
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g | |||
| Other Sizes |
Purity: ≥98%
Elacridar (formerly GW-120918; GF-120918; GG-918; GW-0918; GF120918; GF120918; GW0918; GG918) is a 3rd generationl and potent P-glycoprotein (P-gp or MDR-1) and BCRP inhibitor that has the potential to overcome P-gp-mediated multidrug resistance (MDR) of the cytotoxic drugs such as doxorubicin. Elacridar has been used both in vitro and in vivo as a tool inhibitor of P-gp to investigate the role of transporters in the disposition of various test molecules. In vitro, GF120918A demonstrated high plasma protein binding across species, although a definitive protein binding evaluation was precluded by poor recovery, particularly in buffer and in mouse, rat, and dog plasma.
| Targets |
P-glycoprotein (Pgp); breast cancer resistance protein (BCRP)
|
|---|---|
| ln Vitro |
Elacridar (0.001-1 μM; 2 h) reduces the viability of 786-O cells [2]. Elacridar (5 μM; 24 h) modulates P-glycoprotein and ABCG2 protein expression levels in MCF-7 and 786-O cell lines [2]. Elacridar (5 μM; 24 h) impacts 99mTc-MIBI intracellular accumulation in MCF-7 and 786-O cells [2].
Researchers hypothesized that inhibition of multidrug resistant transporters by Elacridar (dual inhibitor of P-glycoprotein and ABCG 2) might overcome sunitinib resistance in experimental renal cell carcinoma. Human renal carcinoma cell lines 786-O, ACHN, and Caki-1 were treated with sunitinib or elacridar alone, or in combination. It was showed that Elacridar significantly enhanced sunitinib cytotoxicity in 786-O cells. P-glycoprotein activity, confirmed by P-glycoprotein function assay, was found to be inhibited by elacridar. ABCG2 expression was low in all renal carcinoma cell lines, and was suppressed only by combination treatment in 786-O cells. ABCG2 function was inhibited by sunitinib alone or combination with elacridar but not elacridar alone. These findings suggest that sunitinib resistance involves multidrug resistance transporters, and in combination with elacridar, can be reversed in renal carcinoma cells by P-glycoprotein inhibition. Elacridar sensitizes 786-O cells to sunitinib-induced cytotoxicity [2] In this assay, we evaluated the efficacy of elacridar in reversing multidrug resistance. Combined treatment of Elacridar and sunitinib decreased cell viability of 786-O and ACHN significantly compared to each single treatment, and it was elacridar concentration dependent manner (Fig. 1). These effects were not observed in MCF-7 or Caki-1 cells under the same treatments, including those given in combination. These results indicate that elacridar is capable of reversing P-glycoprotein and/or ABCG2-mediated multidrug resistance associated with the clinical anticancer agent sunitinib. Detection of P-glycoprotein and ABCG2 by western blotting [2] Western blot analysis showed a differential expression pattern of P-glycoprotein and ABCG2 proteins among cells. As shown in Fig. 3, the result was consistent with our speculation that the expression of P-glycoprotein was higher in 786-O cells than in other cell lines, although it was not significantly different. In addition, P-glycoprotein expression increased 2-fold in 786-O cells under hypoxia. In Caki-1 and MCF-7 cells, its expression remained stable. ABCG2 protein was relatively low in both RCC cells (Caki-1 and 786-O) compared to MCF-7, it was significantly different under hypoxic condition. Moreover, the effect of sunitinib and elacridar on the expression of P-glycoprotein (Fig. 4A) and ABCG2 (Fig. 4B) was investigated. Both drugs were tested at a concentration of 5 µM as a single treatment, or in combination (each 5 µM). As shown in Fig. 5, the combination of sunitinib and elacridar led to a significant reduction in ABCG2 expression in 786-O, suggesting that elacridar was able to inhibit the overexpression of this protein stimulated by sunitinib, while P-glycoprotein expression was not altered. In vitro imaging of multidrug resistance P-glycoprotein transport function using 99m Tc-MIBI [2] To investigate the potential mechanisms by which Elacridar sensitizes 786-O cells to sunitinib, we used a substrate of P-glycoprotein, 99mTc-MIBI as a radiotracer to visualize the expression of functional P-glycoprotein. MCF-7 and 786-O cells were treated with predetermined concentrations of sunitinib (5 µM) or elacridar (5 µM) alone, or in combination (each 5 µM), for 24 h. Cells were treated with 99mTc-MIBI, and gamma radiation was counted to determine the accumulation of 99mTc-MIBI in each cell line. As a result, intact MCF-7 cells showed higher accumulation of 99mTc-MIBI, while 786-O cells showed almost negligible amounts of the radiotracer (Fig. 5A). These results suggest that 786-O cells are better at extracting P-glycoprotein, which is consistent with the results from western blot analysis which showed that 786-O cells had higher expression of this transporter compared to MCF-7 cells (Fig. 3). As predicted, we noticed a dose-dependent increase in the intracellular accumulation of 99mTc-MIBI in 786-O cells following exposure to Elacridar as a single treatment or in combination with sunitinib (Fig. 5B). Single treatment of sunitinib resulted in a slight increase in the level of intracellular 99mTc-MIBI in 786-O cells, although this effect was less pronounced than that following elacridar treatment. The increase in 99mTc-MIBI accumulation over 60 min was time dependent. We observed an approximate 7-fold difference in the accumulation of 99mTc-MIBI between naïve 786-O cells and in combination-treated cells at 60 min. As predicted, the treatment of MCF-7 cells with elacridar had no effect on the intracellular 99mTc-MIBI accumulation. Neither sunitinib nor elacridar altered the intracellular levels of 99mTc-MIBI in Caki-1 cells, where P-glycoprotein expression was relatively low compared with that in 786-O cells (data not shown). Flow cytometry-based approach for detection of expression and function of ABCG2 transporter [2] In order to assess the functionality of the ABCG2 transporter in 786-O cells, and to evaluate the potency of sunitinib and Elacridar as an in vitro inhibitor of this transporter, we used Ph A, a chlorophyll catabolite, which is reported to be a specific probe to measure ABCG2 function (Robey et al., 2004). Flow cytometry analysis revealed that treating cells with 25 µM elacridar increased the accumulation of Ph A in 786-O cells around 5-fold. Moreover, at a dose of 5 µM, sunitinib led to the Ph A accumulation of 10 times more than in untreated controls (Fig. 6). The combination treatment seemed to further increase the accumulation compared with elacridar single treated cells, although this was almost the same level as that observed in sunitinib single treated cells. The assay was also performed in MCF-7, and exposure to sunitinib treatment alone increased the accumulation of Ph A 12-fold. ACHN cells treated with sunitinib accumulated up to 14.5 times more Ph A than the controls (data not shown). These results suggest that sunitinib causes a dose-dependent increase in the intracellular Ph A levels in RCC cells. In this case, elacridar was less active than sunitinib, although it caused a more potent inhibition of P-glycoprotein function. |
| ln Vivo |
Elacridar (100 mg/kg; intraperitoneally) distributes differently in the brain and plasma[1]. 1.19 Elacridar Plasma Pharmacokinetic Parameters in Mice[1]. AUC0-inf (μg·min/ml) 1460 90.3 161.4 F 0.22 0.013 1 Mice PO 100 mg/kg Mice IP 100 mg/kg Mice IV 2.5 mg/ kg CL/F (ml/min) 2.05 33.2 0.46 Vd/F (liter) 3.5 12.3 0.17 t1/2 (h) 20 4.3 4.4.
Intravenous Administration of Elacridar. [1] The disposition of elacridar in plasma and brain was studied in FVB wild-type mice after an intravenous injection. The plasma concentrations showed a bi-exponential decline indicating distinct distribution and elimination phases (Fig. 1A). Concentrations rapidly reached peak levels in brain within 0.5 h after intravenous dosing with the maximal concentration (Cmax) observed at 0.5 h, the first measured time point. The brain-to-plasma concentration ratio was high at the initial time points (up to 2 h) and decreased thereafter as concentrations in brain declined more rapidly compared with those in plasma (Fig. 1B). This is consistent with the observed terminal t1/2 of 4.4 h in plasma and 1.5 h in brain (Table 1). The total plasma clearance was estimated to be 0.46 ml/min. The blood flow to the liver for a 20-g mouse is 1.8 ml/min (Davies and Morris, 1993), meaning elacridar has at most low to moderate hepatic extraction in the mouse. The AUC from time zero to infinity (AUC0-inf) was 161 μg · min/ml in plasma and 131 μg · min/ml in brain. The resulting brain Kp ratio was 0.82, indicating that after intravenous dosing at 2.5 mg/kg, there is approximately equal partitioning of elacridar into the brain as plasma. Intraperitoneal Administration of Elacridar. [1] The plasma and brain concentrations after an intraperitoneal dose of 100 mg/kg were measured in FVB wild-type mice. After intraperitoneal dosing, brain concentrations were significantly lower than plasma concentrations at all measured time points, except at 4 h after the dose (Fig. 2A). The corresponding brain-to-plasma concentration ratios remained less than one at all measured time points (Fig. 2B). It is important to note that the plasma concentrations after intravenous administration at which we observe a greater than one brain-to-plasma concentration ratio are higher than the maximal plasma concentrations seen after intraperitoneal dosing. The brain distribution ratio (i.e., the partition coefficient) of elacridar is likely to be dependent on its plasma levels. The observed Cmax in plasma after the intraperitoneal dose was 0.295 ± 0.06 μg/ml, and that in brain was 0.061 ± 0.024 μg/ml. The apparent plasma clearance (Cl/F) was estimated to be 33 ml/min by noncompartmental analysis. The AUC0-inf in plasma was 90.3 and 43.5 μg · min/ml in the brain. The elimination-phase t1/2 of elacridar after noncompartmental analysis was estimated to be 4.3 in plasma and 9.2 h in brain. The Kp ratio was 0.48, indicating that the partitioning into the brain was lower after intraperitoneal administration, even at a dose 40 times higher, compared with intravenous administration. Oral Administration of Elacridar. [1] The brain and plasma pharmacokinetics of elacridar were studied in FVB wild-type mice after a 100-mg/kg dose administered orally. The brain concentrations were lower than plasma concentrations until 1 h after the dose, after which the brain concentrations were several-fold higher than plasma concentrations (Fig. 3A). The brain-to-plasma ratio after oral administration was less than one for initial time points and then increased, reaching a maximum of approximately 6 at 4 h after the dose, before showing a slow decline (Fig. 3B). Noncompartmental analysis of plasma and brain concentration-time data showed that the plasma AUC0-inf was 1460 μg · min/ml and the brain AUC0-inf was 6296 μg · min/ml (Table 1). The Cmax in brain after oral dosing was 4.34 ± 0.79 μg/ml, significantly higher than that seen after intraperitoneal dosing. The time to reach Cmax in plasma was 4 h, indicating slow dissolution and absorption from the gut. After oral dosing, the brain Kp was found to be 4.31, suggesting that a high dose of elacridar administered orally gives high distribution in the brain. The t1/2 of the drug in the brain after the oral dose mirrored its plasma t1/2, with the values being 19.8 and 15.6 h, respectively. Determination of Bioavailability after Intraperitoneal and Oral Administration. [1] Absolute bioavailability was determined as a ratio of dose-normalized AUC after intraperitoneal or oral administration to dose-normalized AUC after intravenous administration (Table 2). The bioavailability after intraperitoneal administration was 1.3%, and that after oral administration was 22% with the suspension. The bioavailability after intraperitoneal dosing was very poor, indicating that intraperitoneal may not be a favorable route of administration to get reproducible plasma and brain exposure of elacridar using this simple suspension formulation. The bioavailability of elacridar after oral administration was higher than that after an equivalent dose administered intraperitoneally; this could be due to enhanced dissolution in the gut through greater solvent availability or micellar effects of bile salts. The t1/2 of elacridar after oral administration was approximately 5 times longer than that after intravenous or intraperitoneal administration. There is a possibility that there is nonlinearity in the absorption, distribution, and possibly elimination of elacridar. However, the bioavailability has been calculated with the assumption that there is no change in the clearance of the drug with increases in plasma exposure of the drug. |
| Enzyme Assay |
Analysis of Elacridar by Liquid Chromatography-Tandem Mass Spectrometry.
The concentrations of Elacridar in mouse plasma and brain were determined by high-performance liquid chromatography coupled with mass spectrometry. Frozen brain samples were thawed and homogenized with 3 volumes of 5% bovine serum albumin using a tissue homogenizer. Fifty microliters of plasma and 100 μl of brain homogenate were spiked with 20 ng of internal standard tyrphostin (AG 1478) and 100 μl of a buffer, pH 11 (0.1 M sodium hydroxide and 0.04 M sodium bicarbonate). Samples were extracted by vigorously vortexing with 1 ml of ethyl acetate for 5 min and then centrifugation at 7500 rpm for 15 min at 4°C. Six-hundred microliters of organic layer was transferred to microcentrifuge tubes and dried by a gentle stream of nitrogen. Samples were reconstituted in 100 μl of mobile phase and were transferred to autosampler vials. A 5-μl volume was injected using a temperature-controlled autosampler maintained at 10°C. Chromatographic analysis was performed using an Agilent Technologies Eclipse XDB-C18 RRHT threaded column (4.6 mm i.d. × 12.5 mm, 5 μ). The mobile phase was composed of acetonitrile: 20 mM ammonium formate (with 0.1% formic acid) (42:58 v/v) with a flow rate of 0.25 ml/min. The eluent was monitored using a Thermo Finnigan TSQ Quantum 1.5 detector. The instrument was equipped with an electrospray interface. The samples were ionized by the electrospray probe and analyzed in the positive ionization mode operating at a spray voltage of 4500 V for both and the internal standard. The spectrometer was programmed to allow the [MH+] ion of elacridar at m/z 564.6 and that of the internal standard at m/z 316.67 to pass through the first quadrupole (Q1) and into the collision cells (Q2). The collision energy was set at 39 V for elacridar and 9 V for tyrphostin. The product ions for elacridar (m/z 252.9) and the internal standard (m/z 300.9) were monitored through quadrupole 3 (Q3). The scan width and scan time for monitoring the two product ions were m/z 1.5 and 0.5 s, respectively. The assay was precise and linear over a range of 2.5 to 1500 ng/ml (CV was less than 10% over all concentrations).
Technetium-99m-labeled methoxyisobutyl isonitrile (99mTc-MIBI) in vitro uptake studies [2] 99mTc-MIBI was used as a transport substrate for P-glycoprotein (Piwnica-Worms et al., 1993). Na99mTcO4 was obtained in physiological saline as commercial 99Mo/99mTc generator eluate, and 99mTc-MIBI was prepared by adding Na99mTcO4 into a MIBI-kit vial and heating the mixture at 95 °C for 15 min. The radiopharmaceutical was diluted to 10 MBq/ml with saline. Cells were seeded into 100-mm culture dishes at a density of 2.0×106 per dish. After 24 h incubation, the medium was aspirated and replaced with 10 ml of culture medium with 5% FBS containing the appropriate concentrations of drug. After further 24 h incubation, supernatants were thoroughly aspirated, and single cell suspensions of 5.0×105 cells/ml were prepared. The time course of 99mTc-MIBI accumulation into MCF-7, Caki-1, ACHN, and 786-O cells was studied using single cell suspensions at 5.0×105 cells/ml incubated at 37 °C. Aliquots of 80 µl 99mTc-MIBI were added to vials containing 5 ml aliquots of cell suspension, and the cells were incubated at 37 °C for 1, 15, 30, 60, and 90 min after addition of the tracer. Duplicate samples of 400 µl were removed from each vial and transferred to 1.5 ml microcentrifuge tubes containing 500 µL ice cold saline and then centrifuged at 14,000g for 2 min. The supernatant was aspirated, and the cell pellet was carefully resuspended in 900 µl ice-cold saline and assayed for radioactivity using a gamma cell counter. Dose response curves were generated for sunitinib and Elacridar, and then the radioactivity was measured using a calibrated gamma counter (Ballinger et al., 1996). From the measurement of radioactivity in the cell pellet, the accumulation of radioactivity inside the cell, relative to that outside, (Cin/Cout) was determined. Flow cytometry to measure Ph A uptake [2] Expression of the ABCG2 transporter was determined by flow cytometry using the fluorescent compound pheophorbine A (Ph A), which was identified as an ABCG2 substrate based on Amcg2−/− knockout studies in mice. Cells were trypsinized and 3.0×105 cells were incubated in complete medium (phenol red-free DMEM with 10% FBS) with 10 µM Ph A with the desired concentration of sunitinib or Elacridar for 30 min at 37 °C in 5% CO2. The cells were then washed with cold complete medium, followed by incubation for 1 h at 37 °C in Ph A free complete medium. The cells were subsequently washed with cold complete medium and then incubated for 1 h at 37 °C in Ph A free complete medium. Finally, cells were washed twice with cold PBS and fluorescence was determined by flow cytometry. A total of 2.0×104 events were collected, and debris were eliminated by gating the forward versus side scatter. Intracellular fluorescence was analyzed with an excitation wavelength of 480 nm and an emission wavelength of 530 nm. |
| Cell Assay |
Cell Viability Assay[2]
Cell Types: 786-O cells Tested Concentrations: 2.5 and 5 μM Incubation Duration: 2 hrs (hours) Experimental Results: Dose-dependently inhibited cell viability of 786-O cells and demonstrated better inhibitory effect with sunitnib adding Western Blot Analysis[2] Cell Types: MCF-7, Caki-1, and 786-O cell lines Tested Concentrations: 5 μM Incubation Duration: 24 hrs (hours) Experimental Results: Dreased P-glycoprotein protein expression level in 786-O cells and increased ABCG2 protein expression level in Caki-1 cells. Cell Viability Assay[2] Cell Types: MCF-7 and 786-O cell lines Tested Concentrations: 5 μM Incubation Duration: 24 hrs (hours) Experimental Results: Dose-dependently increased 99mTc-MIBI intracellular accumulation in MCF-7 and 786-O cells. Cytotoxicity assay (MTT assay) [2] To examine the combination effect of sunitinib and Elacridar against cell proliferation, 3.0×103 cells per well were seeded in a 96-well plate. After 24 h incubation, an optimum concentration gradient of sunitinib, Elacridar, or each combination was added to each well, followed by culturing for 48 h. Finally, cell viability was assessed using the proliferation reagent, MTT, according to the manufacturer׳s instructions. Control cells were treated with the vehicle only, 0.1% DMSO. After this final incubation, the medium was aspirated and precipitated formazan crystals were dissolved in DMSO (100 μL/well). The absorbance of each well was measured at 540 nm, and a reference wavelength of 650 nm was read with a multiskan JX microplate reader. Cell viability was calculated as percentage of the control value. Western blotting analysis [2] Expressions of multi-drug resistant proteins were examined by immuno-blotting. Cells were plated at a density of 2.5×105 cells per 60 mm dish and allowed to adhere for 24 h. The cells were then incubated with either sunitinib or Elacridar alone or in combination, in 5% FBS medium for further 24 h. Basal expressions in each cells were analyzed under normoxic or hypoxic conditions, while drug treated cells were analyzed under normoxic condition. Cells were lysed in ice cold buffer containing 0.5 M Tris–HCl, 100 mM β-glycerophosphate, 3 M NaCl, 50 mM EDTA, 100 mM Na3VO4, and 10% Triton-X, the lysates were cleared by centrifugation at 4 °C for 20 min at 12,000g, and protein concentrations were determined using the DC protein assay kit with BSA used as a standard. Protein lysates were loaded at concentrations of 25–30 µg for the ABC Sub-family Member 2, or 5060 µg for P-glycoprotein per lane and were resolved by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) before being electrophoretically transferred onto polyvinylidene fluoride (PVDF) membranes by tank transfer. Cell lysates analyzed for P-glycoprotein were resolved by 7.5% SDS-PAGE, while cell lysates analyzed for ABCG2 were resolved by 10% SDS-PAGE. Non-specific antibody binding was blocked by incubating the membranes in 5% skimmed milk. Membranes were then sequentially probed with primary monoclonal antibody (P-glycoprotein, mouse antibody: 1 h at room temperature at a 1:500 dilution in 1% milk, ABCG2, rabbit antibody) overnight at 4 °C at 1:1000 dilution in 1% milk. Subsequently, the membranes were washed with tris-buffered saline with Tween 20 (TBS-T) and were then incubated with horse-radish peroxidase (HRP)-conjugated secondary antibody (mouse secondary antibody: 1 h at room temperature at 1:1000 dilution in 1% milk, rabbit secondary antibody: 1 h at room temperature at 1:2000 dilution in 1% milk). |
| Animal Protocol |
Animal/Disease Models: FVB wild-type mice[1].
Doses: 100 mg/kg Route of Administration: intraperitoneal (ip)injection; 100 mg/kg once Experimental Results: Showd a higher concertation in brain than plasma except at 4 h after the dose. Intravenous Administration of Elacridar. [1] The elacridar intravenous dosing solution was prepared on the day of the experiment by dissolving elacridar in a vehicle containing dimethyl sulfoxide, propylene glycol, and saline, 2:2:1 (v/v/v), at a concentration of 1.25 mg/ml. FVB wild-type mice were given intravenous doses of 2.5 mg/kg (2 μl volume/g b.wt.) in the tail vein. Blood and brain were collected at 0.5, 1, 2, 4, and 8 h after the dose (n = 4 at each time point). Animals were euthanized by use of a carbon dioxide chamber. Blood was collected by cardiac puncture, and plasma was obtained by centrifugation at 7500 rpm for 10 min at 4°C. The whole brain was quickly removed from skull and rinsed with ice-cold saline. Brains were immediately flash-frozen with liquid nitrogen. The specimens were stored at −80°C until analysis by liquid chromatography-tandem mass spectrometry. Intraperitoneal and Oral Administration of Elacridar. [1] Elacridar for intraperitoneal and oral dosing was prepared on the day of the experiment by preparing a stable suspension of elacridar, using 0.5% hydroxypropylmethylcellulose and 1% Tween 80 to obtain a 10-mg/ml formulation. Mice received an intraperitoneal dose of 100 mg/kg by injection into the peritoneal cavity. For oral administration, mice received a dose of 100 mg/kg by oral gavage. Blood and brain were sampled at 15 min, 0.5, 1, 2, 4, and 8 h after intraperitoneal dosing and at 0.5, 1, 2, 4, 8, 17, and 24 h after oral dosing. Plasma and brain samples were collected and processed in the manner described in the previous section. |
| ADME/Pharmacokinetics |
This study aimed to determine the bioavailability and distribution of elacridar (GF120918; N-(4-(2-(1,2,3,4-tetrahydro-6,7-dimethoxy-2-isoquinolinyl)ethyl)phenyl)-9,10-dihydro-5-methoxy-9-oxo-4-acridinecarboxamide) in plasma and brain tissue of mice after different routes of administration. Elacridar is a potent P-glycoprotein and breast cancer resistance protein inhibitor and has been used to study the effects of these efflux transporters on drug distribution in the brain. In this study, Friend leukemia virus B strain mice were administered 100 mg/kg of elacridar orally or intraperitoneally. The absolute bioavailability of elacridar after oral or intraperitoneal administration was determined with an intravenous dose of 2.5 mg/kg as a reference. At these doses, the absolute bioavailability of oral administration was 0.22, and the absolute bioavailability of intraperitoneal administration was 0.01. The terminal half-life of iracodyl was approximately 4 hours after intraperitoneal and intravenous injection, and nearly 20 hours after oral administration. The brain plasma partition coefficient (Kp,brain) of iracodyl increased with increasing plasma exposure, suggesting that the blood-brain barrier efflux transporter protein had reached saturation. The Kp,brain values after intravenous, intraperitoneal, and oral administration were 0.82, 0.43, and 4.31, respectively. Iracodone has low water solubility and high lipophilicity, leading to poor oral absorption, which is likely limited by the dissolution rate. These results indicate that the route of administration and the resulting plasma exposure are crucial for achieving effective plasma and brain tissue drug concentrations and can serve as a guide for future iracodyl administration studies and the development of formulation strategies to overcome its poor absorption. [1]
Intravenous administration of iracodyl. [1] In this study, the distribution of iracodyl in plasma and brain tissue after intravenous injection was investigated in FVB wild-type mice. Plasma concentrations showed a bi-exponential decrease, indicating the presence of distinct distribution and elimination phases (Figure 1A). Within 0.5 hours of intravenous administration, the drug concentration in brain tissue rapidly reached its peak, with the maximum concentration (Cmax) occurring at 0.5 hours, the time point of the first measurement. At the initial time point (within 2 hours), the concentration ratio in brain tissue to plasma was high, subsequently decreasing as the drug concentration in brain tissue decreased faster than in plasma (Figure 1B). This is consistent with the observed terminal half-life (t1/2) of 4.4 hours in plasma and 1.5 hours in brain tissue (Table 1). The total plasma clearance was estimated at 0.46 ml/min. The hepatic blood flow in a 20-gram mouse was 1.8 ml/min (Davies and Morris, 1993), implying that the liver extraction rate of elacridar in mice was at most low to moderate. The AUC from zero to infinity (AUC0-inf) in plasma was 161 μg·min/ml, and in brain tissue it was 131 μg·min/ml. The resulting brain tissue Kp ratio of 0.82 indicates that after intravenous administration of 2.5 mg/kg, elacridar was distributed approximately equally in brain tissue and plasma. Intraperitoneal administration of elacridar. [1] In FVB wild-type mice, plasma and brain tissue concentrations were determined after intraperitoneal administration of a dose of 100 mg/kg. After intraperitoneal administration, brain tissue concentrations were significantly lower than plasma concentrations at all time points except 4 hours after administration (Fig. 2A). The brain tissue to plasma concentration ratio was less than 1 at all time points (Fig. 2B). Notably, plasma concentrations with a brain tissue to plasma concentration ratio greater than 1 were observed after intravenous administration, which were higher than the maximum plasma concentrations observed after intraperitoneal administration. The brain tissue distribution ratio (i.e., partition coefficient) of elacridar may depend on its plasma concentration. The Cmax observed in plasma after intraperitoneal administration was 0.295 ± 0.06 μg/ml, and the Cmax observed in brain tissue was 0.061 ± 0.024 μg/ml. The apparent plasma clearance (Cl/F) was estimated to be 33 ml/min using a non-compartmental model analysis. The AUC0-inf in plasma was 90.3 μg·min/ml, and the AUC0-inf in brain tissue was 43.5 μg·min/ml. The elimination phase t1/2 of elacridar was estimated to be 4.3 hours in plasma and 9.2 hours in brain tissue using a non-compartmental model analysis. The Kp ratio was 0.48, indicating that even at a dose 40 times higher than that of intravenous administration, the distribution of the drug in brain tissue was low after intraperitoneal administration. Oral administration of Elacridar. [1] The pharmacokinetics of brain and plasma after oral administration of 100 mg/kg elacridar were studied in FVB wild-type mice. Brain tissue concentrations were lower than plasma concentrations within 1 hour after administration, and then brain tissue concentrations were several times higher than plasma concentrations (Figure 3A). Following oral administration, the brain-plasma ratio was less than 1 at the initial time point, then gradually increased, reaching a peak of approximately 6 at 4 hours post-administration, before slowly declining (Figure 3B). Non-compartmental model analysis of plasma and brain tissue concentration-time data showed a plasma AUC0-inf of 1460 μg·min/ml and a brain tissue AUC0-inf of 6296 μg·min/ml (Table 1). The brain tissue Cmax after oral administration was 4.34 ± 0.79 μg/ml, significantly higher than that after intraperitoneal injection. The time to reach plasma Cmax was 4 hours, indicating slow drug dissolution and absorption in the intestine. The brain tissue Kp was 4.31 after oral administration, suggesting that high-dose oral administration of ileclopramide can lead to its widespread distribution in brain tissue. Following oral administration, the brain half-life (t1/2) of the drug was similar to its plasma half-life (t1/2), being 19.8 hours and 15.6 hours, respectively. Determination of bioavailability after intraperitoneal and oral administration. [1] Absolute bioavailability was defined as the ratio of the dose-normalized AUC after intraperitoneal or oral administration to the dose-normalized AUC after intravenous administration (Table 2). The bioavailability after intraperitoneal administration was 1.3%, and the bioavailability after oral suspension was 22%. The extremely low bioavailability after intraperitoneal administration suggests that intraperitoneal administration may not be the ideal route of administration for obtaining reproducible plasma and brain exposures with this simple suspension formulation. The bioavailability of ilexa was higher after oral administration than after an equivalent dose of intraperitoneal administration; this may be due to increased solvent availability in the intestine or the micellar effect of bile acids promoting drug dissolution. The half-life after oral administration of ilexa was approximately 5 times that after intravenous or intraperitoneal administration. The absorption, distribution, and possible elimination of ilexa may be nonlinear. However, the calculation of bioavailability is based on the assumption that drug clearance does not change with increasing plasma drug exposure. |
| References |
|
| Additional Infomation |
Elacridar (GW120918) is an oral bioenhancer targeting multidrug resistance in tumors. In many cases, multidrug resistance in cancer arises due to alterations in the expression of protein inhibitors. As of August 2007, Elacda was not listed in GlaxoSmithKline's (GSK) product pipeline. It is presumed that its development was discontinued. Drug Indications: For the treatment of solid tumors. Mechanism of Action: P-glycoprotein is a well-characterized human ABC transporter in the MDR/TAP subfamily. It is an ATP-dependent efflux pump with broad substrate specificity. It likely evolved as a mechanism for defending against harmful substances. Increased P-glycoprotein expression in the gut reduces the absorption of P-glycoprotein substrate drugs. Therefore, bioavailability is reduced, failing to achieve the required therapeutic plasma concentrations. Elacda works by inhibiting P-glycoprotein, thereby improving the bioavailability of combination therapies.
Pharmacodynamics Iraqda is an oral bioenhancer targeting multidrug resistance in tumors. In many cases, multidrug resistance in cancer arises due to alterations in the expression of protein inhibitors. Iraqda is a third-generation inhibitor of P-gp and BCRP transporters (Hyafil et al., 1993; Witherspoon et al., 1996). It has been used to determine the effects of P-gp and BCRP on the brain distribution of P-gp and BCRP substrate drugs (Breedveld et al., 2005; Bihorel et al., 2007; Chen et al., 2009; Agarwal et al., 2010, 2011b). Studies have also shown that iriqda can serve as a tool to enhance the efficacy of drugs such as sunitinib in the treatment of gliomas (Tang et al., 2012a). This study aimed to investigate the pharmacokinetics of iriqda in the mouse brain and plasma, and to determine its bioavailability after oral and intraperitoneal administration. The physicochemical properties of iridone, such as its low solubility and high lipophilicity, may explain the differences in plasma concentrations observed in this study. Such inter-individual differences have previously been observed in humans after oral administration of iridone (Kuppens et al., 2007) and attributed to variations in iridone solubility. A mouse study found that despite increased doses, the dissolution rate of orally administered iridone remained a limiting factor for its plasma exposure (Ward and Azzarano, 2004). Oral administration is a convenient route of administration in rodents and humans, particularly suitable for long-term administration. Studies have shown that the brain penetration of the TKI sunitinib and its active metabolites is limited by P-gp and BCRP at the blood-brain barrier. In wild-type mice, oral administration of 100 mg/kg iridone increased the brain penetration of sunitinib by nearly 12-fold (Tang et al., 2012a,b). Oral administration of 100 mg/kg iridone resulted in significantly higher plasma and brain tissue exposures compared to intraperitoneal administration. Following oral administration, elacridar had a partition coefficient of 4.31 in the brain, indicating that at this oral dose, it was well distributed from systemic circulation to the brain. The elimination half-life (t1/2) after an oral dose of 100 mg/kg was close to 20 hours, approximately five times that after intraperitoneal or intravenous administration. This phenomenon could have several causes. The prolonged oral t1/2 could be due to a slow dissolution rate (given its poor water solubility), which limits the absorption rate and thus prolongs the drug release time. Therefore, the observed terminal t1/2 may reflect a longer absorption half-life (flip kinetics). The observed time to peak concentration (Tmax) in plasma at 8 hours after administration supports this hypothesis. Another reason for the prolonged t1/2 could be the higher plasma exposure of elacridar, leading to changes in hepatic and renal clearance. However, in vitro studies have shown that elacridar is not a potent inhibitor of any cytochrome P450 enzyme (Ward and Azzarano, 2004), with an IC50 value in the micromolar range. The plasma AUC (dose-normalized) after oral administration was higher than that after intraperitoneal administration, resulting in higher bioavailability. This may be due to the inhibition of intestinal P-gp after oral elacridar, thereby increasing its absorption in the gastrointestinal tract and making its bioavailability higher than that after intraperitoneal administration. Another possible explanation is that enterohepatic circulation occurs after oral elacridar, which may lead to a higher-than-expected AUC value. Regardless of these possibilities, the results indicate that high-dose oral elacridar can result in higher drug exposure in both plasma and brain tissue. A study by Tang et al. (2012a) showed that a dose of 100 mg/kg may be effective in mice, noting that no significant toxicity was observed at this dose. Although toxicity was not assessed in this study, no significant elacridar-related adverse reactions were observed in mice after a single dose. However, toxicity issues cannot be ignored when considering long-term dosing regimens, especially given the high plasma concentrations and long half-life of elacridar at this dose (which may lead to significant drug accumulation at steady state). Intraperitoneal administration is also an ideal option for long-term dosing, especially in preclinical studies. Compared with oral administration, mice receiving intraperitoneal ilagda showed significantly lower plasma exposure at the same dose. This is likely due to poor dissolution and/or absorption of the drug in the peritoneum. Significant inter-animal differences were also observed in drug concentrations in plasma and brain tissue. While intraperitoneal administration facilitates long-term dosing in mice, it leads to reduced ilagda plasma exposure; therefore, the use of this suspension formulation via intraperitoneal administration is currently limited. In this study, the Kp (brain tissue AUC/plasma AUC) ratio was 0.48 after intraperitoneal administration of a 100 mg/kg dose. In a similar study by Padowski and Pollack (2010), the Kp value was 0.0784 after intraperitoneal administration of a 10 mg/kg dose. In summary, these results may indicate a non-linear relationship between plasma and brain tissue exposure with increasing dose, especially after intraperitoneal administration. Exposure after intravenous administration was used as a reference for calculating the absolute bioavailability of the other two routes of administration. Following intravenous administration of a 2.5 mg/kg dose, the AUC values of elacridar in plasma and brain tissue were approximately equal, with a Kp ratio of 0.82 (Table 1). Although widespread distribution of elacridar in brain tissue was observed after intravenous administration, intravenous administration is not a feasible long-term administration protocol for non-catheterized mice due to the difficulty in repeating injections. Furthermore, the administration solution used for intravenous administration is unstable and prone to precipitation. The bioavailability of elacridar is limited by its poor physicochemical properties. The bioavailability after oral administration of a high dose of elacridar is approximately 22%, while the bioavailability after intraperitoneal administration of the same dose is only approximately 1%. The higher plasma AUC after oral administration compared to intraperitoneal administration may be due to the solubilizing effect of bile acids in the intestine. Oral administration of elacridar using this simple suspension appears to be the most effective method to achieve the plasma exposure required for effective inhibition of P-gp and BCRP at the blood-brain barrier. This study found significant differences in brain permeability of elacridar after different routes of administration. First, the Kp ratio of eracrid (a measure of its brain distribution) correlated with its plasma exposure (see Figure 4). When plasma exposure was relatively high (e.g., after oral and intravenous administration), the Kp ratio was greater than 1, with the highest Kp ratio (approximately 5) after oral administration, consistent with the highest plasma exposure. After intraperitoneal administration, plasma exposure was relatively low, and the Kp ratio was less than 1. Second, the brain/plasma concentration ratio curves over time for all routes of administration showed that the ratio first increased to a maximum and then decreased (see Figures 1B, 2B, and 3B). This was unexpected, as the brain/plasma concentration ratio should remain constant after tissue distribution reaches steady state (pseudo-equilibrium). One explanation for these two findings might be that P-gp and BCRP actively remove eracrid from brain tissue. Eracrid inhibits these two transporters at the blood-brain barrier, and the inhibition mechanism may be competitive (as it is a substrate of both transporters). This was demonstrated in a study using positron emission tomography and transporter knockout mice to investigate the effects of P-gp and BCRP on the distribution of eraquadruple at the blood-brain barrier (Kawamura et al., 2011a,b). This finding is currently being further explored in another study. However, this does explain why brain exposure depends on plasma concentration. When plasma concentrations fall below the level required for blood-brain barrier (BBB) efflux saturation, the net efflux from brain tissue will be greater than the amount passively diffused into brain tissue. This will result in a faster decrease in brain tissue concentration than the corresponding plasma concentration, ultimately leading to a decrease in the brain-plasma ratio over time. [1] Concomitant administration of eraquadruple and P-gp and BCRP substrate drugs may improve its distribution within the blood-brain barrier and may enhance efficacy. If we consider the problem of target cells behind the intact blood-brain barrier at the edge of brain tumor invasion or in normal brain tissue, then studying the pharmacokinetics of eraquadruple in the brain becomes particularly important (Agarwal et al., 2011a). The distribution of elacrinar in brain tissue may help address the problem of target cells expressing BCRP and P-gp, thus exhibiting chemotherapeutic resistance (Lu and Shervington, 2008). Intracridar in the brain may effectively inhibit P-gp and BCRP present on these cells, thereby enabling chemotherapeutic drugs to act on these cells and potentially preventing tumor recurrence. Central nervous system-targeted administration of elacrinar could also reduce the dose required to achieve effective brain concentrations, thereby reducing systemic toxicity. Elacridar's preclinical and clinical applications are limited, partly due to its low solubility and bioavailability. Improving the bioavailability and solubility of elacrinar is a pressing issue. Several possible approaches can be explored, including the synthesis of water-soluble prodrugs, the preparation of solid dispersions, and the use of surfactants, cyclodextrins, and penetration enhancers. In summary, this study investigated the pharmacokinetics of elacrinar in mouse plasma and brain tissue and determined its bioavailability after different routes of administration. The results of these experiments can help guide the selection of elacrinar dosage and route of administration in future studies. [1] |
| Molecular Formula |
C34H33N3O5
|
|
|---|---|---|
| Molecular Weight |
563.64
|
|
| Exact Mass |
563.242
|
|
| Elemental Analysis |
C, 72.45; H, 5.90; N, 7.46; O, 14.19
|
|
| CAS # |
143664-11-3
|
|
| Related CAS # |
143851-98-3
|
|
| PubChem CID |
119373
|
|
| Appearance |
Light yellow to brown solid powder
|
|
| Density |
1.3±0.1 g/cm3
|
|
| Boiling Point |
701.6±60.0 °C at 760 mmHg
|
|
| Melting Point |
216-218℃
|
|
| Flash Point |
378.1±32.9 °C
|
|
| Vapour Pressure |
0.0±2.2 mmHg at 25°C
|
|
| Index of Refraction |
1.644
|
|
| LogP |
5.55
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
7
|
|
| Rotatable Bond Count |
8
|
|
| Heavy Atom Count |
42
|
|
| Complexity |
925
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
COC1=CC=CC2=C1NC3=C(C2=O)C=CC=C3C(=O)NC4=CC=C(C=C4)CCN5CCC6=CC(=C(C=C6C5)OC)OC
|
|
| InChi Key |
OSFCMRGOZNQUSW-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C34H33N3O5/c1-40-28-9-5-7-26-32(28)36-31-25(33(26)38)6-4-8-27(31)34(39)35-24-12-10-21(11-13-24)14-16-37-17-15-22-18-29(41-2)30(42-3)19-23(22)20-37/h4-13,18-19H,14-17,20H2,1-3H3,(H,35,39)(H,36,38)
|
|
| Chemical Name |
N-[4-[2-(6,7-dimethoxy-3,4-dihydro-1H-isoquinolin-2-yl)ethyl]phenyl]-5-methoxy-9-oxo-10H-acridine-4-carboxamide
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: 1.67 mg/mL (2.96 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 16.7 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 0.5 mg/mL (0.89 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 0.25 mg/mL (0.44 mM) (saturation unknown) in 5% DMSO + 40% PEG300 + 5% Tween80 + 50% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 0.25 mg/mL (0.44 mM) in 5% DMSO + 95% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 5: 0.05 mg/mL (0.09 mM) in 1% DMSO 99% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 6: 4% DMSO+30% PEG 300+5% Tween 80+ddH2O:1 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7742 mL | 8.8709 mL | 17.7418 mL | |
| 5 mM | 0.3548 mL | 1.7742 mL | 3.5484 mL | |
| 10 mM | 0.1774 mL | 0.8871 mL | 1.7742 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
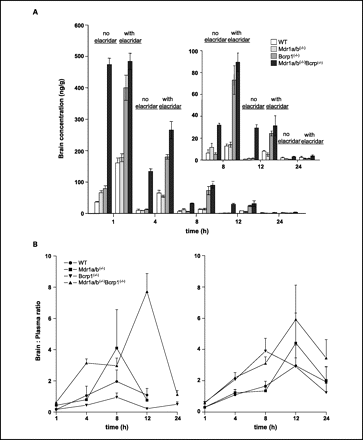 A, brain concentrations of topotecan in WT, Mdr1a/b(−/−) (P-gp knockout), Bcrp1(−/−) (Bcrp1 knockout), and Mdr1a/b(−/−)Bcrp1(−/−) (compound P-gp and Bcrp1 knockout) mice at t = 1, 4, 8, 12, and 24 h after dosing. B, brain versus plasma concentration ratios in mice receiving topotecan (single agent; left) and in combination with elacridar (right).Clin Cancer Res.2007 Nov 1;13(21):6440-9. A, brain concentrations of topotecan in WT, Mdr1a/b(−/−) (P-gp knockout), Bcrp1(−/−) (Bcrp1 knockout), and Mdr1a/b(−/−)Bcrp1(−/−) (compound P-gp and Bcrp1 knockout) mice at t = 1, 4, 8, 12, and 24 h after dosing. B, brain versus plasma concentration ratios in mice receiving topotecan (single agent; left) and in combination with elacridar (right).Clin Cancer Res.2007 Nov 1;13(21):6440-9. |
|---|
|
|