| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
Purity: =98.60%
| Targets |
Acetylcholinesterase
|
|---|---|
| ln Vitro |
Protopine (10–40 μM, 24–96 h) limits the ability of translocated cells (HepG2, Huh7) to migrate, proliferate, and undergo EMT [2]. Protopine (10–40 μM, 24 h) suppresses PI3K/Akt signaling dye and causes cell opening in HepG2 and Huh7 cells via expressing caspase-3 and caspase-9 [2]. Protopine (0–10 μg/mL) inhibits serotonin transporter (SERT) function in S6 cells and N1 cells, while protopine (10–40 μM, 6 hours) increases the formation of reactive oxygen species (ROS) in HepG2 and Huh7 cells [3].
|
| ln Vivo |
Protopine (0.1 and 1 mg/kg, intraperitoneal injection) can attenuate scopolamine (1 mg/kg)-induced memory impairment in mice [1]. Protropine (5-20 mg/kg, i.v.) inhibits tumor growth and inhibits PI3K/Akt, and induces caspase-3 channels in xenografted BALB/c mice (subcutaneously injected with HepG2 or Huh-7 cells) [ 2]. Pratopine (5-20 mg/kg, intraperitoneally) shows antidepressant-like effects in mouse HTR and TST tests [3]. Protopine (1-4 mg/kg, intraperitoneally, once daily for 3 days) has a protective effect against focal brain injury [4].
|
| Enzyme Assay |
Protopine is an isoquinoline alkaloid that possesses various biological activities including the anti-tumour activity. However, the effects of protopine on liver carcinoma cells are still elusive. The aim of this study is to examine the effects of protopine on liver carcinoma cells both in vitro and in vivo.
Methods: MTT assay was performed to measure the cell viability. Wound healing and transwell assays were conducted to assess the motility of cells. Cellular apoptosis and ROS levels were measured by the flow cytometry. Western blotting assay was used to measure the change of proteins. The cytotoxicity of protopine was also evaluated in xenograft mice.
Results: Protopine inhibited viabilities and triggered apoptosis via the intrinsic pathway in a caspase-dependent manner in liver carcinoma cells. Furthermore, protopine also induced accumulation of intracellular ROS which further led to the inhibition of PI3K/Akt signalling pathway. Finally, in vivo study showed that protopine also repressed tumour growth in xenograft mice without noticeable toxicity.
Conclusions: Protopine might be used as a potential therapeutic agent for the treatment of liver carcinoma[2].
|
| Cell Assay |
Western Blot Analysis[2]
Cell Types: HepG2, Huh7 Tested Concentrations: 10, 20, 40 μM Incubation Duration: 24 hrs (hours) Experimental Results: Induced caspase-3 and caspase-9 cleavage. Bcl-2 and Bcl-xl levels are diminished. Induces the release of the mitochondrial protein cytochrome c into the cytoplasm. |
| Animal Protocol |
Animal/Disease Models: 5-Hydroxy-DL-tryptophan (5-HTP)-induced mouse model [3]
Doses: 5, 10, 20 mg/kg Route of Administration: intraperitoneal (ip) injection Experimental Results: Increased 5-HTP-induced head Number of hemispheric twitch responses (HTR). Reduce the immobility time tested in the Tail Suspension Test (TST). The protopine isolated from a Chinese herb Dactylicapnos scandens Hutch was identified as an inhibitor of both serotonin transporter and noradrenaline transporter in vitro assays. 5-hydroxy-DL-tryptophan(5-HTP)-induced head twitch response (HTR) and tail suspension test were adopted to study whether protopine has anti-depression effect in mice using reference antidepressant fluoxetine and desipramine as positive controls. In HTR test, protopine at doses of 5, 10, 20 mg/kg dose dependently increase the number of 5-HTP-induced HTR. Protopine at doses of 3.75 mg/kg, 7.5 mg/kg and 30 mg/kg also produces a dose-dependent reduction in immobility in the tail suspension test. The present results open up new possibilities for the use of protopine in the treatment of mood disorders, such as mild and moderate states of depression.[3] Protopine, an isoquinoline alkaloidis, is known to produce many effects such as vasodilation, down-regulation of glutamate levels in brain and decrease of intracellular calcium. However, so far there is no report on the effect of protopine in cerebral ischaemia. In this study, the effect of protopine on the focal cerebral ischaemia was investigated in rats. Male Sprague-Dawley rats were divided into five groups: sham-operated group, vehicle-treated group and three doses of protopine-treated groups (0.98, 1.96 and 3.92 mg/kg). Protopine was intraperitoneally administered to rats once daily for 3 days prior to the ischaemia and 0.9% normal saline to rats in the vehicle-treated group in the same pattern. Rats in the sham-operated group were given 0.9% normal saline without the ischaemia. The focal cerebral ischaemia was induced by the middle cerebral artery occlusion for 24 hr via the intraluminal filament technique. The results showed that pre-treatment with protopine reduced the cerebral infarction ratio and serum lactate dehydrogenase activity, and improved the ischaemia-induced neurological deficit score and histological changes of brain in a dose-dependent manner. The further studies demonstrated that protopine increased superoxide dismutase activity in serum, and decreased total calcium and terminal deoxynucleotidyl transferase-mediated dUTP nick end labelling (TUNEL)-positive cells in the ischaemic brain tissue in the middle cerebral artery occlusion rats. The results indicate that protopine is able to produce an effective protection on the injury caused by the focal cerebral ischaemia in rats possibly through the multiple effects of calcium antagonism, antioxidation and depression of cell apoptosis.[4] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Xanfu Siwu Decoction (XFSWD) has been widely used clinically for the treatment of primary dysmenorrhea for hundreds of years with significant efficacy. One component of XFSWD, the eluent obtained by eluting a 60% ethanol-water extract through a macroporous adsorption resin (XFSWE), exhibits significant analgesic effects. This study aimed to investigate the pharmacokinetics and tissue distribution of four main active components (berberine, protopine, tetrahydroberberine, and tetrahydropalmatine) in dysmenorrhea-symptomatic rats after oral administration of XFSWE, and to compare the differences between normal rats and dysmenorrhea-symptomatic rats. A dysmenorrhea-symptomatic rat model was established using estradiol benzoate and oxytocin, with an experimental period of 7 days. On the last day of the experimental period, both normal and dysmenorrhea-symptomatic rats were orally administered XFSWE, and blood and tissue samples were collected at different time points. The contents of berberine, protopine, tetrahydroberberine, and tetrahydropalmatine in blood and tissue samples were determined by liquid chromatography-tandem mass spectrometry (LC-MS/MS). Pharmacokinetic parameters were calculated based on plasma concentration-time data using a non-compartmental model. One-way ANOVA was used to test the differences in pharmacokinetic parameters among groups. Results showed statistically significant differences in Cmax, Tmax, AUC(0-t), AUC(0-∞), MRT(0-t), MRT(0-∞), and CL/F between normal rats and dysmenorrhea rats orally administered the same dose of XFSWE (P<0.05). Tissue distribution analysis showed the following overall trend: spleen > liver > kidney > uterus > heart > lung > ovary > brain > thymus, with M-60 min > M-120 min > M-30 min > C-60 min > C-120 min > C-30 min. The levels of protoporphyrin in the liver, spleen, and uterus of rats exhibiting dysmenorrhea symptoms were higher than in other tissues. Compared with normal rats, the levels of compounds in different tissues at different time points were significantly different in rats with dysmenorrhea symptoms (P<0.05). This study reports for the first time the pharmacokinetics and tissue distribution of dysmenorrhea symptoms in animals. The results showed that berberine, protoporphyrin, tetrahydroberberine, and tetrahydropalmatine were absorbed at higher rates and eliminated more slowly in rats with dysmenorrhea syndrome, suggesting that the rate and extent of drug metabolism were altered in these rats. The results also showed significant differences in the concentrations of berberine, protoporphyrin, and tetrahydropalmatine in certain organs and at different time points between normal and dysmenorrhea-symptomatic rats, suggesting altered organ blood flow and perfusion rates in these animals. Metabolites California poppy preparations are used as herbal medicines and medicinal materials. This article describes the metabolic and toxicological analysis of the California poppy alkaloids papaverine and protoporphyrin in rat urine using gas chromatography-mass spectrometry. ...Protopine...first undergoes extensive 2,3-methylenedioxy demethylation, followed by catechol-O-methylation. All phenolic hydroxyl metabolites are partially bound. The authors established a systematic toxicological analysis method using acid hydrolysis, liquid-liquid extraction, and microwave-assisted acetylation, combined with full-scan gas chromatography-mass spectrometry. This method can detect the major metabolites of papaverine and Protopine in rat urine at doses comparable to those in drug users. Therefore, using the authors' systematic toxicological analysis method, the use of papaverine preparations should also be detectable in human urine. |
| Toxicity/Toxicokinetics |
Toxicity Summary
Identification and Uses: Protopine is a solid used as a drug. Human Exposure and Toxicity: Gene reporter assays using transiently transfected HepG2 cells confirmed that protopine-induced CYP1A1 expression was associated with slight or negligible activation of aryl hydrocarbon receptors. Protopine-induced CYP1A mRNA levels in HepG2 cells and human hepatocytes did not lead to increased CYP1A protein or activity levels. Animal Studies: In vitro radiolabeling studies showed that protopine can enhance the binding of γ-aminobutyric acid to rat brain synaptic membrane receptors. Protopine has antiarrhythmic effects and may directly inhibit rapid electrical activity in cardiomyocytes. Studies have found that protopine can inhibit histamine H1 receptors and platelet aggregation, and has analgesic effects. Protopine is a compound with opioid-like biological activity, acting on opioid receptors. Its properties include inducing analgesia or anesthesia. Protopeptides selectively bind to histamine H1 receptors without activating them, thus blocking the effects of endogenous histamine. Classic antihistamines primarily antagonize or prevent the effects of histamine in immediate-type hypersensitivity reactions. They act on the bronchi, capillaries, and other smooth muscles to prevent or alleviate motion sickness, seasonal rhinitis, and allergic dermatitis, as well as to induce drowsiness. Protopeptides can also act as platelet aggregation inhibitors, antagonizing or inhibiting any mechanism leading to platelet aggregation, whether during activation and morphological changes, or after dense granule release and prostaglandin-thromboxane system stimulation. Protopeptides can inhibit the contraction of isolated myocardial papillary muscles and endothelin-induced proliferation of vascular smooth muscle cells. They can also shorten the action potential duration and prolong the effective refractory period of guinea pig myocardial papillary muscles. The protective effect of protopeptides against myocardial ischemia-reperfusion injury in rats and their induced relaxation of the rat thoracic aorta are related to the inhibition of calcium ion influx into voltage-gated and receptor-gated calcium ion channels. Protopine has been a focus of numerous biological studies. Research indicates that it possesses antiparasitic activity and exhibits weaker cytotoxicity compared to other isoquinoline alkaloids. In vitro experiments have shown that protopine has a cytoprotective effect against oxidative stress-induced cell death. Animal model studies have demonstrated that this alkaloid possesses antiarrhythmic, antithrombotic, anti-inflammatory, and hepatoprotective effects. The biological activity of protopine may be related to its ability to inhibit calcium, sodium, and potassium channels. (PMID:15588728; PMID:21419197; L2104) Interactions The antiarrhythmic effects of protopine on experimental arrhythmias have been studied in various animal models. Protopine increased the aconitine dose required to induce ventricular tachycardia (VP), ventricular tachycardia (VT), and ventricular fibrillation (VF) in rats, and increased the dose of strophanthin (strophanthin K) required to induce ventricular tachycardia (VP) in guinea pigs. It also shortened the duration of aconitine-induced central arrhythmias and the duration of phenylephrine-induced arrhythmias in rats. It prevented the occurrence of arrhythmias induced by intravenous calcium chloride and inhalation of chloroform in rats and mice, respectively. In rabbits, the drug increased the incidence of ventricular fibrillation (VFT). These findings suggest that protopine has antiarrhythmic effects and may directly inhibit the rapid electrical activity of cardiomyocytes. Non-human toxicity values Guinea pig intraperitoneal injection LD50: 116 mg/kg Guinea pig oral LD50: 237 mg/kg Mouse intraperitoneal injection LD50: 482 mg/kg |
| References |
|
| Additional Infomation |
Protopine is a dibenzo-azine heptane alkaloid isolated from Codora (Fumaria vaillantii), and it is a plant metabolite. It has also been reported to exist in Codora (Corydalis solida), Codora trifoliata, and other organisms with relevant data. Protopine is a benzylisoquinoline alkaloid found in poppies and other Papaveraceae plants. It has been found to inhibit histamine H1 receptors and platelet aggregation, and possesses opioid analgesic effects. See also: root (part) of Sanguinaria canadensis; inflorescence (part) of Chelidonium majus.
Therapeutic Uses Analgesic, opioid; histamine H1 receptor antagonist; platelet aggregation inhibitor /EXPL THER/ ... This study investigated the anti-cancer cell proliferation and adhesion effects of five alkaloids isolated from Corydalis yanhusuo. We performed MTT assays, cell migration assays, cell invasion assays, and three cell adhesion assays on MDA-MB-231 human breast cancer cells. We further explored the mechanism by which these compounds inhibit heterologous cell adhesion by detecting the expression of epidermal growth factor receptor (EGFR), intercellular adhesion molecule-1 (ICAM-1), αv integrin, β1 integrin, and β5 integrin. Among the five tested alkaloids, only protopine showed anti-adhesion and anti-invasion effects in MDA-MB-231 cells, which contributes to the anti-metastatic effect of Corydalis yanhusuo. The results showed that after 90 minutes of protoporphyrin treatment, the expression of EGFR, ICAM-1, αv integrin, β1 integrin, and β5 integrin was significantly reduced. These results suggest that protoporphyrin appears to inhibit heterogeneous cell adhesion between MDA-MB-231 cells and human umbilical vein endothelial cells by altering the expression of adhesion factors. This study investigated whether protoporphyrin derived from Hypecoum erectum L. could inhibit lipopolysaccharide (LPS)-induced inflammatory responses in mouse macrophages (Raw 264.7 cells). The study found that protoporphyrin reduced the production of nitric oxide (NO), cyclooxygenase-2 (COX-2), and prostaglandin E2 (PGE2) in LPS-stimulated Raw 264.7 cells without cytotoxicity. Pretreatment of Raw 264.7 cells with protoporphyrin reduced the production of pro-inflammatory cytokines. This inhibitory effect is achieved by blocking the phosphorylation of mitogen-activated protein kinase (MAP kinase) and the activation of activated B cell nuclear factor κB (NF-κB). This study investigated the anticancer effects of protopine on human hormone-refractory prostate cancer (HRPC) cells. Protopine exerts its antiproliferative effect by inducing microtubule polymerization and mitotic arrest, ultimately leading to apoptosis. Data showed that protopine enhanced the activity of the cyclin-dependent kinase 1 (Cdk1)/cyclin B1 complex and promoted apoptosis by regulating mitochondrial-mediated signaling pathways, such as Bcl-2 phosphorylation and Mcl-1 downregulation. In summary, the data indicate that protopine is a novel microtubule stabilizer that exerts anticancer activity against HRPC cells via the apoptotic pathway by regulating Cdk1 activity and the Bcl-2 protein family. For more complete data on the therapeutic uses of protopine (12 items in total), please visit the HSDB record page. |
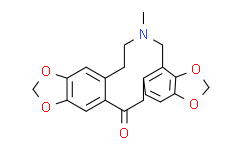
| Molecular Formula |
C20H19NO5
|
|---|---|
| Molecular Weight |
353.374
|
| Exact Mass |
353.126
|
| CAS # |
130-86-9
|
| Related CAS # |
Protopine hydrochloride;6164-47-2
|
| PubChem CID |
4970
|
| Appearance |
White to off-white solid powder
|
| Density |
1.3±0.1 g/cm3
|
| Boiling Point |
547.5±49.0 °C at 760 mmHg
|
| Melting Point |
211ºC
|
| Flash Point |
284.9±29.8 °C
|
| Vapour Pressure |
0.0±1.5 mmHg at 25°C
|
| Index of Refraction |
1.613
|
| LogP |
3.76
|
| Hydrogen Bond Donor Count |
0
|
| Hydrogen Bond Acceptor Count |
6
|
| Rotatable Bond Count |
0
|
| Heavy Atom Count |
26
|
| Complexity |
542
|
| Defined Atom Stereocenter Count |
0
|
| InChi Key |
GPTFURBXHJWNHR-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C20H19NO5/c1-21-5-4-13-7-18-19(25-10-24-18)8-14(13)16(22)6-12-2-3-17-20(15(12)9-21)26-11-23-17/h2-3,7-8H,4-6,9-11H2,1H3
|
| Chemical Name |
15-methyl-7,9,19,21-tetraoxa-15-azapentacyclo[15.7.0.04,12.06,10.018,22]tetracosa-1(17),4,6(10),11,18(22),23-hexaen-3-one
|
| Synonyms |
Fumarine; Biflorine; Corydinine; Fumarine; Biflorine; Macleyine; Protopin; Hypercorine; Protopine
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
DMSO : ~12.5 mg/mL (~35.37 mM)
|
|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.47 mg/mL (4.16 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 14.7 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.25 mg/mL (3.54 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 1.25 mg/mL (3.54 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8299 mL | 14.1495 mL | 28.2990 mL | |
| 5 mM | 0.5660 mL | 2.8299 mL | 5.6598 mL | |
| 10 mM | 0.2830 mL | 1.4149 mL | 2.8299 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
|
|
|