
| Size | Price | Stock | Qty |
|---|---|---|---|
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| 5g |
|
||
| 10g |
|
||
| Other Sizes |
Purity: ≥98%
Erlotinib HCl (formerly OSI-744, OSI744; trade name: Tarceva), the hydrochloride salt of erlotinib, is an EGFR (epidermal growth factor receptor) inhibitor with antitumor activity. In cell-free experiments, it inhibits EGFR with an IC50 of 2 nM, and when compared to human c-Src or v-Abl, it is >1000 times more sensitive to inhibit EGFR. The FDA and other nations have authorized erlotinib, a quinazoline derivative, for the treatment of pancreatic cancer, non-small cell lung cancer (NSCLC), and various other cancer types.
| Targets |
EGFR (IC50 = 2 nM)
|
|---|---|
| ln Vitro |
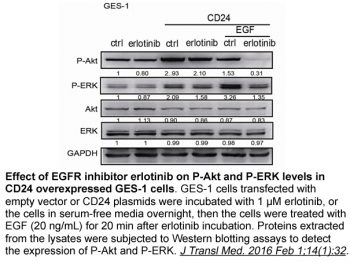
Erlotinib HCl potently inhibits EGFR activation in intact cells, such as MDA MB-468 human breast cancer cells, DiFi human colon cancer cells, and HNS human head and neck tumor cells (IC50 20 nM). DiFi human colon cancer cells undergo apoptosis when exposed to 1 μM erlotinib HCl.[1] With an IC50 ranging from 29 nM to >20 μM, erlotinib inhibits the growth of a panel of NSCLC cell lines, including A549, H322, H3255, H358 H661, H1650, H1975, H1299, and H596.[2] Erlotinib HCl (2 μM) strongly suppresses the proliferation of BxPC-3 and AsPC-1 pancreatic cells.[3] When combined with gemcitabine, the effects of erlotinib HCl are thought to be additive in pancreatic cancer cells that have mutated KRAS. EGFR phosphorylation at the Y845 (Src-dependent phosphorylation) and Y1068 (auto-phosphorylation) sites is inhibited by ten micromolar of erlotinib HCl.[4] When combined with erlotinib HCl, rapamycin-stimulated Akt activity may be down-regulated, and this has a synergistic effect on inhibiting cell growth. [5]
Erlotinib (CP-358774) is also a potent inhibitor, with an IC50 of 1 nM, of the EGFR's recombinant intracellular (kinase) domain. Erlotinib severely inhibits the proliferation of DiFi cells, with an IC50 of 100 nM for an 8-day proliferation assay[1]. When B-DIM and Erlotinib (2 μM) are combined, BxPC-3 cell colony formation is significantly inhibited as opposed to when either agent is used alone. Only in BxPC-3 cells does the combination of B-DIM and Erlotinib (2 μM) significantly induce apoptosis when compared to the apoptotic effect of either agent alone[2]. The epidermal growth factor receptor (EGFR) is overexpressed in a significant percentage of carcinomas and contributes to the malignant phenotype. CP-358,774 is a directly acting inhibitor of human EGFR tyrosine kinase with an IC50 of 2 nM and reduces EGFR autophosphorylation in intact tumor cells with an IC50 of 20 nM. This inhibition is selective for EGFR tyrosine kinase relative to other tyrosine kinases we have examined, both in assays of isolated kinases and whole cells. At doses of 100 mg/kg, CP-358,774 completely prevents EGF-induced autophosphorylation of EGFR in human HN5 tumors growing as xenografts in athymic mice and of the hepatic EGFR of the treated mice. CP-358,774 inhibits the proliferation of DiFi human colon tumor cells at submicromolar concentrations in cell culture and blocks cell cycle progression at the G1 phase. This inhibitor produces a marked accumulation of retinoblastoma protein in its underphosphorylated form and accumulation of p27KIP1 in DiFi cells, which may contribute to the cell cycle block. Inhibition of the EGFR also triggers apoptosis in these cells as determined by formation of DNA fragments and other criteria. These results indicate that CP-358,774 has potential for the treatment of tumors that are dependent on the EGFR pathway for proliferation or survival. [1] Effects of B-DIM and Erlotinib on the Viability of Pancreatic Cancer Cells [2] It is important to note that, during our pilot studies, as indicated in Materials and Methods, different concentrations of B-DIM and erlotinib were used and are presented in Table 1. Moreover, after analyzing the basal level of expression of EGFR, NF-κB, and COX-2, we chose two cell lines having constitutively activated levels of NF-κB, EGFR, and COX-2 expression (BxPC-3) compared with lower level of NF-κB, EGFR, and COX-2 expression (MIAPaCa). Our results prompted us to select the subsequent concentration of B-DIM and erlotinib as presented below. Cell viability of BxPC-3 and MIAPaCa pancreatic cancer cells treated with B-DIM (20 µmol/L), erlotinib (2 µmol/L), and the combination was determined by the MTT assay, and the data are presented in Fig. 1A and B. Significant inhibition of cell viability was seen in BxPC-3 cells treated with either agent, and this was further enhanced by the combination treatment (P = 0.0001). In addition, we have also tested the effects of treatment on cell viability by clonogenic assay as shown below. Similar treatments of MIAPaCa cells resulted in a significant inhibition of viable cells with B-DIM alone but not when exposed to similar concentrations of B-DIM and erlotinib for the same time, and the effect was not enhanced by the combination treatment (P = 0.0890). The insensitivity of MIAPaCa cells to erlotinib is consistent with a recently published report Inhibition of Cell Growth/Survival by Clonogenic Assay [2] To determine the effect of B-DIM and Erlotinib on cell growth, cells were treated with each of the single agents or their combination and assessed for cell viability by clonogenic assay. The combination of B-DIM and erlotinib resulted in a significant inhibition of colony formation in BxPC-3 cells when compared with either agent alone (Fig. 2A and B). Similar treatment of MIAPaCa cells (Fig. 2C) showed inhibition of colony formation with B-DIM alone and also the combination, but the effect was not enhanced with the combination treatment as was seen in BxPC-3 cells (Fig. 2A and B). These results were similar to those obtained from the soft-agar assay. Overall, the results from clonogenic assay was consistent with the MTT data as shown in Fig. 1A and B, suggesting that B-DIM had a differential effect between BxPC-3 and MIAPaCa pancreatic cancer cells. The mechanisms of such differences were further investigated, and the results are presented in the following sections, but first we have determined the effects of B-DIM, erlotinib, and the combination on apoptotic cell death. Induction of Apoptosis by Erlotinib, B-DIM, and the Combination [2] The underlying mechanism on the inhibition of cell viability was further studied by determining the apoptotic effects of different treatments using the Cell Death Detection ELISA. The combination of B-DIM and erlotinib resulted in a significant induction of apoptosis only in BxPC-3 cells when compared with the apoptotic effect of either agent alone (Fig. 1C). Similar treatment of MIAPaCa cells showed no induction of apoptosis with the combination (Fig. 1D). These results are consistent with cell viability assay by MTT. Subsequently, we sought to find further evidence of apoptosis as presented below. B-DIM Enhances Apoptosis Signaling by Erlotinib [2] PARP cleavage was determined in BxPC-3 and MIAPaCa cells that were treated with B-DIM (20 µmol/L), erlotinib (2 µmol/L), and the combination (Fig. 3). We found significant amount of PARP (116 kDa) protein cleavage product (85 kDa fragment) after 72-h treatment only in BxPC-3 cells (Fig. 3). In contrast, MIAPaCa cells treated similarly showed only a small cleavage of PARP with B-DIM alone and also in combination but not with erlotinib alone. The induction of apoptosis could be partly due to inactivation of important survival genes; hence, we investigated whether B-DIM, erlotinib, and their combination could affect key survival proteins. Effect of B-DIM on Molecules Related to Apoptosis [2] BxPC-3 and MIAPaCa cells were used to evaluate the effects of B-DIM and/or Erlotinib on the expression of survivin, Bcl-2, Bcl-xL, and c-IAP1/2. Expression of Bcl-2, Bcl-xL, survivin, and c-IAP1/2 proteins was significantly reduced in cells treated with the combination when compared with either agent alone (Fig. 3). There was no influence on antiapoptotic proteins in MIAPaCa cells treated with either agent alone or the combination. These results suggest that B-DIM, erlotinib, and the combination down-regulate key survival proteins and in turn induced apoptotic cell death in BxPC-3 cells but not in MIAPaCa cells. To further determine the molecular mechanism by which B-DIM sensitized BxPC-3 cells to erlotinib-induced inhibition of cell viability and induction of apoptosis, we investigated the role of EGFR and its downstream signaling pathways. Effect of B-DIM on the Expression of EGFR Protein [2] The expression of EGFR was determined by immunoblotting. No baseline expression of EGFR was found in the MIAPaCa cells. EGFR-expressing BxPC-3 cells showed a significant reduction in the expression of EGFR and phosphorylated EGFR levels when exposed to erlotinib plus B-DIM compared with either agent alone (Fig. 3). It is known that the activation of EGFR could in turn regulate an important transcription factor, NF-κB, which is a known regulator of several survival genes such as survivin, c-IAP1/2, Bcl-2, and Bcl-xL. Because we found a greater degree of down-regulation of survivin, c-IAP1/2, Bcl-2, and Bcl-xL in BxPC-3 cells treated with B-DIM and erlotinib compared with either agent alone, and because these genes are transcriptionally regulated by NF-κB, we investigated the effect of each treatment on the DNA-binding activity of NF-κB. B-DIM Inhibits NF-κBDNA-Binding Activity [2] The activation of NF-κB, a nuclear transcriptional factor, was assessed in B-DIM-treated and Erlotinib-treated cells. There was a significant inhibition of NF-κB activation in BxPC-3 cells exposed to both erlotinib and B-DIM compared with erlotinib alone (Fig. 4A). No such inhibition was shown in the MIAPaCa cells (Fig. 4B). These results suggest that the combination of B-DIM and erlotinib causes greater inhibition of cell growth, induction of apoptosis, inhibition of survival factors, inhibition of EGFR, and inactivation of NF-κB. Because NF-κB plays important roles in the regulation of prosurvival and antiapoptotic processes, we tested whether overexpression of NF-κB by p65 cDNA transfection could abrogate B-DIM-induced and erlotinib-induced apoptotic processes. Moreover, it is known that NF-κB transcriptionally regulates COX-2, which produces PGE2 and in turn induces cell viability. Thus, we tested whether celecoxib, erlotinib, or B-DIM alone could influence the activity of B-DIM and erlotinib in p65 cDNA transfected cells. Erlotinib, B-DIM, and Celecoxib Abrogated Activation of NF-κBActivity Stimulated by p65 cDNA Transfection [2] Cytoplasmic and nuclear proteins from BxPC-3 and MIAPaCa cells transfected with p65 cDNA and then treated with erlotinib (2 µmol/L), B-DIM (20 µmol/L), or celecoxib (5 µmol/L) or left untreated for 48 h were subjected to analysis for NF-κB activity as measured by Western blot analysis and EMSA. The results showed that erlotinib, B-DIM, and celecoxib inhibited the p65 protein and NF-κB DNA-binding activity more in BxPC-3 cells compared with untreated cells (Fig. 5A and B) and very little effect was seen in MIAPaCa cells. Importantly, NF-κB p65 cDNA transfection enhanced the NF-κB p65 protein and DNA-binding activity only in BxPC-3 cells to a significant level as shown in Fig. 5A and B. On the other hand, no such changes were observed in the MIAPaCa cells. Because the activation of NF-κB induces COX-2 expression leading to the production of PGE2 that is released into the culture medium, we measured the levels of PGE2 in untransfected and transfected cells treated with erlotinib, B-DIM, and the COX-2 inhibitor celecoxib. Inhibition of PGE2 Synthesis in p65 cDNA-Transfected Cells [2] We measured the levels of PGE2 in the conditioned medium collected from both BxPC-3 and MIAPaCa cells as an indicator of COX-2 activity. We found a high level of PGE2 secretion by BxPC-3 cells, whereas MIAPaCa cells showed very low levels of PGE2, which is consistent with its low constitutive expression of COX-2. BxPC-3 and MIAPaCa cells were transfected with p65 cDNA followed by treatment with Erlotinib (10 nmol/L), B-DIM (1 µmol/L), or celecoxib (1 nmol/L) to analyze the levels of PGE2 released into the culture medium (Fig. 5C). No change in PGE2 level was noted when cells were treated with erlotinib alone (P = 0.084). However, a significant reduction in PGE2 level was observed in BxPC-3 cells treated with B-DIM (P = 0.006) and celecoxib (P = 0.005). There was a substantial increase in the PGE2 level in p65 cDNA-transfected BxPC-3 cells compared with untransfected cells (P = 0.009), suggesting that NF-κB could induce COX-2 expression. However, there was no change in PGE2 level in MIAPaCa cells with any of the agents. Collectively, these results suggest that the production of PGE2 is mediated through the NF-κB and COX-2 pathway and that celecoxib could down-regulate both NF-κB and COX-2. These results were subsequently correlated with the degree of apoptosis (Fig. 5D) as presented below. Apoptosis through the Inactivation of NF-κB in p65 cDNA-Transfected Cells [1] p65 cDNA was transfected into BxPC-3 and MIAPaCa cells and then treated with Erlotinib (2 µmol/L), B-DIM (20 µmol/L), or celecoxib (5 µmol/L) for 48 h (Fig. 5D). The degree of apoptosis in p65 cDNA-transfected BxPC-3 cells treated with erlotinib (P = 0.034) was much less compared with untransfected cells treated with erlotinib (P = 0.007). Similar results were observed with both B-DIM and celecoxib treatment in BxPC-3 cells. However, in MIAPaCa cells, no such degree of apoptosis was observed. These results suggest that activation of NF-κB by p65 cDNA transfection could abrogate the apoptosis inducing effect of erlotinib, B-DIM, and celecoxib. |
| ln Vivo |
Erlotinib HCl fully inhibits EGF-induced autophosphorylation of EGFR in human HN5 tumors growing as xenografts in athymic mice, as well as of the treated mice's hepatic EGFR, at doses of 100 mg/kg.[1] H460a and A549 tumor models are inhibited by erlotinib HCl (100 mg/Kg) at 71 and 93% inhibition rates, respectively.[5]
In comparison to the untreated control, the combination of B-DIM and Erlotinib (50 mg/kg, i.p.) treatment significantly (P <0.01) reduces tumor weight under experimental conditions[2]. In comparison to the CP+vehicle (V) rats, erlotinib (20 mg/kg, p.o.) significantly attenuates the body weight (BW) loss induced by Cisplatin (CP) (P<0.05). Treatment with erlotinib considerably enhances renal function in CP-N (normal control group, NC) rats. Compared to the CP+V rats, the CP+Erlotinib (E) rats exhibit a significant increase in urine volume (UV) (P<0.05) and Cr clearance (Ccr) (P<0.05), as well as a significant decrease in serum creatinine (s-Cr) (P<0.05), blood urea nitrogen (BUN) (P<0.05), and urinary N-acetyl-β-D-glucosaminidase (NAG) index (P<0.05). B-DIM Augments In vivo Therapeutic Effect of Erlotinib on Primary Tumor [2] Potential therapeutic utility of B-DIM and erlotinib combination in SCID mice bearing orthotopically implanted BxPC-3 pancreatic tumor cells was investigated. A dose of 3.5 mg/d B-DIM per mouse was selected for p.o. administration, whereas erlotinib dose (50 mg/kg body weight i.p.) was based on previously published reports as shown in Fig. 6A. A total of 28 mice were divided into four groups. To ascertain the efficacy of a single-agent treatment compared with combinations, we determined the mean pancreas weight in all treated groups. Under our experimental conditions, administration of B-DIM by gavage treatment and erlotinib alone caused 20% and 35% reduction in tumor weight, respectively, compared with control tumors (Fig. 6C). However, under the experimental conditions, the combination of B-DIM and erlotinib treatment showed significant decrease (P < 0.01) in tumor weight compared with untreated control, B-DIM alone, or erlotinib alone treatment group. These results showed, for the first time, the efficacy of combination of B-DIM and erlotinib in the inhibition of pancreatic tumor growth in an orthotopic model. B-DIM Inhibits NF-κBDNA-Binding Activity In vivo [2] The activation of NF-κB was assessed in B-DIM-treated and Erlotinib-treated tumor tissues. The results show that NF-κB was down-regulated by B-DIM and erlotinib (Fig. 6B). Fig. 6B (bottom) represents results from all seven mice. These in vivo results were similar to our in vitro findings, suggesting that the inactivation of NF-κB is, at least, one of the molecular mechanisms by which B-DIM potentiates erlotinib-induced antitumor activity in our experimental animal model. The effects of blocking the epidermal growth factor receptor (EGFR) in acute kidney injury (AKI) are controversial. Here we investigated the renoprotective effect of Erlotinib, a selective tyrosine kinase inhibitor that can block EGFR activity, on cisplatin (CP)-induced AKI. Groups of animals were given either Erlotinib or vehicle from one day before up to Day 3 following induction of CP-nephrotoxicity (CP-N). In addition, we analyzed the effects of erlotinib on signaling pathways involved in CP-N by using human renal proximal tubular cells (HK-2). Compared to controls, rats treated with erlotinib exhibited significant improvement of renal function and attenuation of tubulointerstitial injury, and reduced the number of apoptotic and proliferating cells. Erlotinib-treated rats had a significant reduction of renal cortical mRNA for profibrogenic genes. The Bax/Bcl-2 mRNA and protein ratios were significantly reduced by erlotinib treatment. In vitro, we observed that erlotinib significantly reduced the phosphorylation of MEK1 and Akt, processes that were induced by CP in HK-2. Taken together, these data indicate that erlotinib has renoprotective properties that are likely mediated through decreases in the apoptosis and proliferation of tubular cells, effects that reflect inhibition of downstream signaling pathways of EGFR. These results suggest that erlotinib may be useful for preventing AKI in patients receiving CP chemotherapy[PLoS One. 2014 Nov 12;9(11):e111728.]. |
| Enzyme Assay |
The process of coating 96-well plates involves incubating 100 μL of 0.25 mg/mL PGT in PBS per well for an entire night at 37 °C. Aspiration is used to remove excess PGT, and three washing buffer washes (0.1% Tween 20 in PBS) are performed on the plate. 50 μL of 50 mM HEPES (pH 7.3) containing 0.1 mM sodium orthovanadate, 125 mM sodium chloride, 24 mM magnesium chloride, 20 μM ATP, 1.6 μg/mL EGF, and 15 ng of affinity-purified EGFR from A431 cell membranes is used for the kinase reaction. A final DMSO concentration of 2.5% is achieved by adding erlotinib HCl in DMSO. When ATP is added, phosphorylation begins and continues for eight minutes at room temperature while being constantly shaken.The kinase reaction is terminated by aspiration of the reaction mixture and is washed 4 times with washing buffer. Phosphorylated PGT is measured by 25 minutes of incubation with 50 μL per well HRP-conjugated PY54 antiphosphotyrosine antibody, diluted to 0.2 μg/mL in blocking buffer (3% BSA and 0.05% Tween 20 in PBS). Antibody is removed by aspiration, and the plate is washed 4 times with washing buffer. The colonmetric signal is developed by addition of TMB Microwell Peroxidase Substrate, 50μL per well, and stopped by the addition of 0.09 M sulfuric acid, 50 μL per well. Phosphotyrosine is estimated by measurement of absorbance at 450 nm. In wells without AlP, EGFR, or PGT, the signal for controls is usually between 0.6 and 1.2 absorbance units, with almost no background, and it is proportional to the incubation period of 10 minutes.
|
| Cell Assay |
Seeded in triplicate, exponentially growing cells are subjected for 72 hours to serial dilutions of erlotinib, pemetrexed, or the combination at a constant concentration ratio of 4:1. Cell count and the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay are used to measure cell viability. The percentage of drug-treated control cells that survive compared to PBS-treated control cells (which are thought to be 100% viable) is known as growth inhibition. The CalcuSyn software determines the IC50 value, which is the concentration at which a 72-hour exposure to drug(s) results in a 50% inhibition of cell growth when compared to untreated control cells.
In order to assess the survival of cells treated with B-DIM, Erlotinib, or both, 3,000–5,000 BxPC-3 and MIAPaCa cells are plated per well in a 96-well plate and incubated at 37°C for the entire night. Initially, B-DIM (10-50 µM) and Erlotinib (1-5 µM) are tested at a range of concentrations. The concentrations of B-DIM (20 µM) and Erlotinib (2 µM) are selected for each assay based on the preliminary findings. The standard MTT assay is used to measure the effects of B-DIM (20 µM), Erlotinib (2 µM), and the combination on BxPC-3 and MIAPaCa cells. The assay is performed three times after 72 hours. The Tecan microplate fluorometer measures the color intensity at 595 nm. Cells treated with DMSO are given a value of 100% and are regarded as the untreated control. We have performed clonogenic assay in addition to the aforementioned assay to evaluate the effects of treatment[2]. Cell Viability Assay [2] To test the viability of cells treated with B-DIM, Erlotinib, or the combination, BxPC-3 and MIAPaCa cells were plated (3,000–5,000 per well) in a 96-well plate and incubated overnight at 37°C. We initially tested a range of concentrations for both B-DIM (10–50 µmol/L) and erlotinib (1–5 µmol/L). Based on the initial results, the concentration of B-DIM (20 µmol/L) and erlotinib (2 µmol/L) were chosen for all assays. The effects of B-DIM (20 µmol/L), erlotinib (2 µmol/L), and the combination on BxPC-3 and MIAPaCa cells were determined by the standard 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay after 72 h and was repeated three times. The color intensity was measured by a Tecan microplate fluorometer at 595 nm. DMSO-treated cells were considered to be the untreated control and assigned a value of 100%. In addition to the above assay, we have also done clonogenic assay for assessing the effects of treatment as shown below. Clonogenic Assay [2] To test the survival of cells treated with B-DIM, Erlotinib, or the combination, BxPC-3 and MIAPaCa cells were plated (50,000–100,000 per well) in a six-well plate and incubated overnight at 37°C. After 72-h exposure to 20 µmol/L B-DIM, 2 µmol/L erlotinib, and the combination, the cells were trypsinized, and the viable cells were counted (trypan blue exclusion) and plated in 100 mm Petri dishes in a range of 100 to 1,000 cells to determine the plating efficiency as well as assess the effects of treatment on clonogenic survival. The cells were then incubated for ~10 to 12 days at 37°C in a 5% CO2/5% O2/90% N2 incubator. The colonies were stained with 2% crystal violet and counted. The surviving fraction was normalized to untreated control cells with respect to clonogenic efficiency, which was 83% for both BxPC-3 and MIAPaCa cells. In addition to this assay, cells were also treated similarly and plated in soft-agar (soft-agar colony assay) and incubated at 37°C. The colonies in the soft agar were also counted in all untreated and treated wells after 12 days. Quantification of Apoptosis by ELISA [2] The Cell Death Detection ELISA kit (Roche Applied Science) was used to detect apoptosis in untreated and treated BxPC-3 and MIAPaCa cells. Cells seeded in six-well plates were treated with B-DIM (20 µmol/L), Erlotinib (2 µmol/L), or the combination. The cells were trypsinized and ~10,000 cells were used as described earlier. Tecan microplate fluorometer was used to measure color intensity at 405 nm. The experiment was repeated three times. Protein Extraction and Western Blot Analysis [2] BxPC-3 and MIAPaCa cells treated with B-DIM (20 µmol/L), Erlotinib (2 µmol/L), or the combination for 72 h were used to evaluate the effects of treatment on survivin, Bcl-2, Bcl-xL, EGFR, EGFR-pTyr1173, c-IAP1/2, Src, poly(ADP-ribose) polymerase (PARP), and β-actin expression. The experiment was carried out for a minimum of three times. Cells were harvested as described previously. The samples were loaded on 7% to 12% SDSPAGE for separation and electrophoretically transferred to a nitrocellulose membrane. Each membrane was incubated with monoclonal antibody against survivin, Bcl-2, Bcl-xL, Src, c-IAP1/2, EGFR, EGFR-pTyr1173, PARP, and β-actin. Blots were incubated with secondary antibodies conjugated with peroxidase. The signal intensity was then measured using chemiluminescent detection system. Electrophoretic Mobility Shift Assay for NF-κB Activation [2] To evaluate the effect of B-DIM and Erlotinib on BxPC-3 and MIAPaCa cells, the cells were either untreated or treated with B-DIM (20 µmol/L), erlotinib (2 µmol/L), or the combination with a minimum repeat of experiment at least three times for 72 h. The cells or the minced tumor tissue were homogenized using a Dounce homogenizer in 400 µL ice-cold lysis buffer as described earlier. |
| Animal Protocol |
Mice: Erlotinib (5 mg/kg) is administered p.o. or i.p. to Bcrp1/Mdr1a/1b-/- and WT mice. The selection of i.p. administration is predicated on full bioavailability and optimal drug absorption. Three series of samples are taken from the lateral tail vein tip. Whole blood samples are taken during the first series at 15, min, 0.5, 1.5, 5, and 10 h following injection. The sampling times of the two subsequent series are adjusted to 5 and 15 minutes and 0.5, 1.5, 4, and 8 hours after injection based on the findings of this initial group. Blood samples are collected, centrifuged right away, and the plasma is kept at -20°C until high-performance liquid chromatographic analysis is performed.
Rats: There are male Crl:CD (SD) rats (244-297 g) that are seven weeks old. Erlotinib hydrochloride (10 mg/kg and 20 mg/kg) is given orally to the animals by gavage. Mice: The treatment groups consist of seven randomly assigned female ICR-SCID mice, aged 6-7 weeks: (a) control (no treatment); (b) B-DIM (50 mg/kg body weight) administered intragastrically once daily; (c) Erlotinib (50 mg/kg body weight) administered daily intraperitoneally for 15 days; and (d) B-DIM and Erlotinib administered according to the schedule for individual treatments. After receiving their last dose of medication, all mice are killed on day three, and their body weight is recorded. A portion of the tissue is immediately frozen in liquid nitrogen and kept cold (−70°C) for later use, while the remaining portion is fixed in formalin and prepared for paraffin block processing. The presence of a tumor or tumors in each pancreas is verified by staining a fixed tissue section with H&E. Rats: Male Sprague-Dawley (SD) rats six weeks of age, weighing 180–210 g, are utilized. On day 0, SD rats (n=28) receive an intraperitoneal injection of 7 mg/kg of freshly prepared ciprofloxacin (CP) at a concentration of 1 mg/mL. For the purpose of examining Erlotinib's effects, 28 CP-N rats are split into two groups. Animals in two groups (n = 14) are given daily oral gavages of either Erlotinib (20 mg/kg) (CP+E, n = 14) or vehicle (CP+V, n = 14) from day -1 (24 hours before the CP injection) to day 3. Groups treated with vehicles are given the same amount of saline. At six weeks of age, a normal control group (NC, n = 5) consists of five male SD rats. From the first to the third day, the NC rats receive an equivalent volume of saline orally via gavage. Day 4 (96 hours post-CP injection): rats are anesthetized, and following a cardiac puncture, they are sacrificed by exsanguination. The kidneys and blood are simultaneously extracted. Renal tissue is sectioned and fixed in 2% paraformaldehyde/phosphate-buffered saline (PBS) for later use, or it can be snap-frozen in liquid nitrogen. In order to reduce suffering as much as possible, diethyl ether gas anesthesia is used during all surgical procedures. Mice were randomized into the following treatment groups (n = 7): (a) untreated control; (b) only B-DIM (50 mg/kg body weight), intragastric once every day; (c) Erlotinib (50 mg/kg body weight), everyday i.p. for 15 days; and (d) B-DIM and Erlotinib, following schedule as for individual treatments. All mice were killed on day 3 following last dose of treatment, and their body weight was determined. One part of the tissue was rapidly frozen in liquid nitrogen and stored at −70°C for future use and the other part was fixed in formalin and processed for paraffin block. H&E staining of fixed tissue section was used to confirm the presence of tumor(s) in each pancreas. [2] Cisplatin (CP) was freshly prepared in saline at a concentration of 1 mg ml−1 and then injected intraperitoneally in SD rats (n = 28) at a dose of 7 mg/kg on day 0. The dose of CP was selected based on a previous stud. To investigate the effect of Erlotinib, 28 CP-N rats were divided into two groups. Separate groups (n = 14) each of animals were administered with either Erlotinib (20 mg/kg, Cugai Pharmaceutical/F. Hoffmann-La Roche, Basel, Switzerland) (CP+E, n = 14) or vehicle (CP+V, n = 14) daily by oral gavage from day -1 (24 hours prior to the CP injection) to day 3. Vehicle-treated groups received an equivalent volume of saline. Five male SD rats at the age of 6 weeks were used as a normal control group (NC, n = 5). The NC rats were given an equivalent volume of saline daily by oral gavage from day -1 to day 3. At day 4 (96 hours after CP injection), each rat was anesthetized and sacrificed by exsanguination after the cardiac puncture; blood was collected by cardiac puncture and kidneys were collected (Figure 1). Renal tissue was divided; separate portions were snap-frozen in liquid nitrogen or fixed in 2% paraformaldehyde/phosphate-buffered saline (PBS) for later use. All surgery was performed under diethyl ether gas anesthesia, and all efforts were made to minimize suffering. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Erlotinib is absorbed approximately 60% after oral administration; food significantly increases its bioavailability to near 100%. Peak plasma concentrations occur 4 hours after administration. Erlotinib solubility is affected by pH; solubility decreases with increasing pH. Smoking also reduces erlotinib exposure. After oral administration of 100 mg, 91% of the dose is recovered, with 83% excreted in feces (1% of the dose in its original form) and 8% excreted in urine (0.3% of the dose in its original form). Apparent volume of distribution = 232 L. Smokers have 24% higher erlotinib clearance. Erlotinib is absorbed approximately 60% after oral administration; food significantly increases its bioavailability to near 100%. Peak plasma concentrations are reached 4 hours after administration. Erlotinib solubility is affected by pH; higher pH results in lower solubility. After absorption, approximately 93% of erlotinib binds to plasma albumin and α1-acid glycoprotein. The apparent volume of distribution of erlotinib is 232 liters. The time required to reach steady-state plasma concentrations is 7–8 days. No significant correlation was observed between clearance and covariates such as patient age, weight, or sex. Erlotinib clearance was 24% higher in smokers. After oral administration of 100 mg, 91% of the dose is recovered: 83% is excreted in feces (1% of the intact parent drug dose) and 8% is excreted in urine (0.3% of the intact parent drug dose). For more complete data on absorption, distribution, and excretion of erlotinib (of 10 parameters), please visit the HSDB record page. Metabolism/MetabolitesMetabolism occurs in the liver. In vitro cytochrome P450 metabolism assays showed that erlotinib is primarily metabolized by CYP3A4, followed by CYP1A2 and the extrahepatic isoenzyme CYP1A1. This study was conducted in healthy male volunteers to investigate the metabolism and excretion of the epidermal growth factor receptor tyrosine kinase inhibitor erlotinib. Following a single oral dose of 14C-erlotinib hydrochloride (equivalent to 100 mg of free base, approximately 91 μCi/subject), unmetabolized erlotinib was detected in plasma. The main circulating component was unchanged erlotinib, while the pharmacologically active metabolite M14 accounted for approximately 5% of the total circulating radioactivity. The three main biotransformation pathways of erlotinib were: O-demethylation of the side chain followed by oxidation to carboxylic acid M11 (29.4% of the dose); partial oxidation of acetylene to carboxylic acid M6 (21.0%); and hydroxylation of the aromatic ring to M16 (9.6%). In addition, O-demethylation of M6 to generate M2, O-demethylation of the side chain to generate M13 and M14, and the binding of oxidative metabolites with glucuronic acid (M3, M8, and M18) and sulfate (M9) play minor roles in the metabolism of erlotinib. Identified metabolites account for more than 90% of the total radioactivity recovered in urine and feces. Metabolites observed in humans are similar to those found in toxic animals (rats and dogs). Known metabolites of erlotinib include erlotinib M14. The median half-life is 36.2 hours. A population pharmacokinetic analysis of 591 patients receiving erlotinib hydrochloride monotherapy as second/third-line therapy showed a median half-life of 36.2 hours. |
| Toxicity/Toxicokinetics |
Hepatotoxicity
Elevated serum transaminase levels are common during erlotinib treatment for pancreatic and lung cancer, with at least 10% of patients experiencing ALT levels exceeding five times the upper limit of normal. However, similar ALT elevations can occur with similar antitumor regimens. These abnormalities are usually asymptomatic and resolve spontaneously, but may require dose adjustment or discontinuation (Case 1). In addition, a few cases have been reported as clinically significant liver injury with erlotinib treatment. Liver injury typically occurs within days or weeks of treatment initiation and can be severe, with at least 12 deaths reported in the literature. Liver injury can occur abruptly, and the pattern of serum enzyme elevation is usually hepatocellular (Case 2). Immune allergic reactions (rash, fever, and eosinophilia) are uncommon, and autoantibody formation has not been reported. Routine monitoring of liver function is recommended during treatment. Patients with a history of cirrhosis or liver dysfunction due to hepatic tumor burden have an increased risk of clinically significant liver injury and liver failure. Probability Score: B (Possible but uncommon, a cause of clinically significant liver injury). Effects during pregnancy and lactation ◉ Overview of use during lactation There is currently no information on the clinical use of erlotinib during lactation. Because erlotinib binds to plasma proteins at a rate of up to 93%, its concentration in breast milk may be low. However, its half-life is approximately 36 hours, which may allow it to accumulate in the infant. Furthermore, erlotinib is used in combination with gemcitabine to treat pancreatic cancer, which may increase the risk to the infant. The manufacturer recommends discontinuing breastfeeding during erlotinib treatment and for two weeks after the last dose. ◉ Effects on breastfed infants No published information found as of the revision date. ◉ Effects on lactation and breast milk No published information found as of the revision date. Protein binding 93% binding to albumin and α-1 acid glycoprotein (AAG) |
| References | |
| Additional Infomation |
Erlotinib hydrochloride is the hydrochloride salt of a quinazoline derivative with antitumor activity. Erlotinib competitively binds to the intracellular catalytic domain of the epidermal growth factor receptor (EGFR) tyrosine kinase, thereby reversibly inhibiting EGFR phosphorylation and blocking signal transduction events and tumorigenic effects associated with EGFR activation. It is a quinazoline derivative and an antitumor drug that acts as a protein kinase inhibitor of EGFR-associated tyrosine kinases. It is used to treat non-small cell lung cancer (NSCLC). See also: Erlotinib (containing the active ingredient). Drug Indications Non-small cell lung cancer (NSCLC): Tarceva may be considered for maintenance therapy in patients with locally advanced or metastatic NSCLC whose disease has stabilized after first-line chemotherapy and who carry EGFR activating mutations. Tarceva may also be considered for patients with locally advanced or metastatic NSCLC who have failed at least one prior chemotherapy regimen. For tumor patients without EGFR activating mutations, Tarceva may be considered when other treatment options are unavailable. Factors related to prolonged survival should be considered when prescribing Tarceva. For tumor patients negative for epidermal growth factor receptor (EGFR) immunohistochemistry (IHC), this treatment has not been proven to provide survival benefit or other clinically relevant efficacy. Pancreatic cancer: Tarceva in combination with gemcitabine is indicated for the treatment of patients with metastatic pancreatic cancer. Factors related to prolonged survival should be considered when prescribing Tarceva.
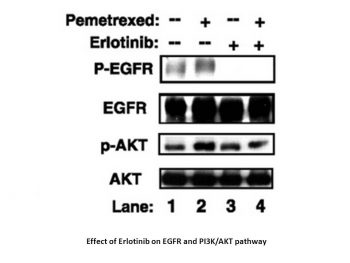
Epidermal growth factor receptor (EGFR) is overexpressed in a significant proportion of cancers and promotes the formation of malignant phenotypes. CP-358,774 is a direct inhibitor of human EGFR tyrosine kinase with an IC50 of 2 nM and reduces EGFR autophosphorylation in intact tumor cells with an IC50 of 20 nM. This inhibitory effect is selective for EGFR tyrosine kinases, superior to other tyrosine kinases we have tested, observed in both isolated kinases and intact cells. At a dose of 100 mg/kg, CP-358,774 completely inhibited EGF-induced autophosphorylation of EGFR in human HN5 tumors (xenografted into athymic mice) and in the livers of treated mice. In cell culture, CP-358,774 inhibited the proliferation of DiFi human colon cancer cells at submicromolar concentrations and arrested the cell cycle in the G1 phase. The inhibitor led to a significant accumulation of hypophosphorylated form of retinoblastoma protein (RP) and p27KIP1 in DiFi cells, which may be the cause of cell cycle arrest. In addition, EGFR inhibition induced apoptosis in these cells, which can be determined by DNA fragmentation and other indicators. These results suggest that CP-358,774 has the potential to treat tumors that depend on the EGFR pathway for proliferation or survival. [1] Objective: This study aimed to screen for the optimal combination therapy of erlotinib and pemetrexed for the treatment of relapsed NSCLC using a range of human non-small cell lung cancer (NSCLC) cell lines. Experimental Design: Human NSCLC cell lines expressing different known erlotinib sensitivity determinants were exposed to different dosing regimens of pemetrexed and erlotinib. Antitumor efficacy was assessed by cell counting and MTT assay to determine growth inhibition rate, flow cytometry to detect cell cycle distribution and apoptosis, and Western blotting to detect the expression of cell cycle mediators. Median effector analysis was used to determine the cytotoxic interaction (i.e., synergistic, additive, or antagonistic) between pemetrexed and erlotinib. Results: Synergistic cytotoxicity was observed in both erlotinib-sensitive and erlotinib-resistant human non-small cell lung cancer (NSCLC) cell lines when cells were co-exposed to pemetrexed and erlotinib, or treated with pemetrexed followed by erlotinib. This was independent of epidermal growth factor receptor or K-Ras gene mutation status. The synergistic effect was related to the effects of the two drugs on the cell cycle. Conversely, in erlotinib-sensitive cells, treatment with erlotinib followed by pemetrexed primarily exhibited an antagonistic effect, while in erlotinib-resistant cells, it was at most additive. The antagonistic effect was associated with erlotinib-induced G1 phase arrest in erlotinib-sensitive cells, which protected cells from the cytotoxic effects of pemetrexed. Pemetrexed induced activation of the epidermal growth factor receptor-mediated phosphatidylinositol 3-kinase/AKT pathway, which can be inhibited by erlotinib and the specific phosphatidylinositol 3-kinase inhibitor LY294002. Conclusion: In vitro, the combination of pemetrexed and erlotinib has a synergistic effect in non-small cell lung cancer, especially in erlotinib-sensitive tumors, if exposure to erlotinib is avoided before pemetrexed treatment. Based on these findings, a randomized phase II study has been initiated to compare progression-free survival in patients with recurrent non-small cell lung cancer (NSCLC) treated with intermittent erlotinib plus pemetrexed (experimental group) versus pemetrexed alone (control group). [2] Objective: This study aims to screen for the optimal erlotinib plus pemetrexed regimen for the treatment of recurrent NSCLC using a range of human NSCLC cell lines. Experimental design: Human NSCLC cell lines expressing different known erlotinib sensitivity molecular determinants were exposed to pemetrexed and erlotinib respectively using different dosing regimens. The antitumor effect was evaluated by measuring growth inhibition rate by cell counting and MTT assay, analyzing cell cycle distribution and apoptosis by flow cytometry, and detecting the expression of cell cycle mediators by immunoblotting. The cytotoxic interaction (i.e., synergistic, additive, or antagonistic effect) between pemetrexed and erlotinib was determined by median effect analysis. Results: A synergistic effect of cytotoxicity was observed in both erlotinib-sensitive and erlotinib-resistant human non-small cell lung cancer (NSCLC) cell lines when cells were simultaneously exposed to pemetrexed and erlotinib, or sequentially treated with pemetrexed followed by erlotinib. This effect was independent of the mutational status of the epidermal growth factor receptor (EGFR) or K-Ras gene. The synergistic effect was related to the effects of the two drugs on the cell cycle. Conversely, in erlotinib-sensitive cells, treatment with erlotinib followed by pemetrexed primarily showed an antagonistic effect; while in erlotinib-resistant cells, at most an additive effect was observed. This antagonistic effect was associated with erlotinib-induced G1 phase arrest in erlotinib-sensitive cells, thereby protecting cells from the cytotoxic effects of pemetrexed. Pemetrexed induces EGFR-mediated activation of the phosphatidylinositol 3-kinase/AKT pathway, which can be inhibited by erlotinib and the specific phosphatidylinositol 3-kinase inhibitor LY294002. Conclusion: In vitro, pemetrexed and erlotinib combination therapy for non-small cell lung cancer (NSCLC) has a synergistic effect if cells are not treated with erlotinib before pemetrexed treatment, especially in tumors sensitive to erlotinib. Based on these findings, a randomized phase II study has been initiated to compare progression-free survival in patients with recurrent NSCLC treated with intermittent combination of erlotinib and pemetrexed (experimental group) versus pemetrexed alone (control group). [3] Epidermal growth factor receptor (EGFR) is overexpressed in a variety of cancers. One of the important signaling pathways regulated by EGFR is the phosphatidylinositol 3-kinase (PI3K)-phosphatidylinositol-dependent kinase 1-Akt pathway. Activation of Akt can stimulate anti-apoptotic pathways and promote cell survival. Akt also regulates the mammalian target of rapamycin (mTOR)-S6K-S6 pathway, thereby controlling the cell response to growth factors and nutrients. Recent studies have shown that the sensitivity of non-small cell lung cancer (NSCLC) cell lines to EGFR inhibitors (such as erlotinib (Tarceva, OSI Pharmaceuticals)) depends on the inhibition of the phosphatidylinositol 3-kinase-phosphatidylinositol-dependent kinase 1-Akt-mTOR pathway. This pathway has multiple inputs, as its activity can be regulated by other receptors or upstream mutations. Therefore, EGFR inhibition alone may not be sufficient to effectively inhibit all tumor cells, highlighting the necessity of multi-point intervention. This study aimed to investigate whether the mTOR inhibitor rapamycin could enhance the sensitivity of different tissue types (NSCLC, pancreatic cancer, colon cancer, and breast cancer) to erlotinib. The results showed that erlotinib inhibited extracellular signal-regulated kinases (ERK), Akt, and S6 only in the cell lines most sensitive to erlotinib. Rapamycin completely inhibited S6 in all cell lines, but with the activation of Akt phosphorylation. However, combination therapy with erlotinib downregulated rapamycin-stimulated Akt activity. Therefore, in certain cell lines, only the combination of erlotinib and rapamycin can simultaneously inhibit S6 and Akt. This combination produced a synergistic effect on cell growth inhibition, an observation that was validated in vivo in xenograft models. These results suggest that the combination of rapamycin and erlotinib may have clinical value and can enhance the efficacy of erlotinib. [4] Our goal was to conduct preclinical pharmacokinetics, monotherapy, and combination antitumor activity assessments of athymic nude mouse models carrying non-small cell lung cancer (NSCLC) xenografts to investigate the pharmacokinetics, monotherapy, and combination antitumor activity of the epidermal growth factor receptor (HER1/EGFR) tyrosine kinase inhibitor erlotinib. Immunohistochemistry was used to determine the HER1/EGFR status of the non-small cell lung cancer (NSCLC) tumor model. Pharmacokinetic studies assessed plasma drug concentrations of erlotinib in tumor-bearing and tumor-free athymic nude mice. The maximum tolerated dose (MTD) of erlotinib with each chemotherapeutic agent was then studied. Subsequently, the efficacy of erlotinib monotherapy and its combination with these chemotherapeutic agents was evaluated in NSCLC xenograft models. Most animals in each study underwent complete necropsy to further assess antitumor or toxic effects. Erlotinib monotherapy dose-dependently inhibited tumor growth in the H460a tumor model, with the inhibitory effect correlated with circulating drug concentration. In both the H460a and A549 tumor models, each investigational drug demonstrated antitumor activity at the maximum tolerated dose (MTD) (erlotinib 100 mg/kg: tumor growth inhibition rates of 71% and 93%, respectively; gemcitabine 120 mg/kg: tumor growth inhibition rates of 93% and 75%, respectively; cisplatin 6 mg/kg: tumor growth inhibition rates of 81% and 88%, respectively). Tumor growth inhibition was less effective when the dose of each compound was below the MTD. To determine efficacy, we evaluated the efficacy of gemcitabine or cisplatin in combination with erlotinib at 25% of the MTD. In two non-small cell lung cancer (NSCLC) models, gemcitabine (30 mg/kg) or cisplatin (1.5 mg/kg) combined with erlotinib (25 mg/kg) was well tolerated at 25% of the MTD. For slow-growing A549 tumors, the combination of gemcitabine/erlotinib and cisplatin/erlotinib significantly inhibited tumor growth (inhibition rates were higher than 100% and 98%, respectively), with some tumor regression. For faster-growing H460a tumors, the above combination also significantly inhibited tumor growth, but the inhibitory effect was not as significant as that of A549 tumors (inhibition rates were 86% and 53%, respectively). These results indicate that erlotinib has significant antitumor activity as a monotherapy or in combination with chemotherapy in non-small cell lung cancer xenografts with similar EGFR expression levels. [5] Epidermal growth factor receptor (EGFR) is overexpressed in a variety of cancers. One of the key signaling pathways regulated by EGFR is the phosphatidylinositol 3-kinase (PI3K)-phosphatidylinositol-dependent kinase 1-Akt pathway. Activation of Akt stimulates anti-apoptotic pathways, promoting cell survival. Akt also regulates the mammalian target of rapamycin (mTOR)-S6K-S6 pathway, thereby controlling cellular responses to growth factors and nutrients. Recent studies have shown that the sensitivity of non-small cell lung cancer cell lines to EGFR inhibitors (such as erlotinib (Tarceva, OSI Pharmaceuticals)) depends on the inhibition of the phosphatidylinositol 3-kinase-phosphatidylinositol-dependent kinase 1-Akt-mTOR pathway. This pathway may have multiple inputs, as its activity can be regulated by other receptors or upstream mutations. Therefore, inhibiting EGFR alone may not be sufficient to effectively suppress all tumor cells, highlighting the necessity of multi-point intervention. This article aims to investigate whether the mTOR inhibitor rapamycin can enhance the sensitivity of cell lines derived from different tissue types (non-small cell lung cancer, pancreatic cancer, colon cancer, and breast cancer) to erlotinib. Erlotinib inhibits extracellular signal-regulated kinases, Akt, and S6 only in the most sensitive cell lines. Rapamycin completely inhibits S6 in all cell lines, but with the activation of Akt phosphorylation. However, rapamycin-stimulated Akt activity is downregulated when used in combination with erlotinib. Therefore, in certain cell lines, only the combination of erlotinib and rapamycin can simultaneously inhibit S6 and Akt. This combination produces a synergistic effect on cell growth inhibition, an observation that has been validated in vivo in xenograft models. These results suggest that the combination of rapamycin and erlotinib may have clinical value and can enhance the efficacy of erlotinib. [6] Background: Epidermal growth factor receptor (EGFR) and mammalian target of rapamycin (mTOR) are key targets for cancer therapy. In vitro and in vivo experiments have shown a synergistic effect of combined inhibition of these two targets in various cancer types. However, the roles of EGFR and mTOR expression and their combined inhibition in other neuroendocrine lung tumors besides small cell lung cancer remain unclear. Materials and Methods: Immunohistochemistry was used to detect the expression and activation of EGFR/AKT/mTOR pathway components in 110 typical and atypical bronchial carcinoid (AC) and large cell neuroendocrine lung cancer (LCNEC) tumor samples, and their correlation with clinicopathological parameters and patient survival was analyzed. Growth inhibition assays were used to evaluate the cytotoxicity of the mTOR inhibitor everolimus and the EGFR inhibitor erlotinib, alone and in combination, on NCI-H720 AC and SHP-77 LCNEC cells. Flow cytometry (FACS) was used to determine cell cycle distribution. The activation of apoptosis-related caspase-3/7 was detected using the Caspase-Glo® kit. Western blotting was used to analyze the activity status of EGFR and mTOR pathway components. Results: EGFR/AKT/mTOR axis activation was detected in all tumor types, and significantly enhanced in high-grade tumors. Neoadjuvant chemotherapy was significantly associated with p-AKT expression and p-ERK loss. Erlotinib combined with everolimus exerted a synergistic effect in AC and LCNEC cells by inducing apoptosis, without affecting cell cycle distribution. These effects can be explained by the synergistic downregulation of phosphorylated mTOR, phosphorylated p70S6 kinase and phosphorylated AKT expression by everolimus and erlotinib. Conclusion: Our study shows that EGFR and mTOR are clinically significant targets in bronchial neuroendocrine tumors, and further in vivo and clinical studies on combined inhibition are necessary. [7] |
| Molecular Formula |
C22H23N3O4.HCL
|
|---|---|
| Molecular Weight |
429.90
|
| Exact Mass |
429.145
|
| Elemental Analysis |
C, 61.47; H, 5.63; Cl, 8.25; N, 9.77; O, 14.89
|
| CAS # |
183319-69-9
|
| Related CAS # |
Erlotinib-d6 hydrochloride;1189953-78-3;Erlotinib;183321-74-6;Erlotinib mesylate;248594-19-6;Erlotinib-13C6 hydrochloride;1210610-07-3;Erlotinib-d6;1034651-23-4
|
| PubChem CID |
176871
|
| Appearance |
White to off-white solid powder
|
| Boiling Point |
553.6ºC at 760 mmHg
|
| Melting Point |
223-225ºC
|
| Flash Point |
288.6ºC
|
| Vapour Pressure |
4.52E-12mmHg at 25°C
|
| LogP |
4.28
|
| Hydrogen Bond Donor Count |
2
|
| Hydrogen Bond Acceptor Count |
7
|
| Rotatable Bond Count |
11
|
| Heavy Atom Count |
30
|
| Complexity |
525
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
Cl[H].O(C([H])([H])C([H])([H])OC([H])([H])[H])C1C([H])=C2C(=NC([H])=NC2=C([H])C=1OC([H])([H])C([H])([H])OC([H])([H])[H])N([H])C1=C([H])C([H])=C([H])C(C#C[H])=C1[H]
|
| InChi Key |
GTTBEUCJPZQMDZ-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C22H23N3O4.ClH/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22;/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25);1H
|
| Chemical Name |
N-(3-ethynylphenyl)-6,7-bis(2-methoxyethoxy)quinazolin-4-amine;hydrochloride
|
| Synonyms |
NSC718781 HCl; NSC-718781 HCl; CP358774 HCl, NSC 718781 HCl; erlotinib HCl; Tarceva; N-(3-ethynylphenyl)-6,7-bis(2-methoxyethoxy)quinazolin-4-amine hydrochloride; OSI-774; OSI 774; Erlotinib (Hydrochloride); CP-358774 HCl; CP 358774 HCl; OSI-774 HCl; OSI 774 HCl; OSI774 HCl; Erlotinib hydrochloride
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 0.5 mg/mL (1.16 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 0.5 mg/mL (1.16 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: 0.5 mg/mL (1.16 mM) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. Solubility in Formulation 4: 15% Captisol: 15 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3261 mL | 11.6306 mL | 23.2612 mL | |
| 5 mM | 0.4652 mL | 2.3261 mL | 4.6522 mL | |
| 10 mM | 0.2326 mL | 1.1631 mL | 2.3261 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
GDC-0449 and Erlotinib Hydrochloride With or Without Gemcitabine Hydrochloride in Treating Patients With Metastatic Pancreatic Cancer or Solid Tumors That Cannot Be Removed by Surgery
CTID: NCT00878163
Phase: Phase 1 Status: Active, not recruiting
Date: 2024-09-19
|
 |
 |