
| Size | Price | Stock | Qty |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g | |||
| Other Sizes |
Purity: ≥98%
Fluvoxamine Maleate (formerly DU 23000; MK264; MK 264; DU-23000; Faverin, Fevarin, Floxyfral, Luvox), the maleate salt of Fluvoxamine which is an approved antidepressant drug, is a potent and selective serotonin (5-HT) reuptake inhibitor (SSRI) used in the treatment of a variety of diseases including obsessive-compulsive disorder, major depressive disorder (MDD), and anxiety disorders such as panic disorder and post-traumatic stress disorder (PTSD).
| Targets |
SSRIs/selective serotonin reuptake inhibitors
|
||
|---|---|---|---|
| ln Vitro |
In vitro activity: Fluvoxamine elevates [DA]ex levels in the striatum and raises [5-HT]ex levels in the rat prefrontal cortex and thalamus.[1] Through its action on 5-HT neurons or spinal 5-HT2A/2C receptors, fluvoxamine maleate reduces tactile allodynia.[2]
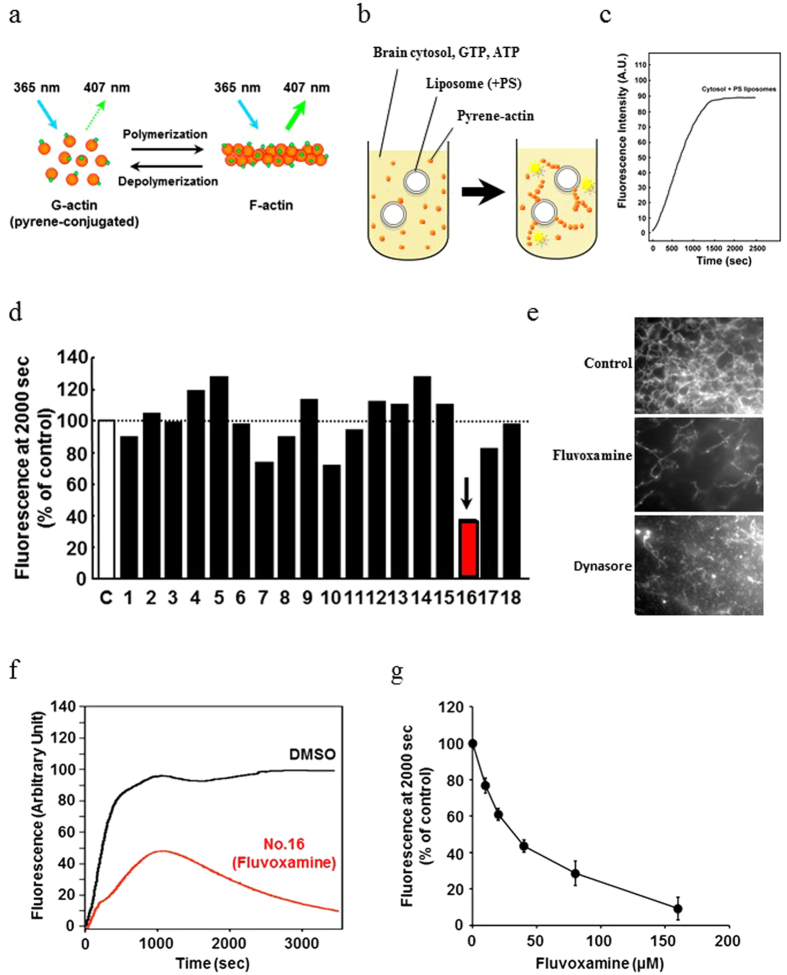
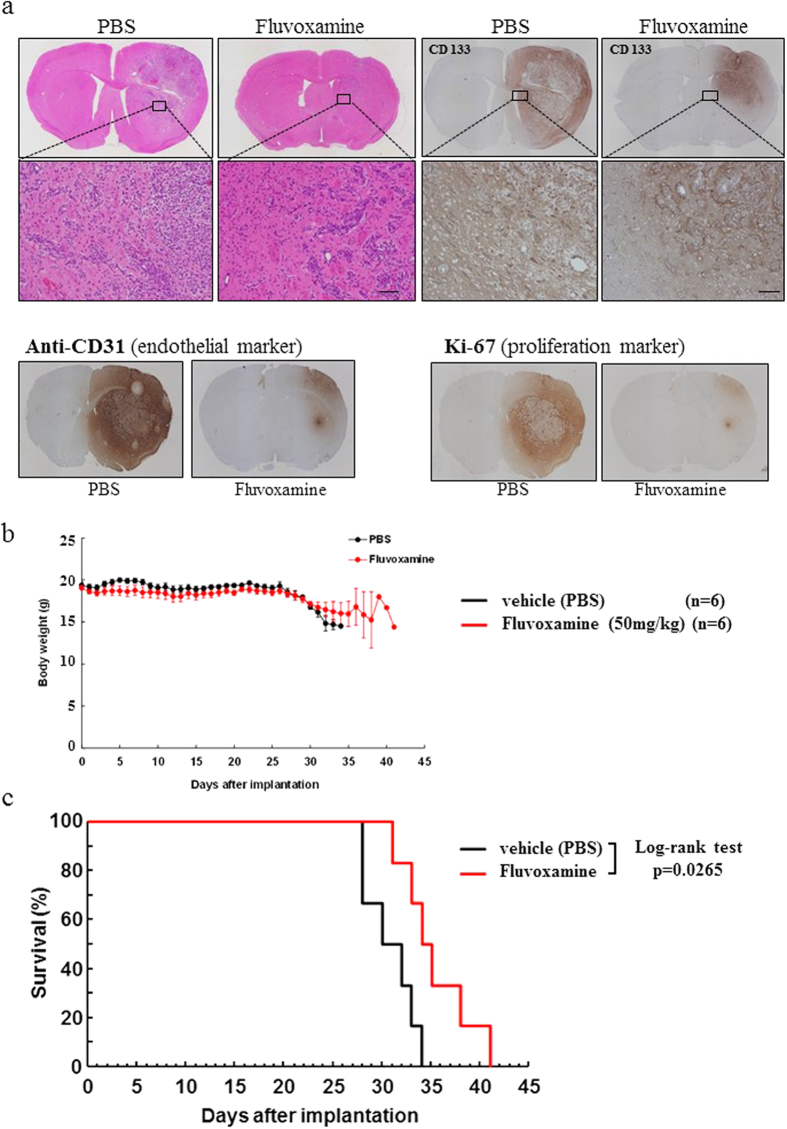
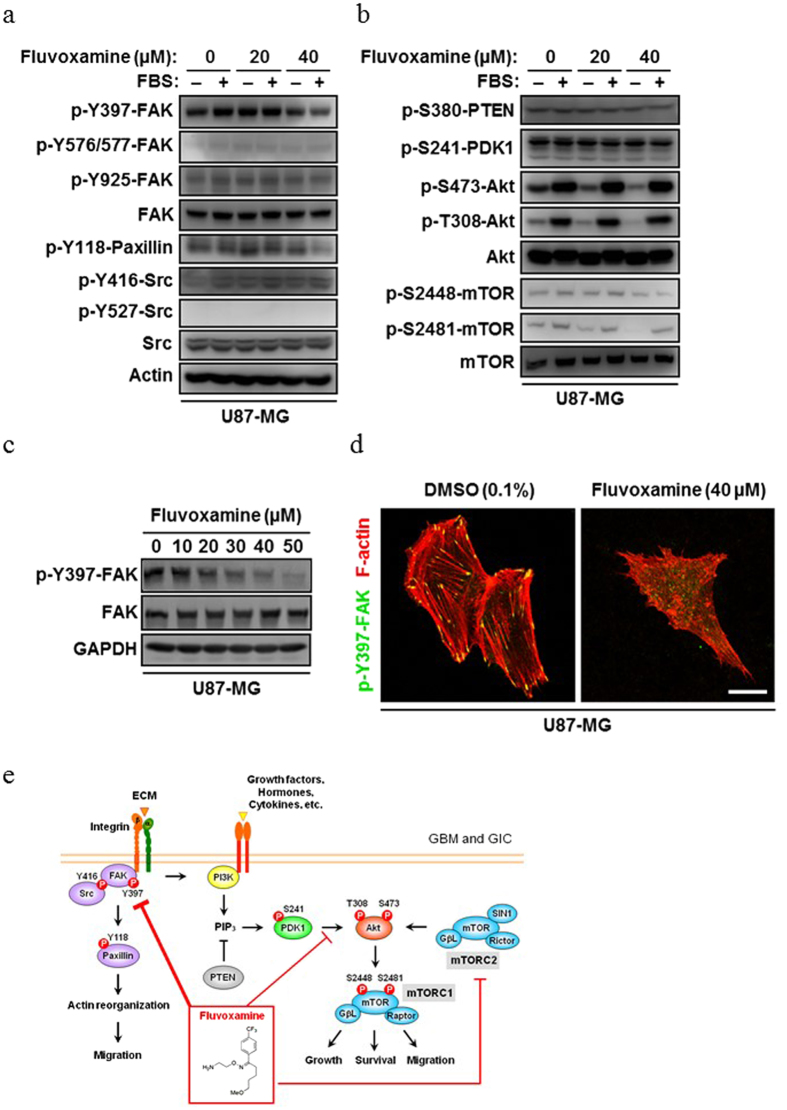
Flv/Fluvoxamine toxicity on SK-N-SH cells [4] The toxicity of Flv on SK-N-SH cells was examined using an MTS assay. We used 10, 25, 50, 75, or 100 μg/ml Flv or a vehicle control to treat SK-N-SH cells. SK-N-SH cells treated with Flv showed 80% (25 μg/ml), 29% (50 μg/ml), 19% (75 μg/ml), and 18% (100 μg/ml) viability compared to the vehicle control cells ([p<0.001] at all doses) (Fig. 1). However, SK-N-SH cells treated with 10 μg/ml Flv did not show reduced viability (102%) compared to the vehicle control (Fig. 1). Based on these data, we used 10 μg/ml Flv in all subsequent experiments. Flv/Fluvoxamine alleviates Px-induced ER stress mediated apoptosis [4] Next we investigated whether Flv could alleviate Px-induced ER stress-mediated apoptosis in SK-N-SH cells by monitoring CHOP, cleaved caspase 4, and cleaved caspase 3, an active form of each caspase. CHOP, cleaved caspase 4, and cleaved caspase 3 were induced in cells treated with Px compared to control cells (Fig. 2a–c, [p<0.01] at each comparison), which is consistent with our previous report [19], On the other hand, when cells were pre-treated with Flv followed by co-treatment with Px/Flv for 24 h, the induction of CHOP, cleaved caspase 4 and cleaved caspase 3 were alleviated compared to the Flv-untreated cells (Fig. 2a–c, p<0.05, p<0.05, and p<0.01, respectively). We next investigated the induction of Sig-1R by Flv in SK-N-SH cells. Flv has been reported not only as a potent Sig-1R agonist with stronger affinity than other SSRIs [27], but also as an inducer of Sig-1R [26]. Sig-1R was induced in cells treated with Flv for 12 h compared to untreated cells (Fig. 2d, p<0.05). This induction continued for at least 24 h (Fig. 2e, p<0.01). Flv/Fluvoxamine alleviates Px-induced neurotoxicity through Sig-1R [4] Finally, using a MTS assay, we quantitatively assessed whether Flv can alleviate Px-induced neurotoxicity. Similar to the results from Western blots, the viability that was decreased by Px treatment was recovered in Flv-pre-treated cells compared to Flv-untreated cells (Fig. 3, p<0.05). This recovery was reversed when cells were incubated with Px, Flv and NE100 (Fig. 3, p<0.05). We recently reported that Fluvoxamine (Flv) alleviates ER stress via induction of sigma-1 receptor (Sig-1R). The purpose of this study was to investigate whether Flv could alleviate Px-induced neurotoxicity in vitro. SK-N-SH cells were pre-treated for 12 h with or without 10 μg/ml Flv followed by treatment with 1 μM Px with or without co-existence of 10 μg/ml Flv for 24 h. To investigate the involvement of Sig-1R in alleviation effect on Px-induced neurotoxicity,1 μM NE100, an antagonist of Sig-1R, was added for 24 h. Neurotoxicity was assessed using the MTS viability assay and ER stress-mediated neurotoxicity was assessed by evaluating the expression of C/EBP homologous protein (CHOP), cleaved caspase 4, and cleaved caspase 3. Pre-treatment with Flv significantly alleviated the induction of CHOP, cleaved caspase 4, and cleaved caspase 3 in SK-N-SH cells. At the same time, pre-treatment with Flv significantly induced Sig-1R in SK-N-SH cells. In addition, viability was significantly higher in Flv-treated cells than in untreated cells, which was reversed by treatment with NE100. Our results suggest that Flv alleviates Px-induced neurotoxicity in part through the induction of Sig-1R. Our findings should contribute to one of the novel approaches for the alleviation of Px-induced neurotoxicity, including chemobrain. |
||
| ln Vivo |
Fluvoxamine maleate likewise demonstrates dose-dependent antinociception in the paw pressure test in non-ligated mice. In the acute paw pressure test, fluoxetine maleate also produces an antinociceptive effect that is countered by granisetron, an antagonist of the 5-HT3 receptor.[2] Fluvoxamine (10 and 30 mg/kg, i.p.) dose-dependently increases synaptic efficacy in the hippocampo-mPFC pathway in the rat hippocampo-medial prefrontal cortex (mPFC).[3] In rats under anesthesia, fluvoxamine (10 and 30 mg/kg, i.p.) inhibits long-term potentiation (LTP) in the hippocampal CA1 field. While the 5-HT(4) receptor antagonist GR 113808 (20 mg/rat, i.c.v.) and the 5-HT(7) receptor antagonist DR 4004 (10 mg/rat, i.c.v.) do not completely reverse the suppression of LTP induced by fluvoxamine (30 mg/kg, i.p.), they do.[4] An isolated rat vas deferens cultured in Krebs-Henseleit solution responds to norepinephrine more strongly when fluvoxamine maleate is added. With an IC50 of 18.2μM and 3.99μM, respectively, fluoxetine hydrochloride and fluoxetine maleate inhibit the contraction brought on by potassium ions in the isolated rat uterus preparation.
Quetiapine had no significant effect on [DA]ex and [5-HT]ex levels in the prefrontal cortex and thalamus, but increased [DA]ex and [5-HT]ex levels in the dorsal striatum. In the accumbens, quetiapine increased [DA]ex levels and decreased [5-HT]ex levels. Fluvoxamine increased [5-HT]ex levels in all brain areas, and also increased [DA]ex levels in the striatum. The combination of quetiapine with fluvoxamine increased [DA]ex and [5-HT]ex levels in all brain areas compared with baseline. Although neither quetiapine nor fluvoxamine in monotherapy affected [DA]ex levels in the prefrontal cortex and thalamus, the combination produced a significant increase of [DA]ex levels in these two brain areas.[1] There is an association between depression and chronic pain, and some antidepressants exert antinociceptive effects in humans and laboratory animals. We examined the effects of Fluvoxamine, a selective serotonin reuptake inhibitor, on mechanical allodynia and its mechanism of action in the mouse chronic pain model, which was prepared by partially ligating the sciatic nerve. The antiallodynic effect was measured using the von Frey test. Fluvoxamine produced antiallodynic effects following both systemic and intrathecal administration. In 5-hydroxytryptamine (5-HT)-depleted mice, prepared by intracerebroventricular injection of 5,7-dihyroxytryptamine, the fluvoxamine-induced antiallodynic effect was significantly attenuated. The antiallodynic effects of systemic fluvoxamine were also reduced by both systemic and intrathecal administration of ketanserin, a 5-HT2A/2C receptor antagonist. In addition, fluvoxamine also induced antinociceptive effect in the acute paw pressure test, and this effect was antagonized by the 5-HT3 receptor antagonist granisetron. These results indicate that fluvoxamine exerts its antiallodynic effects on neuropathic pain via descending 5-HT fibers and spinal 5-HT2A or 5-HT2C receptors, and the antinociception on acute mechanical pain via 5-HT3 receptors. [2] The present studies were conducted to examine the effects of single and repeated treatments with Fluvoxamine, which is a selective serotonin reuptake inhibitor (SSRI), on the synaptic efficacy and synaptic plasticity in the rat hippocampo-medial prefrontal cortex (mPFC) pathway in vivo. It has been reported that the projections arising from the hippocampal structures to the mPFC are involved in the execution of higher cognitive functions in rats. The evoked potentials were recorded in the mPFC by stimulation of the CA1/subicular region of the ventral hippocampus in halothane-anesthetized rats. Single administration of fluvoxamine (10 and 30 mg/kg, i.p.) enhanced synaptic efficacy in the hippocampo-mPFC pathway in a dose-dependent manner. Although repeated treatments with fluvoxamine (30 mg/kg, i.p. after 30 mg/kg/day×21 days, p.o.) caused an enhancement of synaptic efficacy, there was no significant difference between single and repeated treatments. The input/output characteristics showed hypersensitivity to stimulation intensity in the group with repeated fluvoxamine treatments. The establishment of long-term potentiation (LTP) in the hippocampo-mPFC pathway after a single administration of fluvoxamine was not different from that in the saline-injected group. On the other hand, the hippocampo-mPFC LTP was significantly augmented by repeated treatments with fluvoxamine when compared to a single treatment. These findings suggest that the serotonergic system could modulate the synaptic plasticity at hippocampal-mPFC synapses. The present study, furthermore, suggests that the enhancement of LTP in the hippocampo-mPFC pathway produced by repeated treatments with fluvoxamine may be implicated in the SSRI-induced therapeutic effect on psychiatric disorders[3]. |
||
| Cell Assay |
MTS cell viability assays[4]
Cellular viability was assessed using CellTiter 96 Aqueous One Solution Cell Proliferation Assays. Briefly, SK-N-SH cells were seeded in 96-well plates. Cells were allowed to attach for 24 h. For evaluation of the toxicity of Flv on SK-N-SH cells, cells were treated with 10, 25, 50, 75, or 100 μg/ml Flv for 24 h at 37 °C. For evaluation of the alleviation effect of Flv on Px-induced neurotoxicity, SK-N-SH cells were pre-treated with or without 10 μg/ml Flv for 12 h followed by 1 μM Px treatment with or without 10 μg/ml Flv for 24 h. To confirm the involvement of Sig-1 R in alleviation effect on Px- induced neurotoxicity, SK-N-SH cells were incubated with 1 μM Px, 10 μg/ml Flv and 1 μM NE100 for 24 h. Next, 20 μl of MTS reagent was added to each well and cells were incubated for 2 h. Optical density was measured at 490 nm using a Micro Plate Reader. Western blots[4] SK-N-SH cells were pre-treated with or without 10 μg/ml Flv for 12 h followed by 1 μM Px treatment with or without 10 μg/ml Flv for 24 h at 37 °C. Cells were washed in Tris-buffered saline (TBS), harvested, and lysed in RIPA buffer with a protease inhibitor cocktail (Roche, Mannheim, Germany), and a phosphatase inhibitor cocktail. Lysates were sonicated on ice three times for five seconds each, and then incubated for 15 min. After centrifugation for 20 min at 13,000 g, supernatants were retained and boiled in SDS sample buffer. Lysates (10 μg) were separated on SDS-polyacrylamide gels and transferred to polyvinylidene fluoride (PVDF) membranes. Non-specific protein binding was blocked by incubating membranes for 1 h at room temperature in 5% w/v non-fat milk powder in TBS-T [50 mM Tris–HCl (pH 7.6), 150 mM NaCl, and 0.1% v/v Tween-20]. The membranes were incubated overnight at 4 °C with the following primary antibodies: anti-CHOP (1:1000), anti-caspase 4 (1:500), anti-caspase 3 (1:1000), anti-sigma 1 receptor (Sig-1R) (1:250) and anti-GAPDH (1:1000). The membranes were then washed three times in TBS-T for 5 min. Finally, the membranes were incubated for 60 min at room temperature with HRP-conjugated anti-rabbit or anti-mouse antibodies. Protein bands were detected using the ECL Plus kit. The intensity of each band was quantified using NIH image J software. |
||
| Animal Protocol |
|
||
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Fluvoxamine maleate is well absorbed, with a bioavailability of 53%. Following administration of 5 mg of radiolabeled fluvoxamine maleate, nine metabolites were identified, accounting for approximately 85% of fluvoxamine excretion in urine. The major human metabolite is fluvoxamine acid, which, along with its N-acetylated analogues, accounts for approximately 60% of urinary excretion. Approximately 2% of fluvoxamine is excreted unchanged in urine. Following oral administration of 5 mg of 14C-labeled fluvoxamine maleate, an average of 94% of drug-related products are recovered from urine within 71 hours. 25 L/kg.hr Metabolism/Metabolites Fluvoxamine is primarily metabolized in the liver. Known human metabolites of fluvoxamine include fluvoxamine amino alcohol. Hepatic Excretion Route: The main human metabolite is fluvoxamine acid, which and its N-acetylated analogs account for approximately 60% of urinary excretion. Approximately 2% of fluvoxamine is excreted unchanged in the urine. Following oral administration of 14C-labeled fluvoxamine maleate (5 mg), an average of 94% of drug-related products were recovered in the urine within 71 hours. Half-life: 15.6 hours Biological half-life 15.6 hours. |
||
| Toxicity/Toxicokinetics |
Toxicity Summary
The exact mechanism of action of fluvoxamine is not fully understood, but it appears to be related to its inhibition of serotonin uptake by neurons in the central nervous system. Fluvoxamine blocks the reuptake of serotonin by the serotonin reuptake pump on the neuronal membrane, thereby enhancing the effect of serotonin on its 5-hydroxytryptamine 1A autoreceptor. In vitro studies have shown that fluvoxamine, as a serotonin reuptake inhibitor, is more potent than clomipramine, fluoxetine, and desipramine. Studies have also shown that fluvoxamine has almost no affinity for α1- or α2-adrenergic receptors, β-adrenergic receptors, muscarinic receptors, dopamine D2 receptors, histamine H1 receptors, GABA-benzodiazepine receptors, opioid receptors, 5-HT1 receptors, or 5-HT2 receptors. Hepatotoxicity Up to 1% of patients taking fluvoxamine have been reported to experience abnormal liver function, but the elevation is usually small and generally does not require dose adjustment or discontinuation. A small number of patients taking fluvoxamine have reported acute, clinically significant liver injury with markedly elevated liver enzymes, but without or with only mild jaundice. The injury typically occurs within days of starting treatment, with serum enzyme elevations presenting as hepatocellular or mixed. Autoimmune (autoantibodies) and immune hypersensitivity features (rash, fever, eosinophilia) are not mentioned. The number of reported cases is too small to provide a detailed description of the clinical features of liver injury. Fluvoxamine is rarely mentioned in large-scale analyses of adverse liver events associated with antidepressants and selective serotonin reuptake inhibitors (SSRIs). Effects during pregnancy and lactation ◉ Overview of use during lactation Limited information suggests that daily administration of fluvoxamine up to 300 mg results in low concentrations in breast milk and is not expected to have any adverse effects on breastfed infants, especially those older than 2 months. If a mother needs to take fluvoxamine, this is not a reason to discontinue breastfeeding. A safety rating system considers fluvoxamine safe for use during lactation. One case reported elevated serum fluvoxamine levels in an infant, but fluvoxamine levels were undetectable in the serum of most infants tested. Another infant experienced diarrhea, vomiting, and agitation after their mother began taking fluvoxamine. Limited long-term growth and development follow-up studies found no adverse reactions in breastfed infants. Infants exposed to fluvoxamine through breast milk should be monitored for symptoms such as diarrhea, vomiting, decreased sleep, and restlessness. Mothers taking selective serotonin reuptake inhibitors (SSRIs) during pregnancy and postpartum may find breastfeeding more difficult, but this may reflect their disease state. These mothers may require additional breastfeeding support. Breastfed infants exposed to SSRIs in late pregnancy have a lower risk of neonatal maladaptive disorder than formula-fed infants. ◉ Effects on Breastfed Infants One mother started taking 100 mg of fluvoxamine daily at 17 weeks postpartum, and the infant was breastfed from birth until 5 months of age. During the 10-week observation period while the mother was hospitalized, no adverse reactions were observed in the infant. The infant's Bayley Developmental Schedules scores were normal at 4 months and 21 months of age. Two other infants also showed no adverse reactions: a 26-month-old infant whose mother took 150 mg of fluvoxamine daily and was partially breastfed, also had a normal Denver Developmental Schedules score; another 3-week-old infant whose mother took 50 mg of fluvoxamine daily and was exclusively breastfed. The three mothers took an average of 117 mg of fluvoxamine daily, exclusively breastfed their infants for 4 months, and breastfed at least 50% of their babies in the 5th and 6th months. These infants' weight gain at 6 months met national growth standards, and the mothers did not report any adverse reactions in their infants. A study on the side effects of selective serotonin reuptake inhibitors (SSRIs) in breastfeeding mothers found that an infant born to a mother taking fluvoxamine did not experience any adverse reactions requiring medical attention. Specific information regarding maternal fluvoxamine dosage, breastfeeding extent, or infant age was not reported. A woman who had been taking quetiapine 400 mg and fluvoxamine 200 mg long-term throughout her pregnancy and postpartum took these medications. She partially breastfed her infant for 3 months from birth (expansion unspecified). The infant developed normally without any adverse events. A cohort study of 247 infants exposed to antidepressants in utero during late pregnancy assessed neonatal maladaptive disorder (PNA). Of these 247 infants, 154 developed PNA. The risk of PNA was approximately three times higher in exclusively formula-fed infants than in exclusively or partially breastfed infants. Four infants were exposed to low doses of fluvoxamine in utero but did not develop postpartum neuropathic pain. A 5-month-old infant developed severe diarrhea (15 times daily), mild vomiting (2 to 3 times daily), irritability, and reduced sleep two days after her mother began taking fluvoxamine (50 mg daily). The mother's symptoms disappeared 24 hours after stopping the medication; a week later, the symptoms recurred when the mother took fluvoxamine again. No other causes of gastrointestinal symptoms were found. Fluvoxamine is likely the cause of the reaction. The authors speculate that the infant may have a drug metabolism disorder leading to excessively high serum drug concentrations. A retrospective cohort study included 5079 newborns whose mothers had taken selective serotonin reuptake inhibitors (SSRIs) during pregnancy. The results showed that 1.5% of breastfed newborns experienced neonatal withdrawal symptoms, compared to 2.3% of formula-fed newborns, but the difference was not statistically significant. Breastfed newborns had a lower risk of being transferred to the neonatal intensive care unit (NICU) than formula-fed newborns; however, sensitivity analysis did not confirm this result. Only one woman in this study took paroxetine. Effects on lactation and breast milk: Fluvoxamine can cause elevated prolactin levels and galactorrhea in non-pregnant, non-lactating patients. In one case, a 19-year-old male experienced normal prolactinemia-induced gynecomastia and galactorrhea while taking risperidone. A study of cases of hyperprolactinemia and its symptoms (such as gynecomastia) reported by the French National Center for Pharmacovigilance found that fluvoxamine increased the risk of hyperprolactinemia by 4.5 times compared to other medications. Prolactin levels in established lactating mothers may not affect their ability to breastfeed. In a small prospective study, researchers compared eight primiparous women taking serotonin reuptake inhibitors (SRIs; three took fluoxetine, and the remaining one took citalopram, duloxetine, escitalopram, paroxetine, or sertraline, respectively) with 423 mothers not taking SRIs. Mothers taking selective serotonin reuptake inhibitors (SSRIs) experienced a mean delay of 16.7 hours initiation of lactation activation (stage II) compared to the control group (85.8 hours postpartum in the SSRI treatment group vs. 69.1 hours postpartum in the untreated group), which doubled the risk of delayed feeding behavior in the untreated group. However, the delay in stage II lactation may not be clinically significant, as there was no statistically significant difference between the two groups in the proportion of mothers experiencing feeding difficulties after day 4 postpartum. A case-control study compared breastfeeding rates at 2 weeks postpartum in mothers who took SSRIs throughout pregnancy and delivery (n = 167), mothers who took SSRIs only during pregnancy (n = 117), and control mothers who did not take antidepressants (n = 182). In two groups of participants who took selective serotonin reuptake inhibitors (SSRIs), 33 took citalopram, 18 took escitalopram, 63 took fluoxetine, 2 took fluvoxamine, 78 took paroxetine, and 87 took sertraline. Among women taking SSRIs, the breastfeeding rate at two weeks postpartum was 27% to 33% lower than that of mothers not taking antidepressants, but there was no statistically significant difference in breastfeeding rates between the SSRI exposure groups. An observational study investigated the outcomes of 2,859 women who had taken antidepressants in the two years prior to pregnancy. Compared to women who did not take antidepressants during pregnancy, mothers who took antidepressants in all three stages of pregnancy were 37% less likely to breastfeed at discharge. Mothers who took antidepressants only in the third trimester were 75% less likely to breastfeed at discharge. Pregnant women who took antidepressants only in the first and second trimesters were not less likely to breastfeed at discharge. However, the study did not specify the type of antidepressant used by the mothers. A retrospective cohort study analyzed hospital electronic medical records from 2001 to 2008, comparing women taking antidepressants in late pregnancy (n = 575; of whom n = 18 took fluvoxamine), women with mental illness but not taking antidepressants (n = 1552), and mothers not diagnosed with mental illness (n = 30,535). Women receiving antidepressant treatment were 37% less likely to breastfeed at discharge than women not diagnosed with mental illness, but there was no difference in the likelihood of breastfeeding compared to untreated mothers diagnosed with mental illness. A Norwegian study of 80,882 mother-infant pairs from 1999 to 2008 showed that 392 women reported starting antidepressants postpartum, and 201 women reported starting antidepressants during pregnancy. Compared to a control group not exposed to antidepressants, taking antidepressants in late pregnancy was associated with a 7% lower likelihood of initiating breastfeeding, but had no effect on the duration of breastfeeding or the rate of exclusive breastfeeding. Compared to the control group who had not been exposed to antidepressants, recent use or restart of antidepressants was associated with a 63% lower likelihood of primary breastfeeding at 6 months, a 51% lower likelihood of any breastfeeding, and a 2.6-fold increased risk of abrupt cessation of breastfeeding. No specific antidepressant was mentioned. Treatment: Treatment should include general measures for any antidepressant overdose. Ensure a patent airway, oxygenation, and ventilation. Monitor heart rhythm and vital signs. General supportive and symptomatic treatment is recommended. Induction of vomiting is not recommended. If necessary, gastric lavage may be performed shortly after administration or in symptomatic patients using a large-bore gastric tube, with appropriate airway protection. Activated charcoal should be administered. Due to the high distribution of this drug, forced diuresis, dialysis, hemoperfusion, and exchange transfusion may be ineffective. There is currently no specific antidote for fluvoxamine. Protein binding: ~77-80% (plasma proteins). |
||
| References | |||
| Additional Infomation |
Fluvoxamine maleate belongs to the (trifluoromethyl)benzene class of compounds. Fluvoxamine maleate is the maleate salt form of fluvoxamine, a 2-aminoethyl oxime ether of an aralkyl ketone with antidepressant, anti-obsessive-compulsive, and anti-bulimia activities. Fluvoxamine blocks the reuptake of serotonin by inhibiting the serotonin reuptake pump on the presynaptic neuronal membrane, leading to elevated serotonin levels in the synaptic cleft. This results in enhanced serotonergic transmission and decreased serotonin turnover, thus producing antidepressant and anti-obsessive-compulsive effects. It is a selective serotonin reuptake inhibitor used to treat depression and various anxiety disorders. See also: Fluvoxamine (containing the active moiety). Fluvoxamine is an oxime ether in which the benzene ring is substituted at position 1 with (1E)-N-(2-aminoethoxy)-5-methoxypentanoimide and at position 4 with a trifluoromethyl group. It is a selective serotonin reuptake inhibitor used to treat obsessive-compulsive disorder. It has antidepressant, serotonin reuptake inhibitor, and anti-anxiety effects. It is a 5-methoxypentanone O-(2-aminoethyl)oxime, belonging to the (trifluoromethyl)benzene class of compounds. Its function is similar to (trifluoromethyl)benzene. Fluvoxamine is an antidepressant whose pharmacological mechanism of action is selective serotonin reuptake inhibitor (SSRI). Although it belongs to the same class as other SSRIs, it is most commonly used to treat obsessive-compulsive disorder. Fluvoxamine has been used clinically since 1983, and its clinical trial database contains data from approximately 35,000 patients. It was launched in the United States in December 1994 and in Japan in June 1999. By the end of 1995, more than 10 million patients worldwide had received fluvoxamine treatment. Fluvoxamine is a serotonin reuptake inhibitor. The mechanism of action of fluvoxamine is as a serotonin reuptake inhibitor. Fluvoxamine is a selective serotonin reuptake inhibitor (SSRI) used to treat obsessive-compulsive disorder (OCD). Fluvoxamine treatment may cause a transient, asymptomatic increase in serum transaminase levels, and there have been rare reports of clinically significant acute liver injury. Fluvoxamine is a 2-aminoethyl oxime ether of an aralkyl ketone with antidepressant, anti-OCD, and anti-anxiety effects. Unlike other selective serotonin reuptake inhibitors, fluvoxamine selectively blocks the reuptake of serotonin by inhibiting the serotonin reuptake pump on the presynaptic neuronal membrane. This increases serotonin levels in the synaptic cleft, prolongs serotonergic transmission time, and reduces serotonin metabolic turnover, thereby producing antidepressant, anti-anxiety, and anti-OCD effects. In vitro studies have shown that fluvoxamine has no significant affinity for histaminergic receptors, α or β adrenergic receptors, muscarinic receptors, or dopaminergic receptors.
Fluvoxamine is an antidepressant whose mechanism of action is that of a selective serotonin reuptake inhibitor (SSRI). Although it belongs to the same class of SSRIs as other drugs, it is most commonly used to treat obsessive-compulsive disorder (OCD). Fluvoxamine has been used in clinical practice since 1983, and its clinical trial database contains data from approximately 35,000 patients. It was launched in the United States in December 1994 and in Japan in June 1999. By the end of 1995, more than 10 million patients worldwide had received fluvoxamine treatment. Fluvoxamine is a selective serotonin reuptake inhibitor used to treat depression and various anxiety disorders. See also: Fluvoxamine maleate (in salt form); (Z)-Fluvoxamine (note moved here). Drug Indications Primarily used to treat depression and obsessive-compulsive disorder (OCD). It has also been used to treat bulimia nervosa. FDA Label Mechanism of Action The exact mechanism of action of fluvoxamine is not fully understood, but it appears to be related to its inhibition of serotonin uptake by neurons in the central nervous system. Fluvoxamine blocks the reuptake of serotonin by the serotonin reuptake pump on the neuronal membrane, enhancing the effect of serotonin on its own receptor, 5HT1A. Studies have also shown that although fluvoxamine has a binding affinity for σ1 receptors, it has almost no affinity for α1 or α2 adrenergic receptors, β-adrenergic receptors, muscarinic receptors, dopamine D2 receptors, histamine H1 receptors, GABA-benzodiazepine receptors, opioid receptors, 5-HT1 receptors, or 5-HT2 receptors. Pharmacodynamics Fluvoxamine is an aryl ketone derivative, belonging to the class of selective serotonin reuptake inhibitors (SSRIs) antidepressants, and its structure differs from other SSRIs. It is used to treat depression associated with mood disorders. It is sometimes used to treat body dysmorphic disorder and anxiety disorders. The antidepressant, anti-obsessive-compulsive, and anti-bulimia effects of fluvoxamine are thought to be related to its inhibition of serotonin reuptake by neurons in the central nervous system. In vitro studies have shown that fluvoxamine is a potent and selective inhibitor of neuronal serotonin reuptake, with only very weak effects on the neuronal reuptake of norepinephrine and dopamine. Furthermore, besides binding to σ1 receptors, fluvoxamine has no significant affinity for adrenergic (α1, α2, β), cholinergic, GABA, dopaminergic, histaminergic, serotonergic (5HT1A, 5HT1B, 5HT2), or benzodiazepine receptors. Some studies hypothesize that the antagonistic effects of these receptors are related to the various anticholinergic, sedative, and cardiovascular effects of certain psychotropic drugs. In addition, some studies have shown that long-term use of fluvoxamine downregulates norepinephrine receptors in the brain (consistent with observations of other effective drugs for treating major depressive disorder), while other studies have shown the opposite. Reason: Therapy combining atypical antipsychotics with serotonin reuptake inhibitors (STIs) has recently been shown to be beneficial for various neuropsychiatric disorders, such as major depressive disorder, schizophrenia, and obsessive-compulsive disorder. Objective: To investigate the effects of atypical antipsychotics combined with STIs on extracellular serotonin [5-HT]ex and dopamine [DA]ex in different brain regions. Methods: The effects of quetiapine (10 mg/kg) combined with fluvoxamine (10 mg/kg) on [5-HT]ex and [DA]ex in the dorsal striatum, prefrontal cortex, and nuclei of rats were compared. Microdialysis-high performance liquid chromatography-electrochemical detection was used to detect [DA]ex and [5-HT]ex levels in the nucleus accumbens (core and putamen) and thalamus. Results: Quetiapine had no significant effect on [DA]ex and [5-HT]ex levels in the prefrontal cortex and thalamus, but it increased [DA]ex and [5-HT]ex levels in the dorsal striatum. In the nucleus accumbens, quetiapine increases [DA]ex levels and decreases [5-HT]ex levels. Fluvoxamine increases [5-HT]ex levels in all brain regions and increases [DA]ex levels in the striatum. Compared with baseline, the combination of quetiapine and fluvoxamine increases [DA]ex and [5-HT]ex levels in all brain regions. Although neither quetiapine nor fluvoxamine alone affects dopamine efflux (DAex) levels in the prefrontal cortex and thalamus, the combination significantly increases DAex levels in these two brain regions. Conclusion: The combination of quetiapine and fluvoxamine synergistically increases dopamine levels in the prefrontal cortex and thalamus. [1] In summary, the combination of quetiapine and fluvoxamine causes a unique synergistic increase in dopamine levels in the prefrontal cortex and thalamus. Whether this synergistic increase in DAex levels in the prefrontal cortex and thalamus is related to the efficacy of atypical antipsychotics combined with selective serotonin reuptake inhibitors (SSRIs) in treating atypical antipsychotics remains to be further investigated. [1] |
| Molecular Formula |
C19H25F3N2O6
|
|
|---|---|---|
| Molecular Weight |
434.41
|
|
| Exact Mass |
434.166
|
|
| Elemental Analysis |
C, 52.53; H, 5.80; F, 13.12; N, 6.45; O, 22.10
|
|
| CAS # |
61718-82-9
|
|
| Related CAS # |
Fluvoxamine; 54739-18-3; (E)-Fluvoxamine-d4 maleate; 1432075-74-5; Fluvoxamine-d4 maleate
|
|
| PubChem CID |
9560989
|
|
| Appearance |
White to off-white solid powder
|
|
| Boiling Point |
370.6ºC at 760 mmHg
|
|
| Melting Point |
120-121.5ºC
|
|
| Flash Point |
177.9ºC
|
|
| LogP |
3.613
|
|
| Hydrogen Bond Donor Count |
3
|
|
| Hydrogen Bond Acceptor Count |
11
|
|
| Rotatable Bond Count |
11
|
|
| Heavy Atom Count |
30
|
|
| Complexity |
446
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
FC(C1C([H])=C([H])C(=C([H])C=1[H])/C(/C([H])([H])C([H])([H])C([H])([H])C([H])([H])OC([H])([H])[H])=N\OC([2H])([2H])C([2H])([2H])N([H])[H])(F)F.O([H])C(/C(/[H])=C(/[H])\C(=O)O[H])=O
|
|
| InChi Key |
LFMYNZPAVPMEGP-PIDGMYBPSA-N
|
|
| InChi Code |
InChI=1S/C15H21F3N2O2.C4H4O4/c1-21-10-3-2-4-14(20-22-11-9-19)12-5-7-13(8-6-12)15(16,17)18;5-3(6)1-2-4(7)8/h5-8H,2-4,9-11,19H2,1H3;1-2H,(H,5,6)(H,7,8)/b20-14+;2-1-
|
|
| Chemical Name |
(Z)-but-2-enedioic acid;2-[(E)-[5-methoxy-1-[4-(trifluoromethyl)phenyl]pentylidene]amino]oxyethanamine
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.75 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.75 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (5.75 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 20 mg/mL (46.04 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. Solubility in Formulation 5: 20 mg/mL (46.04 mM) in phosphate buffer Saline (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3020 mL | 11.5099 mL | 23.0197 mL | |
| 5 mM | 0.4604 mL | 2.3020 mL | 4.6039 mL | |
| 10 mM | 0.2302 mL | 1.1510 mL | 2.3020 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT04885530 | Active Recruiting |
Drug: Ivermectin Drug: Fluvoxamine Drug: Fluticasone |
Covid19 | Susanna Naggie, MD | June 8, 2021 | Phase 3 |
| NCT04510194 | Active Recruiting |
Drug: Metformin Drug: Placebo Drug: Fluvoxamine |
Covid19 SARS-CoV Infection |
University of Minnesota | January 1, 2021 | Phase 3 |
| NCT04160377 | Recruiting | Drug: Fluvoxamine | Depressive Disorder Endogenous Depression Melancholia |
Lingjiang Li | August 1, 2019 | Phase 2 |
| NCT04963257 | Recruiting | Drug: sertraline fluvoxamine Drug: sertraline |
OCD | Second Affiliated Hospital, School of Medicine, Zhejiang University |
January 1, 2020 | Phase 4 |
| NCT05874037 | Recruiting | Drug: Fluvoxamine | Long COVID | Washington University School of Medicine |
May 15, 2023 | Phase 2 Phase 3 |
 |
|---|
 |
 |