4-PBA sodium; Sodium 4-phenylbutyrate; Sodium phenylbutyrate; 4-phenylbutyrate (4-PBA); Sodium 4-phenylbutyrate; SODIUM PHENYLBUTYRATE; 1716-12-7; sodium 4-phenylbutanoate; Buphenyl; Ammonaps; Benzenebutanoic acid, sodium salt; TriButyrate; 4-phenylbutyric acid; Buphenyl

| Size | Price | Stock | Qty |
|---|---|---|---|
| 100mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| 5g |
|
||
| 10g |
|
||
| Other Sizes |
Purity: ≥98%
Sodium phenylbutyrate (4-Phenylbutyric acid sodium; 4-PBA sodium), an orphan drug marketed by Ucyclyd Pharma, is a novel and potent histone deacetylase (HDAC) inhibitor used as an as adjunctive therapy for chronic treatment of urea cycle disorders involving deficiencies of argininosuccinic acid synthetase (AS), ornithine transcarbamylase (OTC), or carbamylphosphate synthetase (CPS). Because metabolites of sodium phenylbutyrate offer an alternative pathway to the urea cycle for the excretion of excess nitrogen. It has strong anti-cancer properties as well.
| Targets |
HDAC/Histone Deacetylases
Histone Deacetylases (HDACs): In human non-small cell lung cancer (NSCLC) cell lines (A549, H460), the IC50 of Sodium Phenylbutyrate (4-PBA sodium) for HDAC inhibition (assessed by histone H4 acetylation) was 2.5 μM (A549) and 3.1 μM (H460) [2] - Endoplasmic Reticulum (ER) Stress-Related Proteins (GRP78, CHOP): In human neuroblastoma SH-SY5Y cells treated with ER stress inducer (tunicamycin), Sodium Phenylbutyrate (4-PBA sodium) showed an EC50 of 4.2 μM for reducing GRP78 (ER stress marker) expression and an EC50 of 3.8 μM for inhibiting CHOP (pro-apoptotic ER stress protein) expression [5] |
|---|---|
| ln Vitro |
In vitro activity: Phenylbutyrate is a well-known HDAC inhibitor that has neuroprotective properties in addition to increasing the transcription of several genes. In the substantia nigra, phenylbutyrate significantly reduces the effects of MPTP-induced striatal dopamine depletion and tyrosine hydroxylase-positive neuronal loss.[1] In prostate cancer cells, phenylbutyrate reduces the expression of the pro-angiogenic vascular endothelial growth factor, the pro-angiogenic vascular endothelial growth factor, the prostate progression marker caveolin-1, and the double-strand break repair protein DNA-dependent protein kinase. It is discovered that phenylbutyrate and ionizing radiation work together to cause prostate cancer cells to undergo apoptosis.[2]
In human NSCLC cell lines (A549, H460): Sodium Phenylbutyrate (4-PBA sodium) inhibited cell proliferation in a dose- and time-dependent manner. At 72 h treatment, the IC50 for proliferation inhibition was 2.8 μM (A549) and 3.5 μM (H460). Flow cytometry analysis showed that 3 μM treatment for 48 h increased apoptotic rates from 3.2% (control) to 28.5% (A549) and 25.3% (H460). Western blot revealed increased acetylation of histone H3/H4, upregulated p21WAF1/CIP1 (cell cycle inhibitor), and downregulated cyclin D1 (cell cycle promoter) [2] - In rat primary cortical neurons exposed to oxygen-glucose deprivation (OGD, a model of cerebral ischemia): Sodium Phenylbutyrate (4-PBA sodium) at 5 μM improved cell viability from 42% (OGD control) to 76% after 24 h reoxygenation. It reduced lactate dehydrogenase (LDH) release (a marker of cell damage) from 280 U/L (OGD control) to 135 U/L. Western blot showed decreased cleaved caspase-3 (apoptosis marker) and increased Bcl-2 (anti-apoptotic protein) [3] - In human hepatocellular carcinoma HepG2 cells treated with palmitic acid (PA, an inducer of ER stress and lipotoxicity): Sodium Phenylbutyrate (4-PBA sodium) at 4 μM reduced PA-induced cell death (viability increased from 51% to 82% at 48 h). It suppressed PA-induced ER stress: GRP78 expression decreased by 65%, CHOP expression decreased by 70%, and phosphorylated eIF2α (ER stress marker) decreased by 60% (Western blot data) [4] - In human glioblastoma U87MG cells: Sodium Phenylbutyrate (4-PBA sodium) at 3 μM inhibited cell migration (transwell assay: migrated cells reduced by 58% vs. control) and invasion (Matrigel assay: invasive cells reduced by 62% vs. control) at 24 h. PCR results showed downregulated mRNA levels of MMP-2 and MMP-9 (matrix metalloproteinases involved in invasion) by 55% and 60%, respectively [5] - In mouse motor neuron-like NSC-34 cells (a model of amyotrophic lateral sclerosis, ALS): Sodium Phenylbutyrate (4-PBA sodium) at 5 μM reduced mutant SOD1-induced aggregate formation (aggregate-positive cells decreased from 45% to 18% at 72 h). It also increased the clearance of mutant SOD1 aggregates via upregulating autophagy (LC3-II/LC3-I ratio increased by 2.3-fold, Western blot) [6] |
| ln Vivo |
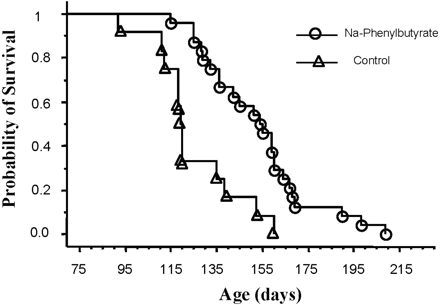
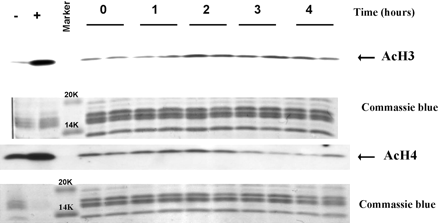
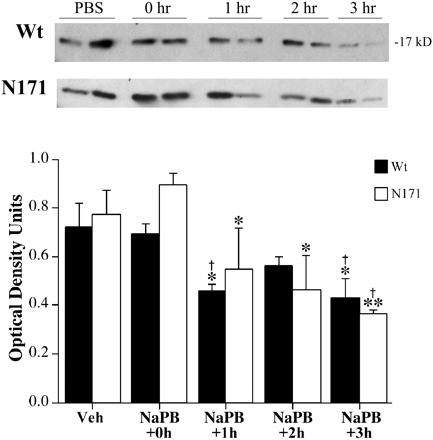
Phenylbutyrate greatly increases survival and improves both the clinical and neuropathological phenotypes in G93A transgenic ALS mice. Administration of phenylbutyrate reduces the expression of cytochrome c and caspase, but ameliorates the histone hypoacetylation seen in G93A mice and induces the expression of nuclear factor-kappaB (NF-kappaB) p50, the phosphorylated inhibitory subunit of NF-kappaB (pIkappaB), and beta cell lymphoma 2 (bcl-2). Phenylbutyrate either directly acetylates NF-kappaB p50 or phosphorylates IkappaB, causing NF-kappaB p50 to be translocated to the nucleus.[3] Phenylbutyrate increases brain histone acetylation and decreases histone methylation levels as determined by immunocytochemistry and Western blots. in a transgenic mousemodel of Huntington's disease (HD). Phenylbutyrate down-regulated caspases linked to apoptotic cell death, increased mRNA for ubiquitin-proteosomal pathway components, and increased caspase 3 immunoreactivity in the striatum.[4]
In nude mice bearing A549 NSCLC xenografts: Mice were divided into control (saline) and Sodium Phenylbutyrate (4-PBA sodium) groups (100 mg/kg, intraperitoneal injection, once daily for 21 days). The treatment group showed a 62% reduction in tumor volume (from 950 mm³ to 361 mm³) and a 58% decrease in tumor weight (from 1.1 g to 0.46 g) vs. control. Immunohistochemistry of tumor tissues showed increased acetyl-histone H4 (by 2.1-fold) and cleaved caspase-3 (by 2.5-fold), and decreased Ki-67 (proliferation marker, by 45%) [2] - In Sprague-Dawley (SD) rats with middle cerebral artery occlusion (MCAO, a model of cerebral ischemia): Sodium Phenylbutyrate (4-PBA sodium) was administered at 150 mg/kg via intraperitoneal injection immediately after MCAO and once daily for 3 days. At 7 days post-MCAO, the infarct volume (assessed by TTC staining) decreased from 45% (control) to 22% of the ipsilateral hemisphere. Neurological deficit scores (0–5 scale) improved from 3.8 (control) to 1.6. Western blot of brain tissues showed increased Bcl-2 and decreased cleaved caspase-3 [3] - In SOD1G93A transgenic mice (an ALS model): Sodium Phenylbutyrate (4-PBA sodium) was given by oral gavage at 200 mg/kg once daily from postnatal day 60 to endpoint. The treatment prolonged median survival by 18 days (control: 128 days; treatment: 146 days). It delayed the onset of motor deficits (rotarod test: time on rod decreased to 50% of baseline at day 112 in control vs. day 126 in treatment). Spinal cord immunohistochemistry showed reduced mutant SOD1 aggregates and increased motor neuron survival (by 35%) [6] |
| Enzyme Assay |
Phenylbutyrate (PB) is a histone deacetylase inhibitor that has been shown to induce differentiation and apoptosis in various cancer cell lines. Although these effects are most likely due to modulation of gene expression, the specific genes and gene products responsible for the effects of PB are not well characterized. In this study, we used cDNA expression arrays and Western blot to assess the effect that PB has on the expression of various cancer and apoptosis-regulatory gene products. We show that PB attenuates the expression of the apoptosis antagonist Bcl-X(L), the double-strand break repair protein DNA-dependent protein kinase, the prostate progression marker caveolin-1, and the pro-angiogenic vascular endothelial growth factor. Furthermore, PB was found to act in synergy with ionizing radiation to induce apoptosis in prostate cancer cells. Taken together, our results point to the possibility that PB may be an effective anti-prostate cancer agent when used in combination with radiation or chemotherapy and for the inhibition of cancer progression.[2]
HDAC Activity Assay (for NSCLC research): Prepare reaction mixtures containing recombinant human HDAC1/2/3, fluorogenic substrate (succinyl-lysine-7-amino-4-methylcoumarin), and Sodium Phenylbutyrate (4-PBA sodium) (0.5–10 μM) in assay buffer. Incubate at 37°C for 60 minutes. Add a developer solution (containing trypsin) to cleave the deacetylated substrate and release fluorescent 7-amino-4-methylcoumarin. Measure fluorescence intensity at excitation 360 nm and emission 460 nm. Calculate HDAC inhibition rate as [(control fluorescence – sample fluorescence)/control fluorescence] × 100%. Plot dose-response curves to determine IC50 values for each HDAC subtype [2] - ER Stress-Related Protein Expression Assay (for neuroblastoma cells): Seed SH-SY5Y cells in 6-well plates and treat with tunicamycin (1 μg/mL) to induce ER stress. Add Sodium Phenylbutyrate (4-PBA sodium) (1–10 μM) and incubate for 24 h. Lyse cells to extract total protein, then perform Western blot. Use primary antibodies against GRP78 and CHOP, and secondary antibodies conjugated to horseradish peroxidase. Detect chemiluminescence signals and quantify band intensity using image analysis software. Calculate the concentration of Sodium Phenylbutyrate (4-PBA sodium) required to reduce protein expression by 50% (EC50) [5] |
| Cell Assay |
In brief, viable cells are seeded at a density of 4 × 104 cells/mL in 60-mm dishes in RPMI 1640 with 10% fetal bovine serum and 0.35% agarose on a base layer of 0.7% agarose. Viable cells are determined by trypan blue dye exclusion. Additions of DMSO, TSA, or PB are made to the upper and lower agarose layers. Colonies are counted after 10–14 days, and assays are run in triplicate at least three times.
NSCLC Cell Proliferation Assay: Seed A549/H460 cells in 96-well plates at 4×10³ cells/well. After 24 h attachment, treat with Sodium Phenylbutyrate (4-PBA sodium) (0.5, 1, 2, 4, 8 μM; control: vehicle). Incubate for 24, 48, 72 h. Add MTT reagent (5 mg/mL) and incubate for 4 h. Remove supernatant, add DMSO to dissolve formazan crystals. Measure absorbance at 570 nm. Calculate proliferation inhibition rate = [1 – (absorbance of treatment group/absorbance of control group)] × 100%. Determine IC50 using GraphPad Prism software [2] - Primary Cortical Neuron OGD Assay: Isolate cortical neurons from E18 SD rat embryos and culture for 7 days. Expose neurons to OGD buffer (glucose-free, 95% N₂/5% CO₂) for 2 h, then reoxygenate in normal medium. Add Sodium Phenylbutyrate (4-PBA sodium) (1, 3, 5, 10 μM) during reoxygenation. After 24 h, measure cell viability using CCK-8 assay (absorbance at 450 nm) and LDH release using a commercial kit (absorbance at 490 nm) [3] - HepG2 ER Stress Assay: Seed HepG2 cells in 6-well plates at 2×10⁵ cells/well. Incubate for 24 h, then treat with palmitic acid (0.5 mM) alone or with Sodium Phenylbutyrate (4-PBA sodium) (1, 2, 4, 8 μM). Incubate for 48 h. For cell viability: use trypan blue exclusion assay (count viable cells using a hemocytometer). For ER stress markers: extract total protein, perform Western blot with antibodies against GRP78, CHOP, and phosphorylated eIF2α. Quantify band intensity relative to β-actin (loading control) [4] - U87MG Migration/Invasion Assay: For migration: seed U87MG cells in the upper chamber of transwell inserts (8 μm pores) at 5×10⁴ cells/chamber, add Sodium Phenylbutyrate (4-PBA sodium) (1, 3, 5 μM) to the upper chamber. For invasion: coat inserts with Matrigel before seeding. Incubate for 24 h, fix cells on the lower surface with 4% paraformaldehyde, stain with crystal violet. Count stained cells under a microscope (5 fields/insert). Calculate migration/invasion inhibition rate vs. control [5] |
| Animal Protocol |
Mice: 10-week-old female IRC's pathogen-free animal facility is home to C57BL/6J mice. The animals are divided into four groups at random: LPS (n=6), vehicle+Benzenebutyric acid (n=6), vehicle control (n=5), and LPS+Benzenebutyric acid (n=6). For three weeks, mice are given 200 μL of phosphate-buffered saline (PBS) once a week at a dose of 5 mg/kg intraperitoneally. Mice are given a daily intraperitoneal injection of 200 μL PBS (or PBS as a vehicle) containing 240 mg/kg of sodium 4-phenylbutyrate (Benzenebutyric acid) solution, which is made by titrating equimolecular amounts of Benzenebutyric acid and sodium hydroxide to reach pH 7.4. The injections last for three weeks. Mice are killed by asphyxia with CO2. The right femur is scanned in order to ascertain the long bone's microarchitecture and bone mineral density (BMD). A 6.9 μm effective detector pixel size and a 77–255 mg/cc threshold are used for the scans. A region of 1.6 mm in length, situated 0.1 mm below the distal femur growth plate, is the subject of analysis for trabecular bone.
NSCLC Xenograft Mouse Model: Female nude mice (6–8 weeks old) were injected subcutaneously with 5×10⁶ A549 cells into the right flank. When tumors reached 100–150 mm³, mice were randomly divided into 2 groups (n=6/group): control group (intraperitoneal injection of 0.9% saline, once daily) and Sodium Phenylbutyrate (4-PBA sodium) group (intraperitoneal injection of 100 mg/kg Sodium Phenylbutyrate (4-PBA sodium) dissolved in 0.9% saline, once daily). Treatments continued for 21 days. Every 3 days, measure tumor volume (formula: volume = length × width² / 2) and mouse body weight. At the end of treatment, sacrifice mice, excise tumors and weigh. Collect tumor tissues for immunohistochemistry (acetylated histone H4, cleaved caspase-3, Ki-67) [2] - Rat MCAO Model: Male SD rats (250–300 g) were anesthetized, and the middle cerebral artery was occluded using a nylon suture for 90 minutes. Immediately after suture removal (reperfusion), rats were randomly divided into control (intraperitoneal injection of saline, once daily) and Sodium Phenylbutyrate (4-PBA sodium) group (intraperitoneal injection of 150 mg/kg Sodium Phenylbutyrate (4-PBA sodium) dissolved in saline, once daily for 3 days). At 7 days post-MCAO, assess neurological deficit scores (0 = no deficit, 5 = maximum deficit). Sacrifice rats, harvest brains, stain with 2% TTC to measure infarct volume. Extract brain tissue proteins for Western blot (Bcl-2, cleaved caspase-3) [3] - SOD1G93A Transgenic Mouse ALS Model: Male SOD1G93A transgenic mice (C57BL/6 background) were used. From postnatal day 60, mice were divided into control (oral gavage of 0.5% carboxymethyl cellulose, once daily) and Sodium Phenylbutyrate (4-PBA sodium) group (oral gavage of 200 mg/kg Sodium Phenylbutyrate (4-PBA sodium) suspended in 0.5% carboxymethyl cellulose, once daily). Monitor mice daily for survival and motor function (rotarod test: 5 rpm, record time on rod until fall). At endpoint, sacrifice mice, collect spinal cords for immunohistochemistry (mutant SOD1 aggregates, motor neuron count) [6] |
| ADME/Pharmacokinetics |
In male SD rats (250–300 g), a single intraperitoneal injection of 150 mg/kg sodium chlorate (4-PBA sodium) was administered: plasma concentration-time curves were determined by high performance liquid chromatography (HPLC). The maximum plasma concentration (Cmax) was 85.2 μg/mL 0.5 h after administration. The area under the plasma concentration-time curve (AUC₀₋∞) was 248.6 μg·h/mL. The elimination half-life (t₁/₂) was 2.3 h. The urinary excretion within 24 h was 38.5% of the administered dose (mainly excreted unchanged) [3]
|
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation
◉ Overview of Drug Use During Lactation Currently, there is no information regarding the clinical use of sodium phenylbutyrate or sodium phenylbutyrate in combination with tauroursodeoxycholic acid during lactation. Both sodium phenylbutyrate and tauroursodeoxycholic acid are highly protein-bound, making it unlikely that they will enter breast milk in clinically significant amounts. If a mother needs to use sodium phenylbutyrate (whether or not in combination with tauroursodeoxycholic acid), this is not a reason to discontinue breastfeeding. These products should be used with caution during lactation, especially when breastfeeding newborns or premature infants, until more data become available. Monitoring the breastfed infant for neurotoxicity (excessive sedation, vomiting) may be advisable while the mother is receiving treatment. ◉ Effects on Breastfed Infants No relevant published information was found as of the revision date. ◉ Effects on Lactation and Breast Milk No relevant published information was found as of the revision date. In nude mice treated with 100 mg/kg sodium chlorate (4-PBA sodium) (intraperitoneal injection, 21 days): no significant weight loss (weight change: -3.2% vs. control group: +2.1%, P > 0.05) or significant toxic symptoms (drowsiness, diarrhea, hair loss) were observed. Serum biochemical parameters: ALT (28.5 U/L vs. control group 26.3 U/L), AST (45.2 U/L vs. control group 42.8 U/L), BUN (15.3 mg/dL vs. control group 14.8 mg/dL) and creatinine (0.8 mg/dL vs. control group 0.75 mg/dL) were not significantly different from the control group [2] - In SD rats treated with 150 mg/kg sodium chlorate (4-PBA sodium) (intraperitoneal injection, 3 days): no significant necrosis or inflammation was observed in liver and kidney histopathology. Plasma protein binding (measured by ultrafiltration) was 42.3% [3] - In SOD1G93A mice treated with 200 mg/kg sodium chlorate (4-PBA sodium) (orally, for approximately 80 days): there were no significant changes in food intake or body weight (treatment group body weight: 28.5 g, control group body weight: 29.2 g, day 120). Serum electrolytes (Na⁺, K⁺, Cl⁻) and pH were within the normal range [6] |
| References | |
| Additional Infomation |
Sodium phenylbutyrate is the organosodium salt of 4-phenylbutyric acid. It is a prodrug of phenylacetic acid used to treat urea cycle disorders. It has multiple functions, including as a prodrug, an EC 3.5.1.98 (histone deacetylase) inhibitor, a neuroprotective agent, an orphan drug, and an anti-aging agent. It contains a 4-phenylbutyrate group. Sodium chlorate is the sodium salt of phenylbutyric acid, a derivative of the short-chain fatty acid butyrate, which has potential antitumor activity. Phenylic acid reversibly inhibits class I and II histone deacetylases (HDACs), which may lead to an overall increase in gene expression, a decrease in cell proliferation, an increase in cell differentiation, and the induction of apoptosis in susceptible tumor cell populations. See also: phenylbutyric acid (containing the active moiety); sodium chlorate; tauroursodeoxycholic acid (component). Drug Indications Tranexamic acid is indicated as adjunctive therapy for chronic urea cycle disorders, including deficiencies in carbamoyl phosphate synthase, ornithine transcarbamoylase, or argininosuccinate synthase. Suitable for all patients with neonatal onset (complete enzyme deficiency, onset within 28 days of birth). Also suitable for patients with late-onset disease (partial enzyme deficiency, onset after the first month of life) with a history of hyperammonemia encephalopathy. Chronic treatment of urea cycle disorders. Sodium chlorate (4-PBA sodium) is a small molecule compound with a dual mechanism: it regulates histone acetylation by inhibiting HDACs and modulates endoplasmic reticulum stress by inhibiting the unfolded protein response (UPR). In non-small cell lung cancer, its antitumor effect is mainly mediated by HDAC inhibition-induced cell cycle arrest and apoptosis[2] - In a model of cerebral ischemia, sodium chlorate (4-PBA sodium) exerts neuroprotective effects by reducing apoptosis and oxidative stress, which is associated with the upregulation of anti-apoptotic protein (Bcl-2) and the downregulation of pro-apoptotic factor (cleaved caspase-3)[3] - Clinically, sodium chlorate (4-PBA sodium) was initially approved for the treatment of urea cycle disorders (UCDs) to reduce ammonia levels. In preclinical studies, sodium chlorate (4-PBA sodium) has shown potential efficacy in neurological diseases (amyotrophic lateral sclerosis, cerebral ischemia) and cancers (non-small cell lung cancer, glioblastoma) due to its properties of inhibiting histone deacetylase (HDAC) and regulating endoplasmic reticulum stress[6]. In hepatocellular lipotoxicity, sodium 4-PBA alleviates palmitic acid-induced cell damage by inhibiting excessive endoplasmic reticulum stress, characterized by decreased expression of GRP78, CHOP and phosphorylated eIF2α [4].
|
| Molecular Formula |
C10H11O2.NA
|
|
|---|---|---|
| Molecular Weight |
186.18
|
|
| Exact Mass |
186.065
|
|
| Elemental Analysis |
C, 64.51; H, 5.96; Na, 12.35; O, 17.19
|
|
| CAS # |
1716-12-7
|
|
| Related CAS # |
1821-12-1 (free acid); 1716-12-7 (Sodium) 4-Phenylbutyric acid-d11;358730-86-6;4-Phenylbutyric acid-d5;64138-52-9;4-Phenylbutyric acid-d2;461391-24-2
|
|
| PubChem CID |
5258
|
|
| Appearance |
White to off-white solid powder
|
|
| Density |
1.095g/cm3
|
|
| Boiling Point |
290.7ºC at 760mmHg
|
|
| Melting Point |
207 °C (dec.)(lit.)
|
|
| Flash Point |
187.9ºC
|
|
| Vapour Pressure |
0.00288mmHg at 25°C
|
|
| LogP |
0.759
|
|
| Hydrogen Bond Donor Count |
0
|
|
| Hydrogen Bond Acceptor Count |
2
|
|
| Rotatable Bond Count |
4
|
|
| Heavy Atom Count |
13
|
|
| Complexity |
142
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
[Na+].[O-]C(C([H])([H])C([H])([H])C([H])([H])C1C([H])=C([H])C([H])=C([H])C=1[H])=O
|
|
| InChi Key |
VPZRWNZGLKXFOE-UHFFFAOYSA-M
|
|
| InChi Code |
InChI=1S/C10H12O2.Na/c11-10(12)8-4-7-9-5-2-1-3-6-9;/h1-3,5-6H,4,7-8H2,(H,11,12);/q;+1/p-1
|
|
| Chemical Name |
sodium;4-phenylbutanoate
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: 100 mg/mL (537.11 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication.
(Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.3711 mL | 26.8557 mL | 53.7115 mL | |
| 5 mM | 1.0742 mL | 5.3711 mL | 10.7423 mL | |
| 10 mM | 0.5371 mL | 2.6856 mL | 5.3711 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT06069375 | Recruiting | Drug: Sodium phenylbutyrate | Medium-chain Acyl-CoA Dehydrogenase Deficiency |
Jerry Vockley, MD, PhD | December 2023 | Phase 2 |
| NCT02111200 | Completed | Drug: Sodium Phenylbutyrate Drug: Sodium Benzoate |
Urea Cycle Disorders, Inborn | Baylor College of Medicine | September 2014 | Not Applicable |
| NCT01529060 | Completed | Drug: Phenylbutyrate Drug: Placebo powder |
Maple Syrup Urine Disease | Brendan Lee | February 2013 | Phase 2 Phase 3 |
| NCT00107770 | Completed | Drug: sodium phenylbutyrate | Amyotrophic Lateral Sclerosis | US Department of Veterans Affairs |
April 2005 | Phase 1 Phase 2 |
 |
|---|
 |
 |