
| Size | Price | Stock | Qty |
|---|---|---|---|
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g | |||
| 10g | |||
| Other Sizes |
Purity: ≥98%
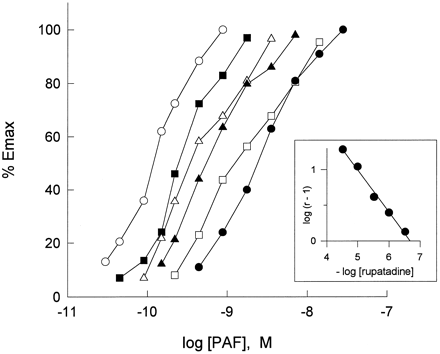
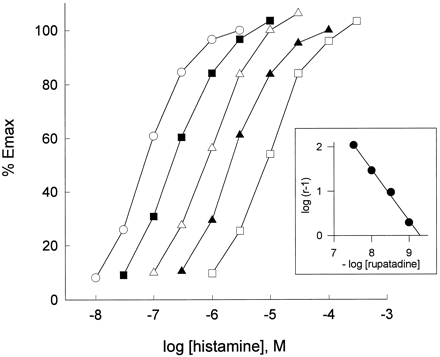
Rupatadine (also known as UR-12592) is a potent inhibitor of PAFR and histamine (H1) receptor with Ki of 550 nM and 102 nM, respectively. Rupatadine inhibits the effects of platelet-activating factor (PAF) and histamine (H1) by interacting with specific receptors. Rupatadine competitively suppresses histamine-induced guinea pig ileum contraction (pA2 = 9.29 ± 0.06) without affecting contraction induced by ACh, serotonin, or leukotriene D4 (LTD4). Additionally, it selectively prevents platelet aggregation caused by PAF in washed rabbit platelets (WRP) (pA2 = 6.68 ± 0.08) and in human platelet-rich plasma (HPRP) (IC50 = 0.68 μM), but not in platelet aggregation caused by arachidonic acid or ADP.
| Targets |
H1 Receptor ( Ki = 0.1 μM ); PAF ( Ki = 0.55 μM )
Histamine H1 Receptor (Ki = 0.4 nM) [1] - Platelet-Activating Factor (PAF) Receptor (IC50 = 0.8 μM for inhibiting PAF-induced platelet aggregation) [1] |
|---|---|
| ln Vitro |
In vitro activity: Rupatadine does not alter contractions induced by ACh, serotonin, or leukotriene D4 (LTD4), but it competitively inhibits histamine-induced guinea pig ileum contraction (pA2=9.29)[1].
Rupatadine does not affect platelet aggregation induced by arachidonic acid or ADP, but it competitively inhibits PAF-induced platelet aggregation in washed rabbit platelets (WRP) (pA2=6.68) and in human platelet-rich plasma (HPRP) (IC50=0.68 μM)[1]. Rupatadine has a concentration-dependent effect on TNF-α secretion, with maximum values of 92.5% when used at concentrations of 0.1-30 μM[2]. Rupatadine exhibited potent and selective binding to histamine H1 receptor: Ki = 0.4 nM, with no significant binding to H2, H3 receptors (Ki > 10 μM). It inhibited histamine-induced guinea pig ileum contraction with an EC50 of 0.3 nM [1] - It blocked PAF-induced biological effects: IC50 = 0.8 μM for inhibiting PAF-induced human platelet aggregation; 1 μM reduced PAF-induced calcium influx in human neutrophils by ~65% [1] - It inhibited mediator release from mast cells: 1-10 μM Rupatadine dose-dependently reduced histamine release from dispersed canine skin mast cells (inhibition rate ~38% at 1 μM, ~72% at 10 μM) and TNF-α release from human mast cell line HMC-1 (inhibition rate ~45% at 5 μM, ~68% at 10 μM) [2] - It attenuated PAF-mediated senescence in human lung fibroblasts: 5 μM Rupatadine reduced senescence-associated β-galactosidase (SA-β-gal) positive cells by ~55%, downregulated senescence markers (p16INK4a, p21CIP1) expression by ~48% and ~52%, and inhibited collagen I and α-SMA production by ~40% and ~45% [3] - No significant cytotoxicity to HMC-1 cells, human lung fibroblasts, or canine skin mast cells at concentrations up to 50 μM (cell viability > 90% by MTT assay) [2][3] |
| ln Vivo |
border-color: rgb(238, 238, 238); text-align: justify;" width="80%">Rupatadine inhibits the effects of histamine and PAF in vivo, including bronchoconstriction in guinea pigs (ID50=113 and 9.6 μg/kg i.v.) and hypotension in rats (ID50=1.4 and 0.44 mg/kg i.v., respectively)[1].
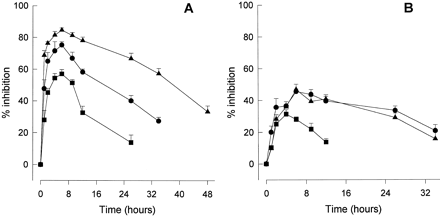
Rupatadine significantly reduces endotoxin-induced mortality in mice and rats (ID50=1.6 and 0.66 mg/kg i.v.) as well as PAF-induced mortality in mice (ID50=0.31 and 3.0 mg/kg i.v. and p.o., respectively)[1]. Rupatadine (6 mg/kg) reduces lung density and facilitates the absorption of lesions[3]. In histamine-induced cutaneous wheal and flare model in guinea pigs, oral Rupatadine (0.1 mg/kg, 0.3 mg/kg, 1 mg/kg) dose-dependently inhibited skin reactions. At 1 mg/kg, wheal area was reduced by ~82% and flare area by ~78% compared to vehicle group, with an ED50 of 0.25 mg/kg [1] - In PAF-induced hypotension model in rats, intravenous Rupatadine (0.3 mg/kg, 1 mg/kg) reversed PAF-mediated blood pressure drop: 1 mg/kg restored mean arterial pressure from 45 mmHg (PAF-induced) to 85 mmHg (normal level) within 15 minutes [1] - In bleomycin-induced pulmonary fibrosis model in mice, oral Rupatadine (10 mg/kg, 20 mg/kg, once daily for 21 days) protected against lung fibrosis: 20 mg/kg reduced lung hydroxyproline content by ~58%, collagen deposition area by ~62%, and inflammatory cell infiltration (neutrophils, macrophages) by ~55%. It also downregulated lung tissue p16INK4a, p21CIP1, TNF-α, and TGF-β1 mRNA levels by ~45-60% [3] |
| Enzyme Assay |
In 0.5 ml of 50 mM PBS, pH 7.5, antagonists are incubated for 30 minutes at 25 °C with guinea pig cerebellum membranes (0.6 mg/ml) and [3H]-pyrilamine (1.2 nM). The incubation is ended by the addition of 5 ml of ice-cold PBS containing 2 μM pyrilamine and the collection of membranes on Whatman GF/B filters. After being cleaned with 3 × 5 ml of ice-cold PBS and 2 μM pyrilamine, the filters are placed in counting vials. Using 3 ml of HiSafe 3, liquid scintillation counting is used to determine the amount of radioactivity retained by each filter. The determination of specific binding is based on the variation in the amount of [3H]-pyrilamine bound when unlabeled promethazine (10 μM) is present compared to when it is not.
Histamine H1 receptor binding assay: Recombinant human histamine H1 receptor was immobilized on a sensor chip. Rupatadine (0.001-10 μM) was injected at a constant flow rate, and surface plasmon resonance (SPR) signals were recorded to calculate Ki value. Competitive binding with radiolabeled histamine was also performed to confirm H1 receptor selectivity [1] - PAF receptor functional assay: Human platelets were incubated with Rupatadine (0.01-10 μM) for 30 minutes, then stimulated with PAF (100 nM). Platelet aggregation was measured by turbidimetry, and IC50 value was calculated [1] |
| Cell Assay |
C18-PAF is used to induce platelet aggregation, which is then measured with a dual-channel aggregometer Chrono-log 560. Platelet aggregation is measured both with and without the test compounds (5-min incubation). The inhibitors' activity is represented by their IC50 values. Rupatadine is tested in WRP against other aggregating agents, such as arachidonic acid (1 mM) and ADP (5 μM), in order to determine its selectivity. Rupatadine is present at different concentrations (3 × 10-7–3 × 10-5 M) and in the absence to obtain dose-response curves for PAF-induced aggregation in WRP.
Mast cell mediator release assay: Dispersed canine skin mast cells or HMC-1 cells were seeded in 24-well plates, pretreated with Rupatadine (1-10 μM) for 1 hour, then stimulated with compound 48/80 (mast cell activator) for 30 minutes. Culture supernatants were collected, and histamine levels were detected by fluorometric assay; TNF-α levels were measured by ELISA [2] - Fibroblast senescence assay: Human lung fibroblasts were seeded in 6-well plates, pretreated with Rupatadine (1-10 μM) for 2 hours, then stimulated with PAF (1 μM) for 72 hours. SA-β-gal staining was performed to count senescent cells; p16INK4a, p21CIP1, collagen I, and α-SMA expression was analyzed by Western blot and PCR [3] - Neutrophil calcium influx assay: Human neutrophils were loaded with fluorescent calcium probe, pretreated with Rupatadine (0.1-10 μM) for 30 minutes, then stimulated with PAF (100 nM). Intracellular calcium concentration ([Ca²⁺]i) was measured by flow cytometry [1] |
| Animal Protocol |
Dissolved in saline; 1 mg/kg; i.v. injection
PAF- and histamine-induced hypotension in normotensive rats Guinea pig cutaneous wheal model: Guinea pigs were randomized into vehicle and Rupatadine treatment groups (0.1 mg/kg, 0.3 mg/kg, 1 mg/kg). The drug was dissolved in 0.5% carboxymethylcellulose sodium and administered by oral gavage. After 1 hour, histamine was injected intradermally, and wheal/flare areas were measured at 30 minutes post-histamine injection [1] - Rat PAF-induced hypotension model: Rats were anesthetized, and mean arterial pressure was monitored via carotid artery catheter. Rupatadine was dissolved in normal saline and administered via intravenous injection (0.3 mg/kg, 1 mg/kg) 5 minutes after PAF (1 μg/kg) injection. Blood pressure was recorded for 30 minutes [1] - Mouse pulmonary fibrosis model: C57BL/6 mice were intratracheally injected with bleomycin (5 mg/kg) to induce pulmonary fibrosis. Rupatadine was dissolved in normal saline and administered by oral gavage at 10 mg/kg or 20 mg/kg once daily for 21 days (starting 1 day post-bleomycin). Mice were sacrificed, and lung tissues were collected for hydroxyproline assay, histological staining (HE, Masson), and PCR/Western blot analysis [3] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Rupatadine is rapidly absorbed, with a time to peak concentration (Tmax) of 1 hour. Concomitant administration with a high-fat meal increases drug exposure by 23% and prolongs Tmax to 2 hours. The apparent volume of distribution is 9799 L. Systemic clearance is 1556.2 L/h in young adults and 798.2 L/h in elderly patients. Metabolism/Metabolites Rupatadine is primarily metabolized via CYP3A4-mediated oxidative metabolism. CYP2C9, CYP2C19, and CYP2D6 are also involved, but to a lesser extent. The metabolite desloratadine and its hydroxylated form retain some H1 receptor antagonistic activity. Known metabolites of rupatadine include desloratadine. Biological half-life The elimination half-life is 15.9 hours for children aged 2-5 years, 12.3 hours for children aged 6-11 years, 5.9 hours for adults, and 8.7 hours for elderly patients. Oral bioavailability: approximately 80% in humans (10 mg orally), approximately 75% in rats [1] -Plasma elimination half-life (t1/2): approximately 24 hours in humans, approximately 12 hours in rats [1] -Peak plasma concentration (Cmax): 130 ng/mL 2 hours after administration of 10 mg orally in humans [1] -Volume of distribution (Vd): 10 L/kg in humans [1] -Plasma protein binding: approximately 98% in humans and rats [1] -Metabolized in the liver by cytochrome P450 enzymes, with approximately 70% of the total clearance excreted in the urine [1] |
| Toxicity/Toxicokinetics |
Protein Binding
Rupatadine binds to human plasma proteins at a rate of 98.5-99.0%. In vitro studies showed that rupatadine at concentrations up to 50 μM had no significant cytotoxicity to mammalian cells [2][3]. In vivo studies showed that oral administration of rupatadine at doses up to 200 mg/kg to rats for 28 consecutive days did not cause significant changes in body weight, organ index, or serum ALT/AST/creatinine levels [1]. The acute oral LD50 in mice was >2000 mg/kg [1]. Clinical adverse reactions were mild to moderate, including somnolence (6-8%), dry mouth (3-4%), and headache (2-3%) [1]. No significant drug interactions with cytochrome P450 substrates were observed [1]. |
| References |
|
| Additional Infomation |
Rupatadine is a benzocyclohepta-pyridine drug. Rupatadine is a dual-recombinant amine H1 receptor and platelet-activating factor (PAF) receptor antagonist used to relieve symptoms of seasonal and perennial allergic rhinitis and chronic spontaneous urticaria. It is approved in Canada under the brand name Rupall, available in adult tablets and pediatric liquid formulations. Indications: For the relief of nasal and non-nasal symptoms of seasonal and perennial allergic rhinitis in patients aged 2 years and older. Also for the relief of symptoms of chronic spontaneous urticaria in patients aged 2 years and older. FDA Label: Mechanism of Action: Rupatadine is a dual-recombinant amine H1 receptor and platelet-activating factor (PAF) receptor antagonist. During an allergic reaction, mast cells degranulate, releasing histamine and other substances. Histamine acts on H1 receptors, producing symptoms such as nasal congestion, runny nose, itching, and swelling. Platelet-activating factor (PAF) is produced by the cleavage of phospholipids by phospholipase A2. It promotes vascular leakage, thereby exacerbating runny nose and nasal congestion. Rupatadine reduces the severity of allergy symptoms by blocking H1 receptors and PAF receptors, preventing these mediators from functioning. Rupatadine is a synthetic, orally effective dual antagonist of histamine H1 receptors and platelet-activating factor (PAF) receptors [1][2][3] - Its core mechanism involves competitive binding to H1 and PAF receptors, inhibiting histamine/PAF-mediated signaling pathways, and inhibiting mast cell release of mediators [1][2] - It also exerts antifibrotic effects by attenuating PAF-mediated cellular senescence and reducing the production of profibrotic cytokines and collagen [3] - Approved indications include allergic rhinitis, chronic idiopathic urticaria, and other allergic diseases [1] - Oral (tablets or oral solution), the recommended daily dose for adults is 10 mg once daily. [1]
- It has good pharmacokinetic characteristics (long half-life, high bioavailability, high plasma protein binding rate) and good tolerability[1] |
| Molecular Formula |
C26H26CLN3
|
|
|---|---|---|
| Molecular Weight |
415.96
|
|
| Exact Mass |
415.181
|
|
| Elemental Analysis |
C, 75.08; H, 6.30; Cl, 8.52; N, 10.10
|
|
| CAS # |
158876-82-5
|
|
| Related CAS # |
Rupatadine Fumarate; 182349-12-8; Rupatadine-d4 fumarate; 1795153-63-7
|
|
| PubChem CID |
133017
|
|
| Appearance |
Solid powder
|
|
| Density |
1.2±0.1 g/cm3
|
|
| Boiling Point |
586.4±50.0 °C at 760 mmHg
|
|
| Melting Point |
58-61ºC
|
|
| Flash Point |
308.4±30.1 °C
|
|
| Vapour Pressure |
0.0±1.6 mmHg at 25°C
|
|
| Index of Refraction |
1.646
|
|
| LogP |
6.11
|
|
| Hydrogen Bond Donor Count |
0
|
|
| Hydrogen Bond Acceptor Count |
3
|
|
| Rotatable Bond Count |
2
|
|
| Heavy Atom Count |
30
|
|
| Complexity |
609
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
CC1=CN=CC(CN2CC/C(CC2)=C3C4=CC=C(Cl)C=C4CCC5=CC=CN=C5\3)=C1
|
|
| InChi Key |
WUZYKBABMWJHDL-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C26H26ClN3/c1-18-13-19(16-28-15-18)17-30-11-8-20(9-12-30)25-24-7-6-23(27)14-22(24)5-4-21-3-2-10-29-26(21)25/h2-3,6-7,10,13-16H,4-5,8-9,11-12,17H2,1H3
|
|
| Chemical Name |
13-chloro-2-[1-[(5-methylpyridin-3-yl)methyl]piperidin-4-ylidene]-4-azatricyclo[9.4.0.03,8]pentadeca-1(11),3(8),4,6,12,14-hexaene
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
|
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4041 mL | 12.0204 mL | 24.0408 mL | |
| 5 mM | 0.4808 mL | 2.4041 mL | 4.8082 mL | |
| 10 mM | 0.2404 mL | 1.2020 mL | 2.4041 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT03770923 | Recruiting | Drug: Rupatadine Drug: Montelukast |
Rheumatoid Arthritis | Sherief Abd-Elsalam | October 1, 2018 | Phase 3 |
| NCT01481909 | Completed | Drug: Rupatadin | Mastocytosis | Marcus Maurer | September 2010 | Phase 2 Phase 3 |
| NCT05356143 | Completed | Drug: Rupatadine | Healthy | J. Uriach and Company | December 2, 2021 | Phase 1 |
| NCT01605487 | Completed | Drug: Rupatadine | Cold Contact Urticaria | Charite University, Berlin, Germany |
June 2012 | Phase 2 |
| NCT01481909 | Completed | Drug: Rupatadin | Mastocytosis | Marcus Maurer | September 2010 | Phase 2 Phase 3 |
 |
|---|
 |
 |