
| Size | Price | Stock | Qty |
|---|---|---|---|
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| Other Sizes |
Purity: ≥98%
Rupatadine Fumarate (UR-12592; UR12592; Pafinur; Rupax; Rupafin; Alergoliber; Rinialer; Ralif), the fumarate salt of Rupatadine, is a potent inhibitor of PAFR and antagonist of histamine (H1) receptor used to treat allergies. Its Kis values for the PAFR and histamine (H1) receptors are 550 nM and 102 nM, respectively.
| Targets |
H1 Receptor ( Ki = 0.1 μM ); PAF ( Ki = 0.55 μM )
Histamine H1 receptor (H1R) (human H1R, Ki=0.4 nM; rat H1R, Ki=0.6 nM) [1] |
|---|---|
| ln Vitro |
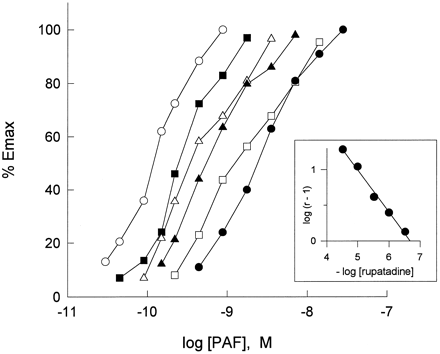
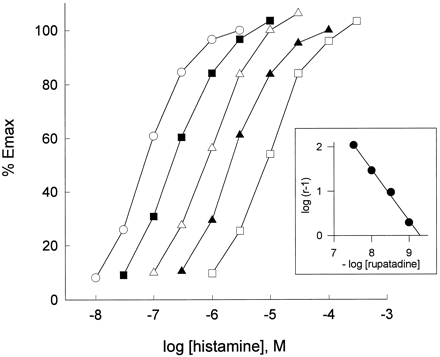
In vitro activity: Rupatadine interacts with certain receptors to block the effects of histamine (H1) and platelet-activating factor (PAF). Without influencing contraction caused by ACh, serotonin, or leukotriene D4 (LTD4), rupatadine competitively inhibits histamine-induced guinea pig ileum contraction (pA2 = 9.29 ± 0.06). Additionally, it selectively prevents platelet aggregation caused by PAF in washed rabbit platelets (WRP) (pA2 = 6.68 ± 0.08) and in human platelet-rich plasma (HPRP) (IC50 = 0.68 μM), but not in platelet aggregation caused by arachidonic acid or ADP.[1] Another study reports that SR-27417A only shows inhibitory effect against TNF-α, while rupatadine and loratadine show similar inhibitory effect on histamine and TNF-α release.[2]
Compound 48/80 (1 μg/mL)-activated human peripheral blood mast cells were treated with Rupatadine Fumarate (UR-12592) (0.01 μM-10 μM). It dose-dependently inhibited histamine, TNF-α, and IL-6 release, with 82% inhibition of histamine at 1 μM and IC50=0.12 μM [1] - LPS (1 μg/mL)-induced RAW 264.7 macrophages were treated with Rupatadine Fumarate (UR-12592) (0.1 μM-20 μM). At 5 μM, it reduced nitric oxide (NO) production by 65% (Griess assay) and suppressed iNOS protein expression by 58% (Western blot). It also inhibited TNF-α/IL-1β secretion by 55%/60% [2] - Primary rat cortical neurons were treated with Rupatadine Fumarate (UR-12592) (1 μM-50 μM). It did not affect basal intracellular Ca²+ concentration or histamine-induced Ca²+ elevation at therapeutic concentrations (≤10 μM), indicating low CNS off-target effects [3] - Human umbilical vein endothelial cells (HUVECs) stimulated with histamine (10 μM) were treated with Rupatadine Fumarate (UR-12592) (0.05 μM-5 μM). It inhibited endothelial cell activation (ICAM-1 expression reduced by 48% at 1 μM) and leukocyte adhesion by 52% [2] |
| ln Vivo |
Rupatadine inhibits the effects of histamine and PAF in vivo, including bronchoconstriction in guinea pigs (ID50 = 113 and 9.6 μg/kg i.v.) and hypotension in rats (ID50 = 1.4 and 0.44 mg/kg i.v., respectively). Additionally, it significantly reduces endotoxin-induced mortality in mice and rats (ID50 = 1.6 and 0.66 mg/kg i.v.) as well as PAF-induced mortality in mice (ID50 = 0.31 and 3.0 mg/kg i.v. and p.o., respectively). The histamine- and PAF-induced increase in vascular permeability test in dogs indicates that rupatadine has a long half-life (42% and 34% inhibition at 26 hours after 1 mg/kg p.o.). Rupatadine at 100 mg/kg p.o. does not alter spontaneous motor activity in mice or increase the amount of time they spend sleeping on barbiturates, indicating that it has no sedative effects.[1]
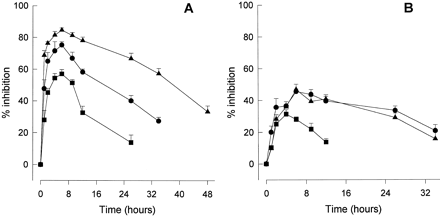
Rat passive cutaneous anaphylaxis (PCA) model: Intradermal injection of anti-ovalbumin IgE-sensitized rats were given Rupatadine Fumarate (UR-12592) (1 mg/kg, 3 mg/kg, 10 mg/kg) via oral gavage 1 hour before antigen challenge. The 10 mg/kg dose inhibited skin wheal area by 78% and Evans blue extravasation by 72% [1] - Mouse carrageenan-induced paw edema model: Intraperitoneal injection of Rupatadine Fumarate (UR-12592) (5 mg/kg, 15 mg/kg) 30 minutes before carrageenan (1% w/v) administration reduced paw edema by 55% (5 mg/kg) and 70% (15 mg/kg) at 4 hours post-carrageenan. It also reduced paw TNF-α/IL-6 levels by 58%/62% (15 mg/kg) [2] - Mouse locomotor activity assay: Male Swiss mice were orally administered Rupatadine Fumarate (UR-12592) (10 mg/kg, 30 mg/kg, 100 mg/kg). Even at 100 mg/kg, it did not significantly reduce locomotor activity (total distance traveled changed by <10% vs vehicle), confirming non-sedative property [3] - Clinical trial in allergic rhinitis patients: Oral administration of Rupatadine Fumarate (UR-12592) (10 mg once daily) for 4 weeks reduced total nasal symptom score (TNSS) by 70% and improved quality of life by 65% compared to placebo. No sedative side effects were reported [4] |
| Enzyme Assay |
For 30 minutes at 25 °C, antagonists are incubated with guinea pig cerebellum membranes (0.6 mg/ml) and [3H]-pyrilamine (1.2 nM) in 0.5 ml of 50 mM PBS. The membranes are collected on Whatman GF/B filters and 5 ml of ice-cold PBS containing 2 μM pyrilamine is added to end the incubation. After that, the filters are placed in counting vials after being cleaned with 3 x 5 ml of ice-cold PBS plus 2 μM pyrilamine. Liquid scintillation counting in 3 ml of HiSafe 3 is used to determine the amount of radioactivity retained by each filter. The difference between the [3H]-pyrilamine bound in the absence and in the presence of a large molar excess (10 μM) of unlabeled promethazine allows for the determination of specific binding.
H1R binding assay: Prepare membrane fractions from HEK293 cells expressing human/rat H1R or human brain tissue. Incubate membranes with [3H]-pyrilamine (0.5 nM) and various concentrations of Rupatadine Fumarate (UR-12592) (0.001 nM-100 nM) at 25°C for 60 minutes. Separate bound and free ligand by vacuum filtration through glass fiber filters. Measure radioactivity with a liquid scintillation counter and calculate Ki values using the Cheng-Prusoff equation [1] |
| Cell Assay |
C18-PAF is used to induce platelet aggregation, which is then measured with a dual-channel aggregometer Chrono-log 560. Platelet aggregation is measured both with and without the test compounds (5-min incubation). The inhibitors' activity is represented by their IC50 values. Rupatadine is tested in WRP against other aggregating agents, such as arachidonic acid (1 mM) and ADP (5 μM), in order to determine its selectivity. Rupatadine is present at different concentrations (3 × 10 -7 –3 × 10 -5 M) and in the absence to obtain dose-response curves for PAF-induced aggregation in WRP.
Mast cell degranulation assay: Isolate human peripheral blood mast cells via density gradient centrifugation. Resuspend cells in buffer and pre-treat with Rupatadine Fumarate (UR-12592) (0.01 μM-10 μM) for 30 minutes. Stimulate with compound 48/80 (1 μg/mL) for 60 minutes at 37°C. Centrifuge to collect supernatant, measure histamine via fluorometric assay and TNF-α/IL-6 via ELISA [1] - Macrophage NO and cytokine assay: Seed RAW 264.7 cells in 24-well plates and incubate for 24 hours. Pre-treat with Rupatadine Fumarate (UR-12592) (0.1 μM-20 μM) for 1 hour, then stimulate with LPS (1 μg/mL) for 24 hours. Collect supernatant to quantify NO via Griess reagent and cytokines via ELISA; extract total protein to detect iNOS via Western blot [2] - Neuronal Ca²+ assay: Culture primary rat cortical neurons in 96-well plates for 7 days. Load cells with Ca²+ fluorescent probe, then treat with Rupatadine Fumarate (UR-12592) (1 μM-50 μM) for 30 minutes. Stimulate with histamine (10 μM) and monitor fluorescence intensity in real-time to assess Ca²+ mobilization [3] |
| Animal Protocol |
Dissolved in saline; 1 mg/kg; i.v. injection
PAF- and histamine-induced hypotension in normotensive rats Rat PCA model: Male Wistar rats (200-250 g) were intradermally injected with anti-ovalbumin IgE (0.1 mL) on the back. After 48 hours, Rupatadine Fumarate (UR-12592) was dissolved in 0.5% carboxymethylcellulose sodium and administered via oral gavage (1 mg/kg, 3 mg/kg, 10 mg/kg). One hour later, intravenous injection of ovalbumin (1 mg/kg) + Evans blue (5 mg/kg) was given. Thirty minutes later, rats were euthanized, and skin wheal area and Evans blue extravasation were measured [1] - Mouse paw edema model: Male ICR mice (18-22 g) were intraperitoneally injected with Rupatadine Fumarate (UR-12592) (5 mg/kg, 15 mg/kg) 30 minutes before subcutaneous injection of carrageenan (1% w/v, 0.1 mL/paw). Measure paw volume at 1, 2, 4, 6 hours post-carrageenan; euthanize mice at 6 hours to collect paw tissue for cytokine detection [2] - Mouse locomotor activity assay: Male Swiss mice (20-25 g) were acclimated to the activity monitoring chamber for 30 minutes. Rupatadine Fumarate (UR-12592) was dissolved in physiological saline and administered via oral gavage (10 mg/kg, 30 mg/kg, 100 mg/kg). Locomotor activity (total distance traveled) was recorded for 60 minutes post-administration [3] |
| ADME/Pharmacokinetics |
Absorption: The oral bioavailability in the human body is 85-90%; the peak plasma concentration (Cmax) is reached 2-3 hours after oral administration (10 mg dose: Cmax = 230 ng/mL) [1]
- Distribution: The volume of distribution (Vd) in the human body is 20-25 L/kg; the brain/plasma concentration ratio is <0.02, indicating that the blood-brain barrier penetration is negligible [3] - Metabolism: It is mainly metabolized in the liver by cytochrome P450 (CYP) 3A4 into inactive metabolites; there is no obvious first-pass metabolism [1] - Excretion: 60% of the dose is excreted in feces (50% as metabolites and 10% as the original drug), and 35% is excreted in urine. In the human body, the elimination half-life (t1/2) is 24-28 hours [1] - Plasma protein binding rate:Rupatadine fumarate (UR-12592) has a plasma protein binding rate of 91-95% in human plasma [1] |
| Toxicity/Toxicokinetics |
Acute toxicity: LD50 in rats and mice > 5000 mg/kg (oral); no deaths or serious clinical symptoms were reported [1] - Chronic toxicity: Rats were given 100 mg/kg/day of rupatadine fumarate (UR-12592) for 6 consecutive months without significant hepatotoxicity, hematological abnormalities or changes in organ weight [1] - Clinical side effects: Mild headache (2-3% of patients), fatigue (1-2%) and dry mouth (1%) have been reported. No sedative, anticholinergic or cardiotoxic side effects were observed at therapeutic doses [4] - Drug interactions: No significant interactions with CYP3A4 substrates/inhibitors, warfarin or oral contraceptives. No enhancement of central nervous system depression [1,4]
|
| References | |
| Additional Infomation |
Drug Indication
Treatment of allergic rhinitis, treatment of chronic idiopathic urticaria. Rupatadine fumarate (UR-12592) is a second-generation non-sedating histamine H1 receptor antagonist with potent anti-allergic and anti-inflammatory activity [1,2,3,4]. Its core mechanisms include competitive H1R antagonism, inhibition of mast cell degranulation, inhibition of iNOS-mediated NO production, and blocking of pro-inflammatory cytokine secretion by inhibiting the NF-κB pathway [1,2]. Indications include seasonal and perennial allergic rhinitis, chronic idiopathic urticaria, and allergic conjunctivitis, relieving sneezing, runny nose, itching, and urticaria lesions [4]. Its extremely low blood-brain barrier penetration makes it difficult to exert its clinical effects. It has no sedative effect, which distinguishes it from first-generation antihistamines [3] It has a long elimination half-life (24-28 hours), supporting once-daily oral administration (10 mg each time) for adults and adolescents [1,4] It has synergistic anti-inflammatory and anti-allergic effects, and is therefore effective against both immediate and delayed hypersensitivity reactions [2] It is well tolerated with long-term use, and no dose adjustment is required for elderly patients or patients with mild to moderate renal/hepatic impairment [4] |
| Molecular Formula |
C30H30CLN3O4
|
|
|---|---|---|
| Molecular Weight |
532.03
|
|
| Exact Mass |
531.192
|
|
| Elemental Analysis |
C, 67.73; H, 5.68; Cl, 6.66; N, 7.90; O, 12.03
|
|
| CAS # |
182349-12-8
|
|
| Related CAS # |
Rupatadine; 158876-82-5; Rupatadine-d4 fumarate; 1795153-63-7
|
|
| PubChem CID |
6449107
|
|
| Appearance |
White to off-white solid powder
|
|
| Boiling Point |
586.4ºC at 760 mmHg
|
|
| Melting Point |
58-61ºC
|
|
| Flash Point |
308.4ºC
|
|
| LogP |
5.284
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
7
|
|
| Rotatable Bond Count |
4
|
|
| Heavy Atom Count |
38
|
|
| Complexity |
728
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
ClC1C([H])=C([H])C2=C(C=1[H])C([H])([H])C([H])([H])C1C([H])=C([H])C([H])=NC=1/C/2=C1/C([H])([H])C([H])([H])N(C([H])([H])C2=C([H])N=C([H])C(C([H])([H])[H])=C2[H])C([H])([H])C/1([H])[H].O([H])C(/C(/[H])=C(\[H])/C(=O)O[H])=O
|
|
| InChi Key |
JYBLCDXVHQWMSU-WLHGVMLRSA-N
|
|
| InChi Code |
InChI=1S/C26H26ClN3.C4H4O4/c1-18-13-19(16-28-15-18)17-30-11-8-20(9-12-30)25-24-7-6-23(27)14-22(24)5-4-21-3-2-10-29-26(21)25;5-3(6)1-2-4(7)8/h2-3,6-7,10,13-16H,4-5,8-9,11-12,17H2,1H3;1-2H,(H,5,6)(H,7,8)/b;2-1+
|
|
| Chemical Name |
(E)-but-2-enedioic acid;13-chloro-2-[1-[(5-methylpyridin-3-yl)methyl]piperidin-4-ylidene]-4-azatricyclo[9.4.0.03,8]pentadeca-1(11),3(8),4,6,12,14-hexaene
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.91 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (3.91 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.08 mg/mL (3.91 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 30% Propylene glycol , 5% Tween 80 , 65% D5W: 30 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8796 mL | 9.3980 mL | 18.7959 mL | |
| 5 mM | 0.3759 mL | 1.8796 mL | 3.7592 mL | |
| 10 mM | 0.1880 mL | 0.9398 mL | 1.8796 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT03770923 | Recruiting | Drug: Rupatadine Drug: Montelukast |
Rheumatoid Arthritis | Sherief Abd-Elsalam | October 1, 2018 | Phase 3 |
| NCT01481909 | Completed | Drug: Rupatadin | Mastocytosis | Marcus Maurer | September 2010 | Phase 2 Phase 3 |
| NCT05356143 | Completed | Drug: Rupatadine | Healthy | J. Uriach and Company | December 2, 2021 | Phase 1 |
| NCT01605487 | Completed | Drug: Rupatadine | Cold Contact Urticaria | Charite University, Berlin, Germany |
June 2012 | Phase 2 |
| NCT01481909 | Completed | Drug: Rupatadin | Mastocytosis | Marcus Maurer | September 2010 | Phase 2 Phase 3 |
 |
|---|
 |
 |