor weight and MVD (CD31 staining) were evaluated at the end of the study [2] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Multiple administrations over seven consecutive days can result in a 2.5 to 7-fold higher accumulation of the drug compared to a single dose. Steady-state plasma concentrations are reached within seven days, with a peak-to-trough ratio of less than 2. After twice-daily oral administration of 400 mg, the increase in mean Cmax and AUC is less than the dose-proportional increase. Tmax is approximately three hours. The mean relative bioavailability of sorafenib tablets is 38% to 49%. A high-fat diet can reduce bioavailability by 29%. After oral administration of 100 mg sorafenib, approximately 96% of the dose is recovered within 14 days, with 77% excreted in feces as glucuronidated metabolites and 19% excreted in urine as glucuronidated metabolites. Sorafenib is excreted unchanged in feces, accounting for 51% of the total dose. Sorafenib is widely distributed in tissues, indicating its lipophilic nature. After oral administration of 100 mg sorafenib solution, 96% of the dose was recovered within 14 days, with 77% excreted in feces as glucuronidated metabolites and 19% excreted in urine as glucuronidated metabolites. Unreacted sorafenib (51% of the total dose) was present in feces but not detected in urine. After administration of Nexavar tablets, the mean relative bioavailability was 38-49% compared to the oral solution. Peak plasma concentrations of sorafenib were reached approximately within 3 hours after oral administration. Bioavailability was similar after a moderate-fat meal (30% fat; 700 calories) compared to the fasting state. Bioavailability decreased by 29% after a high-fat meal (50% fat; 900 calories) compared to the fasting state. Nexavar is recommended to be taken on an empty stomach. Following a twice-daily oral dose of 400 mg, the increase in mean Cmax and AUC was less than the proportion of dose increases. Sorafenib binds to human plasma proteins in vitro in 99.5%. The absorption and basic pharmacokinetics of sorafenib tosylate following a single dose were evaluated in female CD-1 mice, male Wistar rats, and female beagle dogs. To determine sorafenib absorption in rats, bile duct cannulated rats (n=5 per group) were used. Twenty-four hours post-surgery, rats were administered (14)C-sorafenib tosylate orally or intravenously at a dose of 5 mg/kg. Absorption of sorafenib was nearly complete in female CD-1 mice (78.6%) and male Wistar rats (79.2%). In beagle dogs, the absorption rate (67.6%, calculated based on AUC values after intravenous and oral administration) and absolute bioavailability (59.9%) were lower than in rodents. Peak plasma radioactivity was observed in all species within 1.5 to 2 hours following oral administration. Following intravenous administration of 14C-sorafenib tosylate, the elimination of the radioactive material from plasma in mice, rats, and dogs exhibited similar terminal half-lives of 6.8 h, 8.8 h, and 7.3 h, respectively. After oral administration, the radioactive terminal half-lives in mice and dogs were 6.1 h and 5.8 h, respectively. In rats, the terminal half-life after oral administration (11.2 h) was longer than that after intravenous administration. In rats, the elimination rate of the parent compound (half-life: 9.3 h) was slower than that in mice (half-life: 6.5 h) and dogs (half-life: 4.3 h). The total plasma clearance in rats was 0.044 L/(hr/kg), equivalent to a blood clearance of 0.049 L/(hr/kg). The total plasma clearance in mice and dogs was 0.13 L/(hr/kg) and 0.15 L/(hr/kg), respectively. The steady-state volume of distribution ranges from 0.65 L/kg to 0.74 L/kg, depending on the species. Metabolisms/Metabolites Sorafenib is oxidatively metabolized in the liver via CYP3A4 and glucuronidated in the liver and kidneys via UGT1A9. At steady state, sorafenib accounts for 70-85% of the circulating analytes in plasma. Approximately eight metabolites of sorafenib have been identified, five of which are detectable in plasma. The major circulating metabolite is pyridine N-oxide, which accounts for approximately 9-16% of the total circulating dose at steady state; the pharmacological activity of this metabolite is comparable to that of the parent drug. Sorafenib is oxidatively metabolized in the liver via CYP3A4 and also glucuronidated via UGT1A9. CYP3A4 activity inducers can reduce systemic exposure to sorafenib. At steady state, sorafenib accounts for approximately 70-85% of the circulating analytes in plasma. Eight metabolites of sorafenib have been identified, five of which are detectable in plasma. The major circulating metabolite of sorafenib, pyridine N-oxide, accounts for approximately 9-16% of the circulating analyte at steady state and has similar in vitro activity to sorafenib. Known metabolites of sorafenib include sorafenib and AD-glucuronide (discontinued). Sorafenib is primarily metabolized in the liver, mainly through CYP3A4-mediated oxidative metabolism and UGT1A9-mediated glucuronidation metabolism. At steady state, sorafenib accounts for approximately 70-85% of the circulating analyte in plasma. Eight metabolites of sorafenib have been identified, five of which are detectable in plasma. The major circulating metabolite of sorafenib in plasma, pyridine N-oxide, has similar in vitro activity to sorafenib. This metabolite accounts for approximately 9-16% of the circulating analyte at steady state. Elimination route: Following oral administration of 100 mg sorafenib solution, 96% of the dose was recovered within 14 days, of which 77% was excreted in feces and 19% in urine as glucuronidated metabolites. Half-life: 25–48 hours Biological half-life The mean elimination half-life of sorafenib is approximately 25 to 48 hours. Intravenous injection of (14)C-sorafenib tosylate into mice, rats, and dogs resulted in similar terminal half-lives of radioactive material elimination from plasma, at 6.8 hours, 8.8 hours, and 7.3 hours, respectively. The terminal half-life of radioactive material after oral administration was 6.1 hours in mice and 5.8 hours in dogs. The terminal half-life after oral administration in rats (11.2 hours) was longer than that after intravenous administration. The elimination rate of unmetabolized compounds in rats (half-life: 9.3 h) was slower than that in mice (half-life: 6.5 h) and dogs (half-life: 4.3 h). The mean elimination half-life of sorafenib was approximately 25 to 48 hours. The oral bioavailability of sorafenib (10 mg/kg) in rats was 38%. Sorafenib distributed well in mice, with the highest concentrations in the liver, kidneys, and tumor tissues. Two hours after oral administration (30 mg/kg), the concentrations in tumor tissue were 2.8 μg/g, in liver 4.2 μg/g, and in kidney 3.5 μg/g. |
| Toxicity/Toxicokinetics |
Toxicity Summary
Sorafenib interacts with a variety of intracellular kinases (CRAF, BRAF, and mutant BRAF) and cell surface kinases (KIT, FLT-3, VEGFR-2, VEGFR-3, and PDGFR-2). Some of these kinases are believed to be involved in angiogenesis, thus sorafenib can reduce blood flow to tumors. Sorafenib is unique in that it targets the Raf/Mek/Erk pathway. By inhibiting these kinases, the transcription of genes involved in cell proliferation and angiogenesis is suppressed. Hepatotoxicity Elevated serum transaminase levels are common in large clinical trials of sorafenib, occurring in up to half of patients, but only 1% to 3% of subjects had values exceeding the upper limit of normal (ULN) by more than 5 times. Furthermore, there have been several case reports of clinically significant liver injury during sorafenib treatment; these injuries are usually severe and occasionally fatal. Acute liver injury occurs from several days to 8 weeks after starting sorafenib, typically in a hepatocellular pattern with significantly elevated serum transaminase levels. No immune hypersensitivity or autoimmune features were observed. Patients usually recover rapidly after discontinuing sorafenib, but some cases develop progressive liver injury and liver failure. Most reports of severe liver injury occur in patients receiving treatment for hepatocellular carcinoma with concurrent cirrhosis, or in patients taking other potentially hepatotoxic drugs.31 Probability score: B (Possibly the cause of clinically significant liver injury). Use during pregnancy and lactation ◉ Overview of use during lactation There is currently no information on the clinical use of sorafenib during lactation. Due to the high plasma protein binding rate of sorafenib (99.5%), its concentration in breast milk is likely to be low. However, its half-life of 25 to 48 hours may allow it to accumulate in the infant. The manufacturer recommends discontinuing breastfeeding during sorafenib treatment and for 2 weeks after the last dose. ◉ Effects on breastfed infants No published information found as of the revision date. ◉ Effects on lactation and breast milk No published information found as of the revision date. Protein binding In vitro studies showed that sorafenib binds to human plasma proteins in 99.5%. Interactions Sorafenib does not appear to affect the metabolism of warfarin (CYP2C9 substrate) in vivo; the mean baseline change in prothrombin time (PT)/international normalized ratio (INR) in patients treated with sorafenib did not appear to be greater than in the placebo group. However, some patients receiving both warfarin and sorafenib have reported occasional bleeding events or elevated INR. Sorafenib may have pharmacokinetic interactions with doxorubicin and irinotecan (increased area under the serum concentration-time curve (AUC) of doxorubicin, irinotecan, and their active metabolite SN-38). The clinical significance of these findings is unclear, and caution is advised. In vitro studies have shown that sorafenib inhibits glucuronidation via the uridine diphosphate glucuronyl transferase (UGT) 1A1 and 1A9 pathways; pharmacokinetic interactions may exist (increased systemic exposure to UGT 1A1 or 1A9 substrates). Caution should be exercised when sorafenib is used concomitantly with drugs primarily metabolized via the UGT 1A1 pathway (e.g., irinotecan, whose active metabolite SN-38 is metabolized by UGT 1A1). In vitro studies using human liver microsomes showed that sorafenib inhibited CYP isoenzymes 2B6, 2C8, 2C9, 2C19, 2D6, and 3A4; however, sorafenib did not appear to alter exposure to dextromethorphan (CYP2D6 substrate), midazolam (CYP3A4 substrate), or omeprazole (CYP2C19 substrate). The manufacturer states that sorafenib is unlikely to alter the metabolism of CYP isoenzymes 2C19, 2D6, or 3A4 substrates in vivo, nor is it likely to induce CYP isoenzymes 1A2 or 3A4. However, sorafenib may increase systemic exposure to CYP2B6 or CYP2C8 substrates; caution should be exercised when sorafenib is used concomitantly with CYP2B6 or CYP2C8 substrates. For more complete data on sorafenib interactions (6 items in total), please visit the HSDB record page. In vitro experiments showed that treatment with sorafenib (at concentrations up to 30 μM) for 72 hours had no effect on the viability of normal human foreskin fibroblasts (NHF), indicating that it has selective toxicity to tumor cells[2]. - No significant weight loss (>10%) or behavioral abnormalities were observed in mice after treatment with sorafenib (30-50 mg/kg, orally) for 21-28 days. Serum ALT, AST and creatinine levels were all within the normal range, suggesting no significant toxicity[1][2][3]. - Sorafenib has a plasma protein binding rate of 99.5% in human plasma[2]. |
| References | |
| Additional Infomation |
Therapeutic Uses
Anti-tumor drug; protein kinase inhibitor Nexavar is indicated for the treatment of patients with unresectable hepatocellular carcinoma (HCC). /US product label includes/ Nexavar is indicated for the treatment of patients with locally recurrent or metastatic progressive differentiated thyroid cancer (DTC) that is unresponsive to radioactive iodine therapy. /US product label includes/ Nexavar is indicated for the treatment of patients with advanced renal cell carcinoma (RCC). /US product label includes/ Drug Warnings Hand-foot syndrome (commonly known as hand-foot syndrome) and rash are common adverse reactions to sorafenib. In clinical studies, these adverse reactions occurred in 30% and 40% of patients receiving this drug, respectively, compared to 7% and 16% of patients receiving placebo, respectively. Cumulative event rate analysis showed that rash and hand-foot syndrome are usually grade 1 or 2 and generally occur within the first 6 weeks of sorafenib treatment. Management of skin toxicity may include topical treatment to relieve symptoms, temporary interruption of treatment, and/or adjustment of the sorafenib dose; for severe or persistent cases, permanent discontinuation of sorafenib treatment may be necessary. The risk of bleeding may be increased. In clinical studies, bleeding was reported in 15.3% of patients treated with sorafenib (regardless of causality) and in 8.2% of patients treated with placebo. The incidence of grade 3 and 4 bleeding was 2% and 0% in patients treated with sorafenib, respectively, compared to 1.3% and 0.2% in patients treated with placebo, respectively. One fatal bleeding event occurred in each treatment group. Permanent discontinuation of sorafenib should be considered if any bleeding event requires medical intervention. Gastrointestinal perforation is rare in patients treated with sorafenib, sometimes with obvious intra-abdominal tumors. If gastrointestinal perforation occurs, sorafenib treatment should be discontinued. Based on its mechanism of action and animal studies, sorafenib may cause fetal harm when taken by pregnant women. In animal studies, sorafenib caused embryo-fetal toxicity when maternal exposure was significantly lower than the human exposure at the recommended dose (400 mg twice daily). Due to the potential harm to the fetus, women of childbearing age should be advised to avoid pregnancy while taking sorafenib. For more complete data on sorafenib warnings (16 in total), please visit the HSDB record page. Pharmacodynamics Sorafenib reduces tumor cell proliferation in vitro. It inhibits the growth of human tumor xenografts in immunodeficient mice, reduces tumor angiogenesis, and increases tumor cell apoptosis in hepatocellular carcinoma, renal cell carcinoma, and differentiated thyroid carcinoma models. Some studies have shown that sorafenib can induce apoptosis in various tumor cell lines, but this effect is not consistent across different cell lines. The antiviral activity of sorafenib has been confirmed; in vitro experiments have shown that it can inhibit hepatitis C virus replication. Sorafenib (Bay 43-9006) is a multi-target kinase inhibitor with a dual mechanism of action: inhibiting tumor angiogenesis by targeting VEGFR/PDGFR and inhibiting tumor cell proliferation by targeting RAF kinase [2][3]. - Sorafenib has oral bioavailability and has shown broad-spectrum antitumor activity against a variety of solid tumors, including colon cancer, melanoma, and hepatocellular carcinoma, in preclinical models [2]. - Sorafenib's anti-angiogenic effect is achieved by inhibiting VEGF-induced endothelial cell proliferation, migration, and tubular formation, while its anti-proliferative effect involves blocking the RAF-MEK-ERK signaling pathway in tumor cells [1][3]. |
| Molecular Formula |
C21H16CLF3N4O3
|
|---|---|
| Molecular Weight |
464.82
|
| Exact Mass |
464.086
|
| Elemental Analysis |
C, 54.26; H, 3.47; Cl, 7.63; F, 12.26; N, 12.05; O, 10.33
|
| CAS # |
284461-73-0
|
| Related CAS # |
Sorafenib Tosylate;475207-59-1;Sorafenib-d3;1130115-44-4;Sorafenib-d4;1207560-07-3;Sorafenib-13C,d3;1210608-86-8
|
| PubChem CID |
216239
|
| Appearance |
white solid powder
|
| Density |
1.5±0.1 g/cm3
|
| Boiling Point |
523.3±50.0 °C at 760 mmHg
|
| Melting Point |
202-204°C
|
| Flash Point |
270.3±30.1 °C
|
| Vapour Pressure |
0.0±1.4 mmHg at 25°C
|
| Index of Refraction |
1.626
|
| LogP |
5.16
|
| Hydrogen Bond Donor Count |
3
|
| Hydrogen Bond Acceptor Count |
7
|
| Rotatable Bond Count |
5
|
| Heavy Atom Count |
32
|
| Complexity |
646
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
FC(F)(F)C1C=C(NC(NC2C=CC(=CC=2)OC2=CC=NC(C(=O)NC)=C2)=O)C=CC=1Cl
|
| InChi Key |
MLDQJTXFUGDVEO-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C21H16ClF3N4O3/c1-26-19(30)18-11-15(8-9-27-18)32-14-5-2-12(3-6-14)28-20(31)29-13-4-7-17(22)16(10-13)21(23,24)25/h2-11H,1H3,(H,26,30)(H2,28,29,31)
|
| Chemical Name |
4-[4-[[4-chloro-3-(trifluoromethyl)phenyl]carbamoylamino]phenoxy]-N-methylpyridine-2-carboxamide
|
| Synonyms |
BAY 43-9006; BAY-439-006; BAY439006; BAY-439006; BAY 439006; BAY 549085; trade name: Nexavar; SFN
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: 4 mg/mL (8.61 mM) in 2% DMSO + 40% PEG300 + 5% Tween80 + 53% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication.
Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.08 mg/mL (4.47 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.08 mg/mL (4.47 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: ≥ 2.08 mg/mL (4.47 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL corn oil and mix evenly. Solubility in Formulation 5: 5%DMSO+45%PEG400+50%H2O: 0.375mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1514 mL | 10.7569 mL | 21.5137 mL | |
| 5 mM | 0.4303 mL | 2.1514 mL | 4.3027 mL | |
| 10 mM | 0.2151 mL | 1.0757 mL | 2.1514 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Prospective, Non-interventional, Post-authorization Safety Study That Includes All Patients Diagnosed as Unresectable Differentiated Thyroid Carcinoma and Treated With Sorafenib
CTID: NCT02185560
Phase: Status: Active, not recruiting
Date: 2024-11-20
The number of nuclei breaking the internal limiting membrane (ILM). A: Controlled group; B: ROP group; C: Vehicle-treated ROP group; D: Low doses sorafenib-treated ROP group; E: Middle doses sorafenib-treated ROP group; F: High dose sorafenib-treated ROP group. |
 |
 |