
| Size | Price | Stock | Qty |
|---|---|---|---|
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
Purity: ≥98%
Palonosetron HCl (formerly RS-25259, RS-25259, RS 25259 197, RS-25259197; trade names: Aloxi and Akynzeo), an approved antiemetic drug, is a potent 5-HT3 antagonist that has been used in the prevention and treatment of chemotherapy-induced nausea and vomiting. In order to avoid acute and delayed nausea and vomiting that are linked to both initial and repeated courses of highly emetogenic cancer chemotherapy, the FDA approved the use of fosnetupitant and palonosetron in April of 2018. With a ± hundred-fold higher 5-HT3 receptor binding affinity than other 5-HT3 receptor antagonists (pKi 10.5 compared with 8.91 for granisetron, 8.81 for tropisetron, 8.39 for ondansetron, and 7.6 for dolasetron), palonosetron is a highly potent, selective, second-generation antagonist of the 5-HT3 receptor pathway.
| Targets |
5-HT3 Receptor
|
|
|---|---|---|
| ln Vitro |
|
|
| ln Vivo |
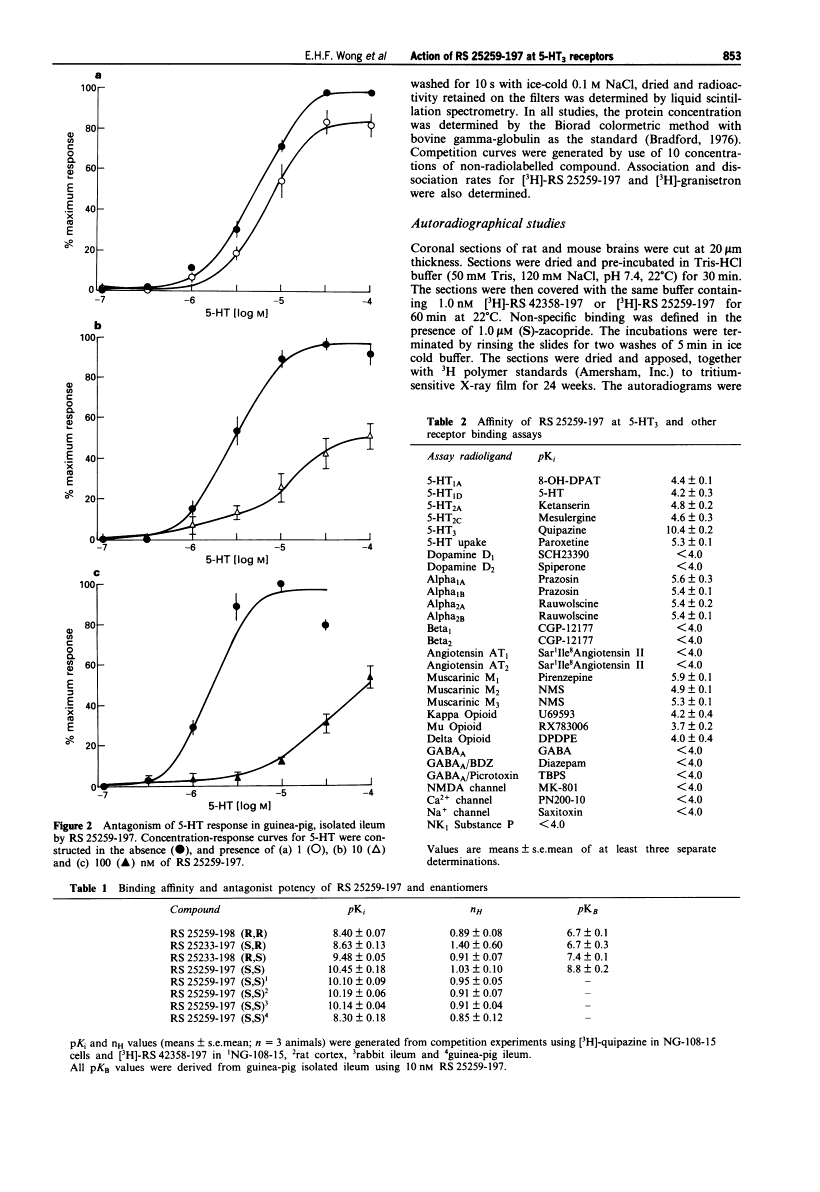
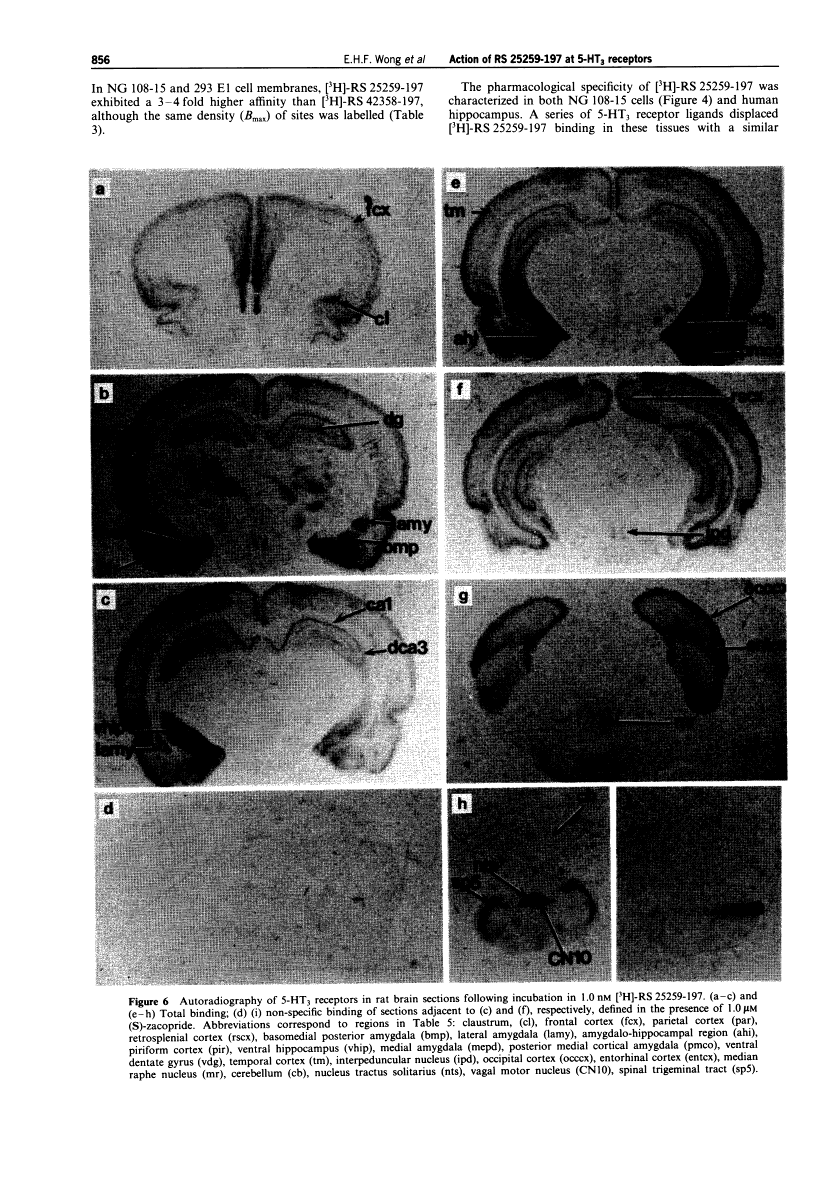
Quantitative autoradiographic studies in rat brain indicated a differential distribution of 5-HT3receptor sites by [3H]-RS 25259-197. High densities of sites were seen in nuclear tractus solitaris and area postrema, a medium density in spinal trigeminal tract, ventral dentate gyrus and basal medial amygdala,and a low density of sites in hippocampal CAl, parietal cortex, medium raphe and cerebellum.7 In conclusion, the functional, binding and distribution studies undertaken with the radiolabelled and non-radiolabelled RS 25259-197 (S,S enantiomer) established the profile of a highly potent and selective5-HT3 receptor antagonist[1].
|
|
| Enzyme Assay |
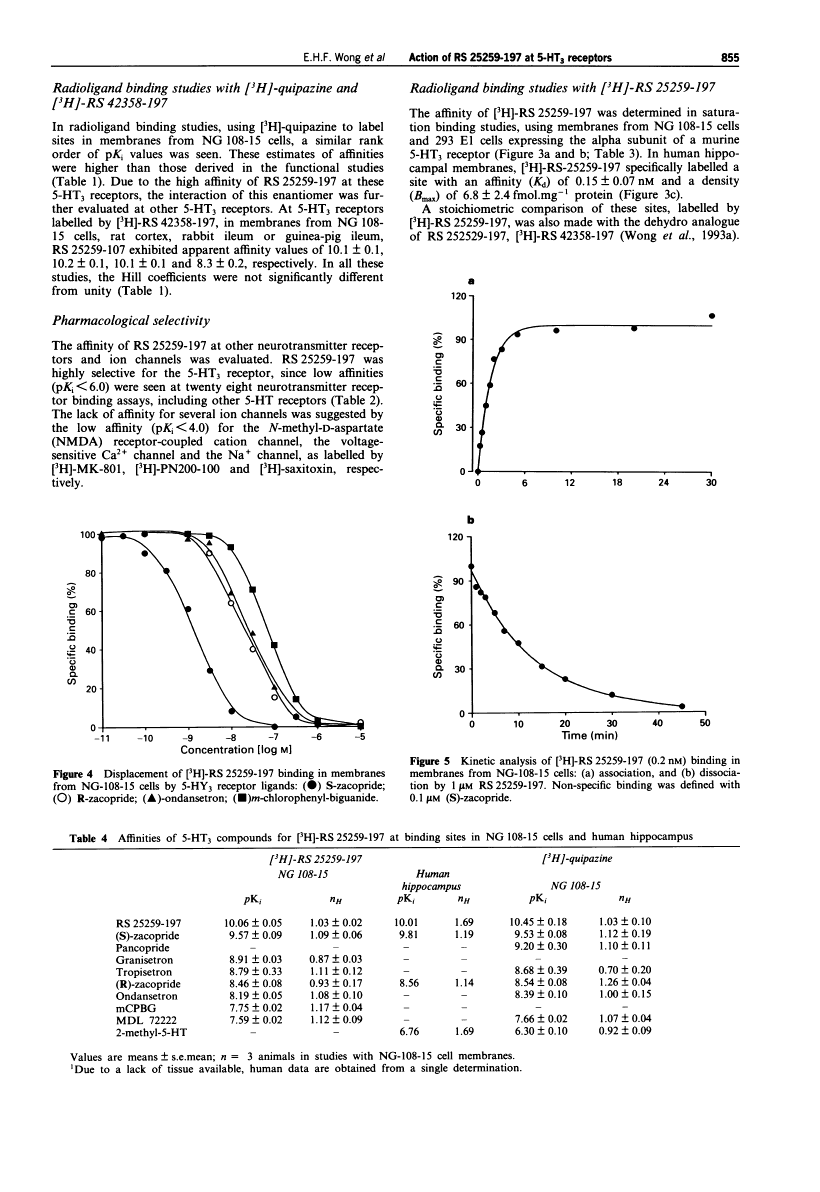
Palonosetron is a second-generation, highly selective, potent antagonist of the 5-HT3 receptor with a binding affinity for the receptor that is approximately 100 times higher than that of other antagonists of the 5-HT3 receptor (pKi 10.5 compared with 8.91 for granisetron, 8.81 for tropisetron, 8.39 for ondansetron, and 7.6 for dolasetron).
Radioligand binding assays at 5-HT3 receptors were conducted with four 5-HT3 receptor ligands, [3H]-quipazine, [3H]- granisetron, [3H]-RS 42348-197 and [3H]-RS 25259-197. Membranes were prepared according to the methods of Wong et al. (1993a) in a Tris-Krebs buffer (composition mM: NaCl 154, KCI 5.4, KH2PO4 1.2, CaCI2 2.5, MgCI2 1.0, Dglucose 11, Tris25, pH7.4 at 25°C) and incubations were performed in 0.5 ml total volume at 25°C for 60 min. Saturation studies were conducted with eight concentrations of radioligand, ranging from 4 pm to 4 nm. Competition studies were conducted with 0.1 to 0.4 nM of radioligand. Nonspecific binding was defined with 0.1 M (S)-zacopride. Reactions were terminated by vacuum filtration over GF/B filters pretreated with 0.3% polyethyleneimine. The filters were then washed for 10 s with ice-cold 0.1 M NaCI, dried and radioac- * tivity retained on the filters was determined by liquid scintillation spectrometry. In all studies, the protein concentration was determined by the Biorad colormetric method with bovine gamma-globulin as the standard (Bradford, 1976). Competition curves were generated by use of 10 concentrations of non-radiolabelled compound. Association and dissociation rates for [3H]-RS 25259-197 and [3H]-granisetron were also determined[1]. |
|
| Cell Assay |
Palonosetron is a 5-HT3 antagonist used to treat and prevent nausea and vomiting brought on by chemotherapy (CINV). IC50 Value: Among the 5-HT3 antagonists, 5-HT3 Receptor Palonosetron is the most successful in managing delayed CINV nausea and vomiting that manifests over a 24-hour period following the initial dosage of a chemotherapy regimen.
|
|
| Animal Protocol |
Autoradiographical studies[1]
Coronal sections of rat and mouse brains were cut at 20 ,um thickness. Sections were dried and pre-incubated in Tris-HCl buffer (50 mM Tris, 120 mM NaCl, pH 7.4, 22°C) for 30 min. The sections were then covered with the same buffer contain- -4 ing 1.0 nM [3H]-RS 42358-197 or [3H]-RS 25259-197 for 60 min at 22°C. Non-specific binding was defined in the presence of 1.0 tLM (S)-zacopride. The incubations were ter- -n minated by rinsing the slides for two washes of 5 min in ice cold buffer. The sections were dried and apposed, together with 3H polymer standards (Amersham, Inc.) to tritiumsensitive X-ray film for 24 weeks. The autoradiograms were then analysed by digital image analysis with the MCID imaging system (Imaging Research, Inc.). Brain areas were verified on cresyl violet stained sections after autoradiography, using the areas described in the rat brain atlas of Paxinos & Watson (1985). |
|
| References | ||
| Additional Infomation |
Palonosetron hydrochloride is a hydrochloride obtained by combining palonosetron with one molar equivalent of hydrogen chloride; an antiemetic used in combination with netupitant (under the trade name Akynzeo) to treat nausea and vomiting in patients undergoing cancer chemotherapy. It has a role as an antiemetic and a serotonergic antagonist. It contains a palonosetron(1+).
Palonosetron Hydrochloride is the hydrochloride salt of palonosetron, a carbazole derivative and a selective serotonin receptor antagonist with antiemetic activity. Palonosetron competitively blocks the action of serotonin at 5-hydroxytryptamine type 3 (5-HT3) receptors located on vagal afferents in the chemoreceptor trigger zone (CTZ), resulting in suppression of chemotherapy-induced nausea and vomiting. The CTZ is located in the area postrema on the dorsal surface of the medulla oblongata at the caudal end of the fourth ventricle and outside the blood-brain barrier (BBB). Isoquinoline and quinuclidine derivative that acts as a 5-HT3 RECEPTOR antagonist. It is used in the prevention of nausea and vomiting induced by cytotoxic chemotherapy, and for the prevention of post-operative nausea and vomiting. See also: Palonosetron (has active moiety); Fosnetupitant; palonosetron hydrochloride (component of); Netupitant; palonosetron hydrochloride (component of) ... View More ... Drug Indication Aloxi is indicated in adults for: the prevention of acute nausea and vomiting associated with highly emetogenic cancer chemotherapy,the prevention of nausea and vomiting associated with moderately emetogenic cancer chemotherapy. Aloxi is indicated in paediatric patients 1 month of age and older for: the prevention of acute nausea and vomiting associated with highly emetogenic cancer chemotherapy and prevention of nausea and vomiting associated with moderately emetogenic cancer chemotherapy. Palonosetron Hospira is indicated in adults for: the prevention of acute nausea and vomiting associated with highly emetogenic cancer chemotherapy; the prevention of nausea and vomiting associated with moderately emetogenic cancer chemotherapy. Palonosetron Hospira is indicated in paediatric patients 1 month of age and older for: the prevention of acute nausea and vomiting associated with highly emetogenic cancer chemotherapy and prevention of nausea and vomiting associated with moderately emetogenic cancer chemotherapy. |
| Molecular Formula |
C19H25CLN2O3
|
|---|---|
| Molecular Weight |
332.87
|
| Exact Mass |
332.165
|
| Elemental Analysis |
C, 68.56; H, 7.57; Cl, 10.65; N, 8.42; O, 4.81
|
| CAS # |
135729-62-3
|
| Related CAS # |
Palonosetron-d3 hydrochloride; 1246816-81-8; Palonosetron; 135729-61-2; (R,R)-Palonosetron Hydrochloride; 135729-75-8
|
| PubChem CID |
6918303
|
| Appearance |
White to off-white solid powder
|
| Boiling Point |
470.4ºC at 760 mmHg
|
| Melting Point |
>290ºC
|
| Flash Point |
209.5ºC
|
| Vapour Pressure |
5.07E-09mmHg at 25°C
|
| LogP |
3.334
|
| Hydrogen Bond Donor Count |
1
|
| Hydrogen Bond Acceptor Count |
2
|
| Rotatable Bond Count |
1
|
| Heavy Atom Count |
23
|
| Complexity |
456
|
| Defined Atom Stereocenter Count |
2
|
| SMILES |
O=C1N(C[C@@]([H])(CCC2)C3=C2C=CC=C13)[C@@H]4CN5CCC4CC5.[H]Cl
|
| InChi Key |
OLDRWYVIKMSFFB-SSPJITILSA-N
|
| InChi Code |
InChI=1S/C19H24N2O.ClH/c22-19-16-6-2-4-14-3-1-5-15(18(14)16)11-21(19)17-12-20-9-7-13(17)8-10-20;/h2,4,6,13,15,17H,1,3,5,7-12H2;1H/t15-,17-;/m1./s1
|
| Chemical Name |
(3aS)-2-[(3S)-1-azabicyclo[2.2.2]octan-3-yl]-3a,4,5,6-tetrahydro-3H-benzo[de]isoquinolin-1-one;hydrochloride
|
| Synonyms |
RS-25233-197; RS25233-198; RS 25259, RS 25259 197; Palonosetron hydrochloride; RS 25233-197; RS25233-197; RS-25233-198; RS 25233-198; RS-25259-197; US brand name: Aloxi; Akynzeo
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 0.33 mg/mL (0.99 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 3.3 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 0.33 mg/mL (0.99 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 3.3 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 0.33 mg/mL (0.99 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 100 mg/mL (300.42 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0042 mL | 15.0209 mL | 30.0418 mL | |
| 5 mM | 0.6008 mL | 3.0042 mL | 6.0084 mL | |
| 10 mM | 0.3004 mL | 1.5021 mL | 3.0042 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT05956899 | Recruiting | Drug: Palonosetron Drug: Ondansetron |
Idiopathic Scoliosis Postoperative Nausea and Vomiting |
University of Malaya | June 1, 2023 | Phase 4 |
| NCT04507711 | Recruiting | Drug: 0 ul of palonosetron Drug: 1 ul of palonosetron |
Blood Coagulation Disorder | Seoul National University Bundang Hospital |
September 16, 2020 | Not Applicable |
| NCT03817970 | Recruiting | Drug: Granisetron Drug: Palonosetron |
Nephrotoxicity | University of Colorado, Denver | November 15, 2019 | Phase 3 |
| NCT05199818 | Recruiting | Drug: Palonosetron HCl Buccal Film 0.5 mg Drug: IV Palonosetron 0.25 mg |
Chemotherapy-induced Nausea and Vomiting |
Xiamen LP Pharmaceutical Co., Ltd |
March 1, 2022 | Phase 3 |
| NCT05841849 | Not yet recruiting | Drug: Aprepitant Drug: Palonosetron |
Breast Cancer Chemotherapy-induced Nausea and Vomiting |
Second Affiliated Hospital, School of Medicine, Zhejiang University |
July 2023 | Phase 4 |
 |
|---|
 |
 |