
| Size | Price | Stock | Qty |
|---|---|---|---|
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g | |||
| 5g | |||
| Other Sizes |
Purity: ≥98%
Prucalopride (R 108512; R108512; R 108512; trade names: Resolor; Resotran; Motegrity), a novel enterokinetic and benzofuran-based compound, is a novel, selective, high affinity 5-HT (serotonin) receptor agonist for 5-HT4A and 5-HT4B receptor with anti-idiopathic constipation activity. It shows >290-fold selectivity for 5-HT4A and 5-HT4B over other 5-HT receptor subtypes and inhibits 5-HT4A and 5-HT4B with Ki values of 2.5 nM and 8 nM, respectively. With a pEC50 of 7.5, prucalopride causes contractions in a concentration-dependent manner. Following electrical field stimulation, prucalopride dramatically increases the guinea pig proximal colon's rebound contraction. The FDA approved prucalopride in December 2018 for the treatment of idiopathic chronic constipation.
| Targets |
5-HT4A Receptor ( pKi = 8.6 ); 5-HT4B Receptor ( pKi = 8.1 )
Prucalopride Succinate (R-108512, R-93877) is a highly selective agonist of the 5-hydroxytryptamine 4 (5-HT4) receptor, with subtype selectivity: - Human recombinant 5-HT4a receptor: Ki = 1.3 nM (using [³H]-GR113808 as the radioligand) [1] - Human recombinant 5-HT4b receptor: EC50 = 3.8 nM (functional assay measuring cAMP accumulation, full agonism with 95% efficacy vs. 5-HT) [2] - No significant binding to other 5-HT receptor subtypes (5-HT1A/2A/3/7, Ki > 1000 nM) or neurotransmitter transporters (SERT, DAT, NET, Ki > 1000 nM) [1,2] |
|---|---|
| ln Vitro |
In vitro activity: Prucalopride has a pEC50 of 7.5 and causes contractions in a concentration-dependent manner. When electrical field stimulation is applied to the proximal colon of guinea pigs, the rebound contraction is greatly enhanced by pruralopride (1 mM). A monophasic concentration–response curve is produced when prucalopride relaxes the rat oesophagus preparation of rat oesophagus tunica muscularis mucosae, with a pEC50 of 7.8. [1] The amplitude of submaximal cholinergic contractions and acetylcholine release triggered by electrical field stimulation in pig stomach circular muscle is concentration-dependently increased by pracilocin (0.1 μM), and this effect is enhanced by IBMX (10 μM). [2] In pigs, descending colon electrically induced cholinergic contractions are significantly enhanced by prucalopride (1 μM), and rolipram significantly amplifies the facilitating effect. [3]
5-HT4 receptor-mediated cAMP accumulation: HEK293 cells expressing human 5-HT4b receptors were treated with Prucalopride Succinate (0.1–100 nM) for 30 minutes. At 10 nM, intracellular cAMP levels increased by 85% vs. baseline; EC50 = 3.8 nM (ELISA detection). Maximal efficacy was 95% of 5-HT (1 μM) [2] - Stimulation of colonic smooth muscle contraction: Isolated human colonic smooth muscle strips were incubated with Prucalopride Succinate (1–1000 nM) for 1 hour. At 100 nM, the contractile amplitude increased by 60% vs. vehicle; EC50 = 25 nM (force transducer measurement) [4] - No effect on neuronal BDNF expression: Primary rat cortical neurons treated with Prucalopride Succinate (up to 10 μM) for 72 hours showed no significant change in BDNF protein levels (Western blot) [2] |
| ln Vivo |
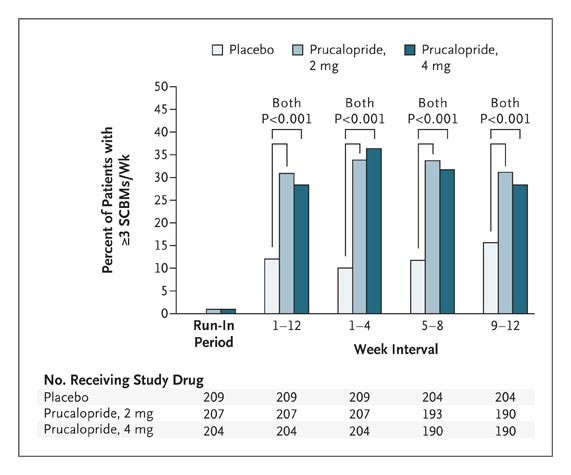
Prucalopride stimulates high-amplitude clustered contractions in the proximal colon and inhibits contractile activity in the distal colon of fasting dogs, modifying colonic contractile motility patterns in a dose-dependent manner. The first giant migrating contraction (GMC) is also dose-dependently shortened by prucalopride; at higher doses, the first GMC typically happens within the first half-hour following treatment.[4]
Enhancement of gastrointestinal motility in loperamide-induced constipated mice: Male ICR mice (20–25 g) were subcutaneously injected with loperamide (10 mg/kg) to induce constipation, then orally administered Prucalopride Succinate (0.1, 0.3, 1 mg/kg). At 0.3 mg/kg, the number of fecal pellets over 6 hours increased by 75% vs. vehicle; fecal water content increased from 32% to 48% [3] - Reduction of gastrointestinal transit time in rats: Male Sprague-Dawley rats (250–300 g) were orally given Prucalopride Succinate (0.05, 0.2, 0.5 mg/kg). At 0.2 mg/kg, the transit time of a charcoal meal through the gastrointestinal tract decreased by 40% (from 120 minutes to 72 minutes) vs. vehicle [1] - No central nervous system (CNS) effects: Mice treated with oral Prucalopride Succinate (up to 10 mg/kg) showed no changes in locomotor activity (open field test) or anxiety-like behavior (elevated plus maze) [2] |
| Enzyme Assay |
Human 5-HT4a Receptor Binding Assay: The 200 μL reaction system contained 50 μg of membrane protein from HEK293 cells expressing human 5-HT4a receptors, 0.5 nM [³H]-GR113808 (radioligand), and Prucalopride Succinate (0.01–100 nM). Incubated at 25°C for 60 minutes in 50 mM Tris-HCl (pH 7.4, 10 mM MgCl₂). Filtered through glass fiber filters pre-soaked in 0.3% polyethyleneimine, washed 3× with cold buffer. Radioactivity measured via liquid scintillation counter. Non-specific binding (NSB) determined with 10 μM unlabeled GR113808; Ki calculated via Cheng-Prusoff equation [1]
- 5-HT4b Receptor cAMP Functional Assay: HEK293-5-HT4b cells were seeded in 96-well plates (5×10⁴ cells/well) and cultured in DMEM + 10% FBS for 24 hours. Medium replaced with serum-free DMEM containing 0.5 mM IBMX (phosphodiesterase inhibitor), then Prucalopride Succinate (0.1–100 nM) added. Incubated at 37°C for 30 minutes, cells lysed with 0.1 M HCl. cAMP levels measured via competitive ELISA; EC50 derived from dose-response curves [2] |
| Cell Assay |
Isolated Human Colonic Smooth Muscle Cell Contraction Assay: Human colonic smooth muscle tissue was dissected into 2 mm×10 mm strips, suspended in Krebs-Ringer buffer (37°C, 95% O₂/5% CO₂). Strips equilibrated for 1 hour, then Prucalopride Succinate (1–1000 nM) added. Contractile force was recorded continuously for 1 hour using a force transducer. Data normalized to the maximal contraction induced by 1 μM carbachol [4]
- HEK293-5-HT4b Cell Viability Assay: HEK293 cells expressing 5-HT4b receptors were seeded in 96-well plates (1×10⁴ cells/well) and treated with Prucalopride Succinate (0.1–100 μM) for 72 hours. MTT (5 mg/mL) added, incubated for 4 hours, formazan crystals dissolved in DMSO. Absorbance at 570 nm measured; cell viability >95% vs. vehicle at all concentrations [2] |
| Animal Protocol |
In the prefrontal cortex, prucalopride maximally increased ACh and histamine levels at 5 and 10 mg/kg, while PRX-03140 significantly increased cortical histamine levels at 50 mg/kg, failing to affect ACh release at doses below 150 mg/kg, according to studies using microdialysis in rats.
Loperamide-Induced Constipation Mouse Model: Male ICR mice (6–8 weeks old, 20–25 g) housed at 22±2°C (12 h light/dark cycle). Randomized into 4 groups (n=10/group): 1. Vehicle: Oral gavage of 0.5% carboxymethylcellulose sodium (CMC-Na, 10 mL/kg); 2. Prucalopride 0.1 mg/kg: Oral gavage of Prucalopride Succinate (0.1 mg/kg, dissolved in 0.5% CMC-Na); 3. Prucalopride 0.3 mg/kg: Oral gavage of Prucalopride Succinate (0.3 mg/kg); 4. Prucalopride 1 mg/kg: Oral gavage of Prucalopride Succinate (1 mg/kg). Mice received loperamide (10 mg/kg, s.c.) 1 hour before drug administration. Fecal pellets were collected for 6 hours to count number and measure water content [3] - Rat Gastrointestinal Transit Model: Male Sprague-Dawley rats (8 weeks old, 250–300 g) fasted for 18 hours, randomized into 4 groups (n=6/group): 1. Vehicle: Oral gavage of 0.5% CMC-Na (10 mL/kg); 2. Prucalopride 0.05 mg/kg: Oral gavage of Prucalopride Succinate (0.05 mg/kg); 3. Prucalopride 0.2 mg/kg: Oral gavage of Prucalopride Succinate (0.2 mg/kg); 4. Prucalopride 0.5 mg/kg: Oral gavage of Prucalopride Succinate (0.5 mg/kg). Thirty minutes after drug administration, rats received a charcoal meal (10% charcoal in 5% gum arabic, 1 mL/100 g, p.o.). Rats euthanized 60 minutes later; gastrointestinal tract dissected. Transit time was calculated as the time for charcoal to reach the cecum [1] |
| ADME/Pharmacokinetics |
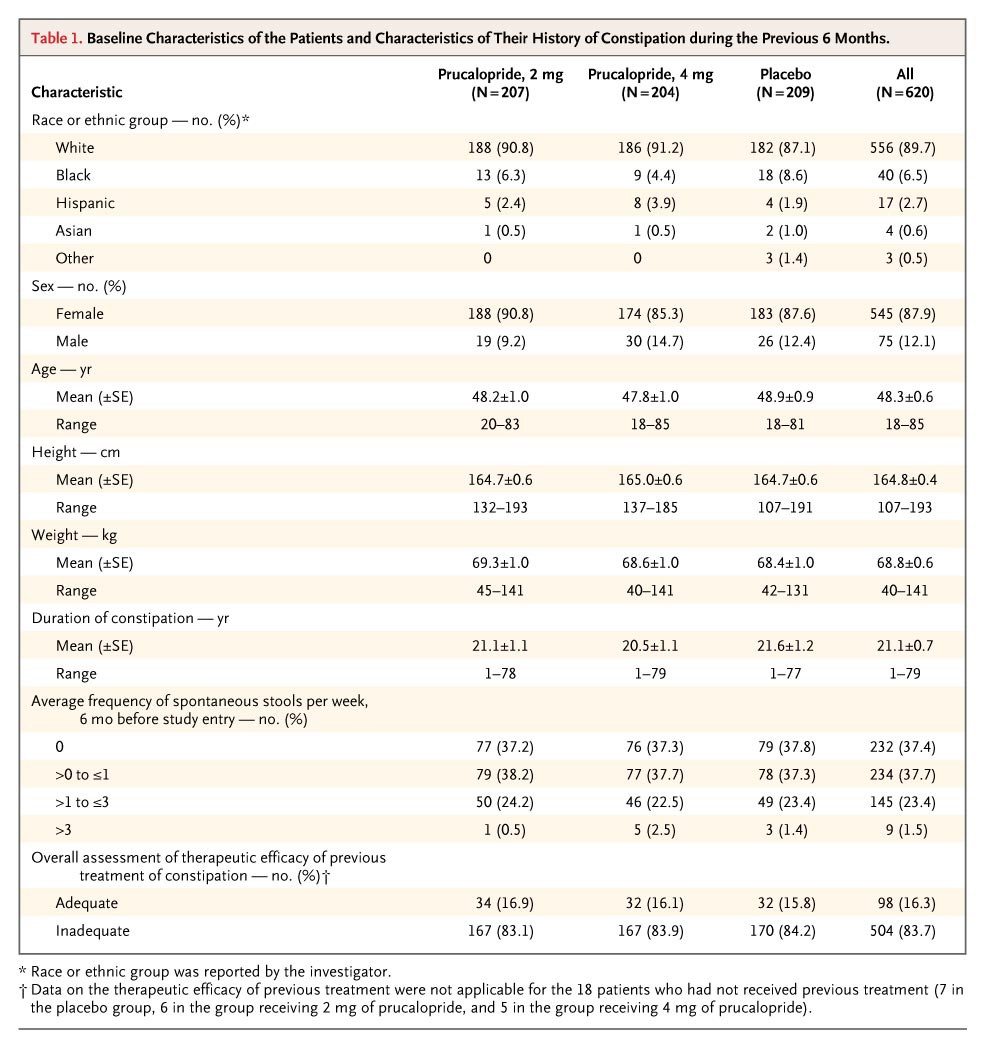
Oral bioavailability: In male Sprague-Dawley rats, the oral bioavailability of prucalopride succinate (1 mg/kg) was 82%, while that of intravenous administration (0.5 mg/kg) was 82% [1]
- Plasma pharmacokinetics: In rats receiving 0.5 mg/kg prucalopride succinate intravenously: Cmax = 0.8 μg/mL, Tmax = 5 min, elimination half-life (t1/2) = 3.5 h. Oral administration of 1 mg/kg: Cmax = 0.6 μg/mL, Tmax = 1.0 h, t1/2 = 4.2 h (HPLC-UV detection) [1] - Plasma protein binding rate: The protein binding rate of prucalopride succinate in human plasma was 30% (ultrafiltration method, plasma concentration range: 0.1–10 μg/mL) [1] - Tissue distribution: One hour after oral administration of 1 mg/kg prucalopride succinate to mice, the highest drug concentrations were found in the gastrointestinal tract (colon: 2.8 μg/g) and kidney (1.5 μg/g); the brain/plasma concentration ratio was 0.05 (low central nervous system penetration) [1] |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation
◉ Overview of Use During Lactation There is currently no published experience regarding the use of prucalopride during lactation. However, the manufacturer has reported an unpublished study showing relatively low levels of the drug in breast milk. Breastfed infants should be monitored for diarrhea until more data are available. ◉ Effects on Breastfed Infants As of the revision date, no relevant published information was found. ◉ Effects on Lactation and Breast Milk As of the revision date, no relevant published information was found. Acute In Vivo Toxicity: The LD50 of prucalopride succinate in male ICR mice (intraperitoneal injection) is 450 mg/kg. Mice given doses >300 mg/kg experienced transient diarrhea and lethargy, while no deaths were observed at doses ≤200 mg/kg [1] - Subacute toxicity: Rats were orally administered prucalopride succinate (0.1, 1, 10 mg/kg/day) for 28 days, with no significant changes in body weight (change <5%), serum ALT/AST/BUN/creatinine levels, or histopathological damage to the liver, kidneys, or gastrointestinal tract [1] |
| References | |
| Additional Infomation |
Prucalapride succinate is the succinate form of prucalapride, a highly bioavailable dihydrobenzofuran carboxamide compound and a selective serotonin (5-HT4) receptor agonist with gastrointestinal prokinetic effects. After oral administration, prucalapride specifically targets, binds to, and stimulates 5-HT4 receptors. This alters colonic motility patterns and stimulates overall colonic motility. This may help restore normal bowel regularity and relieve chronic constipation. Additionally, prucalapride can alleviate inhalation-related symptoms by enhancing esophageal and gastric motility.
See also: Prucalapride (containing the active ingredient). Drug Indications Resolor is indicated for the treatment of chronic constipation symptoms in adults, particularly in patients for whom laxatives do not adequately relieve symptoms. Treatment of chronic constipation, treatment of opioid-induced constipation Mechanism of action:Prucalopride succinate (R-108512, R-93877) exerts its prokinetic effect by selectively activating 5-HT4 receptors on gastrointestinal smooth muscle cells and enteric neurons. This activation increases intracellular cAMP levels, enhances smooth muscle contraction, and accelerates gastrointestinal motility [1,4] -Therapeutic potential:Prucalopride succinate has been clinically approved for the treatment of chronic idiopathic constipation (CIC), especially in patients who do not respond to laxatives. Preclinical data have confirmed that it can reverse drug-induced constipation without central nervous system side effects [3] -Chemical properties:Prucalopride succinate (R-108512, R-93877) is a white crystalline powder, soluble in water (25 mg/mL) and DMSO (50 mg/mL). It is stable for 7 days in aqueous solutions at pH 4.0–8.0 at room temperature [1] |
| Molecular Formula |
C22H32CLN3O7
|
|
|---|---|---|
| Molecular Weight |
485.96
|
|
| Exact Mass |
485.192
|
|
| Elemental Analysis |
C, 54.38; H, 6.64; Cl, 7.29; N, 8.65; O, 23.05
|
|
| CAS # |
179474-85-2
|
|
| Related CAS # |
Prucalopride; 179474-81-8
|
|
| PubChem CID |
9870009
|
|
| Appearance |
White to off-white solid powder
|
|
| LogP |
3.117
|
|
| Hydrogen Bond Donor Count |
4
|
|
| Hydrogen Bond Acceptor Count |
9
|
|
| Rotatable Bond Count |
9
|
|
| Heavy Atom Count |
33
|
|
| Complexity |
538
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
ClC1=C(C2C([H])([H])C([H])([H])OC=2C(=C1[H])C(N([H])C1([H])C([H])([H])C([H])([H])N(C([H])([H])C([H])([H])C([H])([H])OC([H])([H])[H])C([H])([H])C1([H])[H])=O)N([H])[H].O([H])C(C([H])([H])C([H])([H])C(=O)O[H])=O
|
|
| InChi Key |
QZRSNVSQLGRAID-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C18H26ClN3O3.C4H6O4/c1-24-9-2-6-22-7-3-12(4-8-22)21-18(23)14-11-15(19)16(20)13-5-10-25-17(13)14;5-3(6)1-2-4(7)8/h11-12H,2-10,20H2,1H3,(H,21,23);1-2H2,(H,5,6)(H,7,8)
|
|
| Chemical Name |
4-amino-5-chloro-N-[1-(3-methoxypropyl)piperidin-4-yl]-2,3-dihydro-1-benzofuran-7-carboxamide;butanedioic acid
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.14 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.14 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (5.14 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0578 mL | 10.2889 mL | 20.5778 mL | |
| 5 mM | 0.4116 mL | 2.0578 mL | 4.1156 mL | |
| 10 mM | 0.2058 mL | 1.0289 mL | 2.0578 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT02047045 | Completed | Procedure: acupuncture Drug: Prucalopride |
Constipation | Guang'anmen Hospital of China Academy of Chinese Medical Sciences |
April 2014 | Not Applicable |
| NCT01870674 | Completed | Drug: YH12852 Drug: Prucalopride Drug: Placebo |
Healthy | Yuhan Corporation | August 2013 | Phase 1 |
| NCT03279341 | Completed | Drug: Prucalopride Drug: Bisacodyl Drug: polyethylene glycol |
Chronic Constipation | University Hospital, Gasthuisberg |
December 3, 2012 | Phase 4 |
| NCT05966246 | Completed | Drug: Arm I : Experimental (Prucalopride succinate group) Drug: Arm II : Control (Mosapride citrate group) |
Gastric Cancer | Gangnam Severance Hospital | January 25, 2022 | Not Applicable |
| NCT01807000 | Completed | Drug: Radiolabeled Prucalopride Succinate |
Healthy | Shire | March 18, 2013 | Phase 1 |
 |
|---|
 |
 |