
| Size | Price | Stock | Qty |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g | |||
| Other Sizes |
Purity: ≥98%
Doxorubicin HCl (formerly known as Adriamycin; FI 106; Adriblastina; DOXOCELL; Doxolem; NSC-123127 etc.) is an anthracycline antibiotic agent with anticancer activity. It causes DNA damage and apoptosis in tumor cells and inhibits DNA topoisomerase II. The use of it as an antineoplastic agent was authorized. Doxorubicin, the hydroxylated congener of daunorubicin, is isolated from Streptomyces peucetius var. caesius. By intercalating between base pairs in the DNA helix, doxorubicin inhibits the synthesis of proteins by preventing DNA replication. Furthermore, topoisomerase II is inhibited by doxorubicin.
| Targets |
Topoisomerase I (IC50 = 0.8 μM); Topoisomerase II (IC50 = 2.67 μM); Daunorubicins/Doxorubicins; HIV-1; DNA topoisomerase II (induces DNA double-strand breaks) [5][9]
Histone eviction from chromatin [5] |
|---|---|
| ln Vitro |
Doxorubicin, an antibiotic anthracycline, is widely believed to exhibit its anti-tumor activity on two basic levels: it modifies DNA and generates free radicals to cause DNA damage that causes cancer cells to undergo apoptosis. Doxorubicin inhibits DNA topoisomerase II (TOP2) and can intercalate into DNA strands to prevent DNA synthesis. When cells are multiplying quickly and expressing a lot of TOP2, doxorubicin works best. Additionally, doxorubicin can cause apoptosis by releasing cytochrome c from the mitochondria, ceramide (which activates p53 or other downstream pathways like JNK), the degradation of Akt by serine threonine proteases, an increase in the production of FasL (death receptor Fas/CD95 ligand) mRNA, and an increase in free radical production.[2]
Pre-treatment with GSNO (nitrosoglutathione) results in increased protein glutathionylation and doxorubicin accumulation in the nucleus of the doxorubicin-resistant breast cancer cell line MCF7/Dx, which in turn suppresses resistance.[3] Doxorubicin increased expression of cyclin G2 (CycG2) and phosphorylation of proteins in the ATM, ATM and Rad3-related (ATR) signaling pathways are responsible for induced G2/M checkpoint arrest.[5] Doxorubicin inhibits AMP-activated protein kinase (AMPK), which can be further sensitized by pre-inhibition of AMPK. This results in SIRT1 dysfunction, p53 accumulation, and increased cell death in mouse embryonic fibroblasts (MEFs) and cardiomyocytes.[6] Doxorubicin causes a noticeable heat shock response, and in neuroblastoma cells, it increases the apoptotic effect by either inhibiting or silencing heat shock proteins. When nanomolar Doxorubicin is administered to neuroblastoma cells, it results in a dose-dependent over-ubiquitination of a particular set of proteins without any detectable proteasome inhibition. Additionally, it causes a decrease in the activity of ubiquitinated enzymes like lactate dehydrogenase and α-enolase, whose protein ubiquitination patterns resemble those of the proteasome inhibitor Bortezomib, suggesting that Doxorubicin may also cause protein damage. [8] - Doxorubicin shows potent cytotoxicity against MDA-MB-231 breast cancer cells (IC50 = 0.1 μM) and MCF-7 cells (IC50 = 0.05 μM) after 72h exposure [4] - Induces senescence in human cardiac progenitor cells via p53/p21 pathway activation (SA-β-gal+ cells increase to 80% at 0.5 μM) [1] - Synergistic effect with Bcl-2 siRNA in multidrug-resistant NCI/ADR-RES cells (combination index=0.3 at 50 nM) [4] - Generates ROS in H9c2 cardiomyocytes (2-fold increase at 1 μM) [3] |
| ln Vivo |
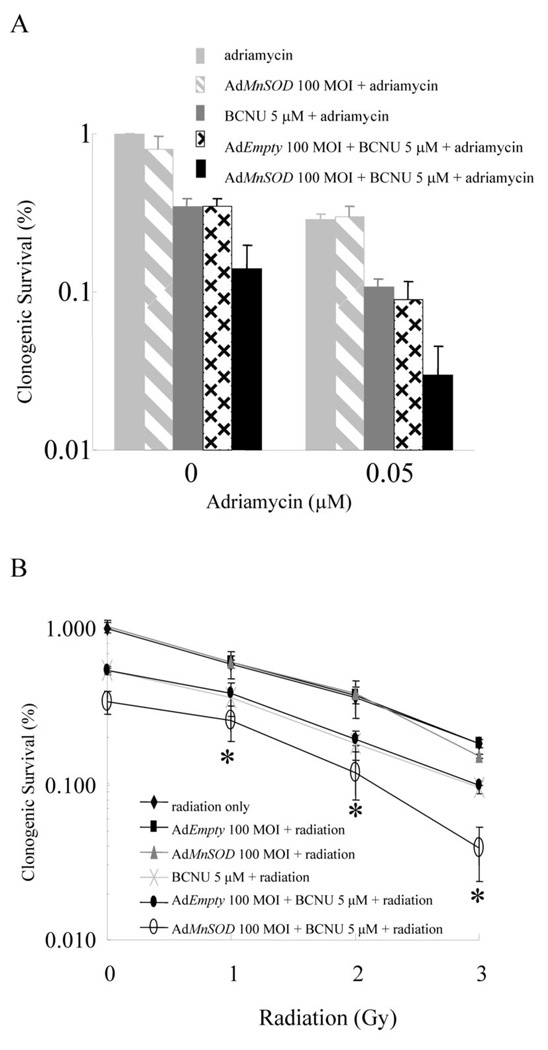
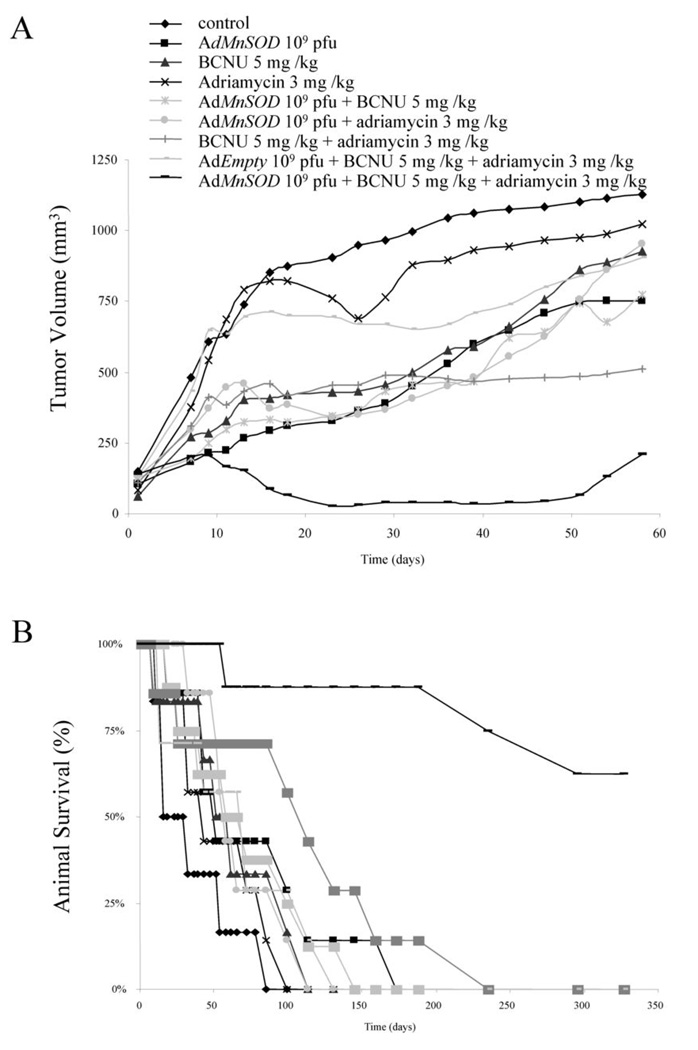
In vivo, the most effective combination for reducing the volumes of MB231 tumors and extending mouse survival is doxorubicin plus 1,3-bis(2-chloroethyl)-1-nitrosourea (BCNU) in conjunction with adenoviral MnSOD (AdMnSOD).[1]
Doxorubicin is indispensable in the treatment of solid tumors in childhood, soft tissue sarcomas, Hodgkin and non-Hodgkin lymphomas, osteosarcomas, Kaposi's sarcoma, and breast and oesophageal carcinomas, despite the fact that its use is restricted by the toxic side effects, both acute and chronic.[2] - Doxorubicin (5 mg/kg weekly × 4, i.v.) reduces tumor volume by 70% in MDA-MB-231 xenograft mice [7] - Magnetic nanoparticle-loaded formulation (3 mg/kg, i.v.) increases tumor drug concentration 5-fold vs free drug [7] - Cumulative dose (15 mg/kg) induces chronic cardiotoxicity in rats: LVEF decrease 40%, fibrosis area 25% [2] |
| Enzyme Assay |
Purified human DNA topoisomerase I was assayed quantitatively by enzyme titrations with supercoiled pHC624 DNA in the presence of 0-2.0 microM doxorubicin. Supercoiled and relaxed DNAs were resolved by agarose gel electrophoresis in the presence of ethidium bromide, and the percentage of conversion of supercoiled DNA to relaxed DNA was quantified by scanning microdensitometry. The inhibition of DNA topoisomerase I activity was measured at varying concentrations of doxorubicin. Doxorubicin inhibited enzyme activity at an IC50 value (the concentration required to inhibit 50% of the total activity) of 0.8 microM. Similar inhibition was observed for daunomycin, a structurally related anthracycline antitumor drug. These results indicate that anthracyclines inhibit human DNA topoisomerase I activity at concentrations that cause DNA damage and cytotoxicity in vivo [11].
- Topoisomerase II decatenation assay: Kinetoplast DNA incubated with enzyme and Doxorubicin (0.1-10 μM) for 30 min at 37°C. DNA breaks visualized by agarose gel electrophoresis [5] - Caspase-3 activity assay: Cell lysates treated with Doxorubicin (1 μM) incubated with DEVD-pNA substrate. Cleavage measured at 405 nm [1] |
| Cell Assay |
Cell Culture[7]
LS141 primary human cell line was derived from a patient with high-grade retroperitoneal dedifferentiated liposarcoma and the MPNST cells were derived from a patient with a high-grade peripheral nerve sheath tumor of the thigh. These were grown in RPMI1640 supplemented with 15% heat-inactivated fetal bovine serum plus penicillin and streptomycin.[7] Colony Assays [7] MPNST cells were treated with doxorubicin, flavopiridol, or the combination of the two drugs together in sequence. MPNST cells were chosen given that LS141 (and other CDK4 dependent) cells are exquisitely sensitive to CDK4 inhibition in vitro, thus making combination studies uninterruptable. MPNST cells were plated, in triplicate, at a density of 1000 cells/100 mm2 per plate. Twenty-four hours after plating, cells were treated for 24 hours with the IC50 of doxorubicin (D, 15 nM), flavopiridol (F, 150 nM), drug free media (control), or a combination of the two drugs, either concomitantly or sequentially for 24 hours each. After treatment, drug-containing medium was removed and cells were allowed to grow for 10 days to form colonies. The resulting colonies were stained with 0.01% crystal violet for 30 minutes and colonies counted using an automated colony counter. Results are presented as the percentage of untreated controls and the statistical significance of the experimental results was determined by the two-sided t test. - Apoptosis assay: Cells stained with Annexin V/PI after 48h Doxorubicin treatment (0.1-5 μM). Quantified by flow cytometry [4] - ROS measurement: H9c2 cells loaded with DCFH-DA, treated with Doxorubicin (0.5-5 μM). Fluorescence read at 488/525 nm [3] - Senescence assay: SA-β-gal staining performed after 72h exposure to 0.5 μM Doxorubicin. Blue-stained cells counted microscopically [1] |
| Animal Protocol |
Female athymic nude mice injected s.c. with MB231 cells; 3 mg/kg/day; Delivered intratumorly
Female athymic nude mice injected s.c. with MB231 cells In vivo studies LS141 xenografts were established by directly implanting into severe combined immunodeficient (SCID) mice. Once tumors reached 100 mm3 , groups of five mice were treated with the maximum tolerated dose (MTD) of flavopiridol (9 mg/kg), doxorubicin (0.9 mg/kg), or doxorubicin (0.7 mg/kg) followed by flavopiridol (7 mg/kg) at selected time points (1, 4 and 7 hours). In addition, one set of animals was treated in reverse order of flavopiridol followed by doxorubicin, administered 7 hours apart. All treatments were administered in intraperitoneal fashion, twice weekly, for a total of 5 treatments. Tumors were measured every 2 to 3 days with calipers, and tumor volumes were calculated by the formula π/ 6 × (large diameter) × (small diameter)2. Tumor volume was compared between groups of mice at various points in time based on the experiment and the statistical significance of the experimental results was determined by the two-sided t test. Given the aggressive morbidity of the tumors, animal survival data could not be estimated. Toxicity was monitored by weight loss. These studies were done in accordance with the Principles of Laboratory Animal Care, under an IACUC-approved protocol. - Xenograft model: MDA-MB-231 cells implanted subcutaneously in nude mice. Doxorubicin (5 mg/kg in saline) administered i.v. weekly × 4. Tumors measured biweekly [7] - Cardiotoxicity model: Rats injected i.p. with Doxorubicin (2.5 mg/kg cumulative dose weekly × 6). Echocardiography performed pre/post treatment [2] - Biodistribution: Tumor-bearing mice injected with 99mTc-labeled liposomal Doxorubicin (3 mg/kg). Organs harvested at 24h for gamma counting [10] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
In patients with HIV-associated Kaposi's sarcoma, after administration of 10 mg/m² liposomal doxorubicin, the calculated Cmax and AUC values were 4.12 ± 0.215 μg/mL and 277 ± 32.9 μg/mL•h, respectively. Approximately 40% of the dose was found in bile within 5 days, while only 5% to 12% of the drug and its metabolites were found in urine during the same period. Less than 3% of the doxorubicin dose was recovered in urine within 7 days. The steady-state volume of distribution of doxorubicin ranged from 809 L/m² to 1214 L/m². The plasma clearance of doxorubicin ranged from 324 mL/min/m² to 809 mL/min/m², primarily through metabolism and bile excretion. There was also a sex difference in doxorubicin clearance, with males having a higher clearance than females (1088 mL/min/m² vs. 433 mL/min/m²). Following administration of doxorubicin hydrochloride at doses ranging from 10 mg/m² to 75 mg/m², plasma clearance was estimated at 1540 mL/min/m² in children over 2 years of age and 813 mL/min/m² in infants under 2 years of age. Unencapsulated doxorubicin hydrochloride is unstable in gastric acid, and animal studies have shown that the drug is almost not absorbed from the gastrointestinal tract. The drug is highly irritating to tissues and therefore must be administered intravenously. In patients with AIDS-associated Kaposi's sarcoma, after a single intravenous infusion of 10 or 20 mg/m² of liposomal doxorubicin hydrochloride, the mean peak plasma concentrations of doxorubicin (primarily bound to liposomes) were 4.33 or 10.1 μg/mL at 15 minutes post-infusion and 4.12 or 8.34 μg/mL at 30 minutes post-infusion. In adult patients with AIDS-associated Kaposi's sarcoma, the mean peak plasma concentration 15 minutes after intravenous infusion of 40 mg/m² doxorubicin hydrochloride was 20.1 μg/mL. Unencapsulated (conventional) doxorubicin hydrochloride exhibited linear pharmacokinetic characteristics; polyethylene glycol-stabilized liposomal doxorubicin hydrochloride also showed dose-proportional linear pharmacokinetic characteristics in the dose range of 10–20 mg/m². It has been reported that the pharmacokinetics of liposomally encapsulated doxorubicin at a dose of 50 mg/m² is non-linear. It is expected that at a dose of 50 mg/m², its elimination half-life will be longer, its clearance lower, and the increase in the area under the plasma concentration-time curve will be greater than that at a dose-proportional rate. Encapsulating doxorubicin hydrochloride in polyethylene glycol (PEG)-stabilized (stealthy) liposomes significantly alters its pharmacokinetics compared to conventional intravenous formulations (i.e., unencapsulated drugs), resulting in reduced distribution in peripheral tissues, increased distribution in Kaposi's sarcoma lesions, and decreased plasma clearance. Conventionally administered doxorubicin is widely distributed in plasma and tissues. It is detectable in the liver, lungs, heart, and kidneys within just 30 seconds of intravenous injection. Doxorubicin is absorbed by cells and binds to cellular components, especially nucleic acids. The volume of distribution for conventionally intravenously administered doxorubicin hydrochloride is approximately 700-1100 liters/m². Unencapsulated doxorubicin binds to plasma proteins at a rate of approximately 50-85%... Intravenous injection of liposome-encapsulated doxorubicin hydrochloride resulted in greater distribution in Kaposi's sarcoma lesions than in healthy skin. Following a single intravenous injection of 20 mg/m² of liposome-encapsulated doxorubicin hydrochloride, doxorubicin concentrations in Kaposi's sarcoma lesions were 19 times higher (range: 3–53 times) than in healthy skin; however, this study did not consider blood drug concentrations in either the lesions or healthy skin. Furthermore, intravenous injection of liposome-encapsulated doxorubicin resulted in 5.2–11.4 times higher distribution in Kaposi's sarcoma lesions compared to intravenous injection of the same dose of conventional (unencapsulated) doxorubicin. The mechanism by which liposome encapsulation enhances doxorubicin distribution in Kaposi's sarcoma lesions is not fully elucidated, but previous studies have shown that polyethylene glycol (PEG)-like stable liposomes containing colloidal gold as a marker can penetrate Kaposi's sarcoma-like lesions in animals. Liposomes may also exude through the intercellular spaces of Kaposi's sarcoma endothelial cells. After drug entry into the lesion, liposome degradation makes them permeable in situ, potentially leading to local drug release. For more complete data on the absorption, distribution, and excretion of doxorubicin (16 in total), please visit the HSDB records page. Metabolism/Metabolites Doxorubicin is metabolized via three pathways: single-electron reduction, two-electron reduction, and deglycosylation. However, approximately half of the dose is excreted unchanged. Two-electron reduction is the primary metabolic pathway of doxorubicin. In this pathway, doxorubicin is reduced to secondary doxorubicin alcohol by several enzymes, including alcohol dehydrogenase [NADP(+)], carbonyl reductase [NADPH]1, carbonyl reductase [NADPH]3, and aldehyde-ketone reductase 1 family member C3. Multiple oxidoreductases in the cytoplasm and mitochondria promote single-electron reduction, generating doxorubicin semiquinone radicals. These enzymes include mitochondrial and cytoplasmic NADPH dehydrogenases, xanthine oxidase, and nitric oxide synthase. This semiquinone metabolite can be reoxidized to doxorubicin, but this process generates reactive oxygen species (ROS) and hydrogen peroxide. The primary cause of doxorubicin-related adverse reactions (especially cardiotoxicity) is the reactive oxygen species (ROS) generated by this pathway, rather than the formation of doxorubicin semiquinone. Deglycosylation is a minor metabolic pathway, accounting for only 1% to 2% of doxorubicin metabolism. Doxorubicin can be reduced to doxorubicin deoxyaglycone or hydrolyzed to doxorubicin hydroxyaglycone by cytoplasmic NADPH quinone dehydrogenase, xanthine oxidase, and NADPH-cytochrome P450 reductase. Non-encapsulated doxorubicin is metabolized by NADPH-dependent aldehyde-ketone reductases to the hydrophilic 13-hydroxy metabolite doxorubicinol, which has antitumor activity and is the major metabolite of doxorubicin; these reductases are present in most cells, and possibly all cells, especially in erythrocytes, liver, and kidneys. Although not fully identified, doxorubicinol also appears to be a major culprit in the cardiotoxicity of this drug. Reports indicate that after a single intravenous injection of 10–50 mg/m² of polyethylene glycol-stabilized liposome-encapsulated doxorubicin hydrochloride, plasma doxorubicin alcohol concentrations are extremely low or undetectable (i.e., 0.8–26.2 ng/mL). It remains unclear whether these liposome-encapsulated anthracyclines are less cardiotoxic than conventional (unencapsulated) drugs, and whether current prophylactic measures for unencapsulated drugs should also apply to liposomal formulations. Following the use of polyethylene glycol (PEG)-stabilized liposome injections, a significant decrease or disappearance of plasma concentrations of the major doxorubicin metabolite has been observed, suggesting that the drug may not be released in large quantities from the liposomes during circulation, or that some doxorubicin may be released, but the elimination rate of doxorubicin alcohol is much higher than the release rate; doxorubicin hydrochloride not encapsulated in PEG-stabilized liposomes is metabolized to doxorubicin alcohol. Other non-therapeutic metabolites include poorly soluble aglycones such as doxorubicinone (doxorubicin) and 7-deoxydoxorubicinone (17-deoxydoxorubicin), and their conjugates. These aglycones are formed in microsomes via NADPH-dependent cytochrome reductase-mediated partial cleavage of aminoglycosides. The enzymatic reduction of doxorubicin to 7-deoxyaglycone is key to its cytotoxic effects, as this process generates hydroxyl radicals, leading to widespread cell damage and death. For unencapsulated doxorubicin, more than 20% of the drug in plasma is present as a metabolite within 5 minutes of administration; 70% after 30 minutes; 75% after 4 hours; and 90% after 24 hours. …At least six metabolites have been identified, the most prominent being doxorubicin alcohol. This product is produced by the reduction of the C13 ketone group by an enzyme present in leukocytes and erythrocytes (presumably also present in malignant tissues). Doxorubicin can be converted into doxorubicin alcohol, aglycones, and other derivatives. For more complete data on the metabolism/metabolites of doxorubicin (a total of 6 metabolites), please visit the HSDB record page. Doxorubicin is metabolized via three metabolic pathways: single-electron reduction, two-electron reduction, and deglycosylation. However, approximately half of the dose is excreted unchanged from the body. Two-electron reduction produces doxorubicin alcohol, a secondary alcohol. This pathway is considered the primary metabolic pathway. Single-electron reduction is catalyzed by various oxidoreductases, generating doxorubicin semiquinone radicals. These enzymes include mitochondrial and cytoplasmic NADPH dehydrogenases, xanthine oxidase, and nitric oxide synthase. Deglycosylation is a minor metabolic pathway (1-2% of the dose is metabolized via this pathway). The resulting metabolites are deoxyaglycones or hydroxyaglycones, formed through reduction or hydrolysis, respectively. Enzymes potentially involved in this pathway include xanthine oxidase, NADPH-cytochrome P450 reductase, and cytoplasmic NADPH dehydrogenase. Excretion pathway: 40% of the dose is excreted via bile within 5 days. 5-12% of the drug and its metabolites are excreted via urine within the same time period. Less than 3% of the dose recovered in urine is doxorubicin alcohol. Half-life: Terminal half-life = 20-48 hours. Biological Half-Life The terminal half-life of doxorubicin is 20 to 48 hours. The distribution half-life of doxorubicin is approximately 5 minutes. For the liposomal formulation, in patients with AIDS-associated Kaposi's sarcoma, the first-phase half-life and second-phase half-life of 10 mg/m² doxorubicin were calculated to be 4.7 ± 1.1 hours and 52.3 ± 5.6 hours, respectively. Plasma concentrations of unencapsulated doxorubicin and its metabolites exhibit a biphasic or triphasic decrease. In the first phase of the triphasic model, unencapsulated doxorubicin is rapidly metabolized, presumably through the first-pass effect in the liver. Most of the metabolism appears to be completed before administration. In the triphasic model, unencapsulated doxorubicin and its metabolites are rapidly distributed into the extravascular space, with a plasma half-life of doxorubicin of approximately 0.2–0.6 hours and that of its metabolites of approximately 3.3 hours. Subsequently, plasma concentrations of doxorubicin and its metabolites are maintained at relatively long levels, likely due to tissue binding. In the second phase, the plasma half-life of unencapsulated doxorubicin was 16.7 hours, while that of its metabolites was 31.7 hours. In the biphasic model, the average initial distribution half-life was reported to be approximately 5–10 minutes, and the average terminal elimination half-life was approximately 30 hours. The plasma concentrations of liposome-encapsulated doxorubicin hydrochloride appear to decrease in a biphasic manner. In patients with AIDS-associated Kaposi's sarcoma, following a single intravenous injection of 10–40 mg/m² doses of doxorubicin hydrochloride liposomes, the average initial plasma half-life (t1/2α) of doxorubicin was 3.76–5.2 hours, while the average terminal elimination half-life (t1/2β) was 39.1–55 hours. The approximately 5-minute initial distribution half-life indicates rapid tissue absorption of doxorubicin, while its slow elimination from tissue is reflected in the 20–48-hour terminal half-life. The plasma half-life of doxorubicin in patients is about 17 hours, while the plasma half-life of its metabolites is about 32 hours. - The plasma half-life in humans is 20-48 hours, and the volume of distribution is 25 L/kg [11] - Plasma protein binding rate is 90% [11] - Major metabolites: doxorubicin alcohol (active ingredient), aglycone [9] - Bile excretion >60% [9] |
| Toxicity/Toxicokinetics |
The chemotherapy drug doxorubicin (DOX) exhibits dose-dependent cardiotoxicity, leading to heart failure. This study aimed to evaluate the timing and severity of cardiotoxicity in rats administered three different regimens (DOX1: a single intraperitoneal injection of 10 mg/kg DOX; DOX2: daily intraperitoneal injections of 1 mg/kg DOX for 10 days; DOX3: weekly intraperitoneal injections of 2 mg/kg DOX for 5 weeks). Weekly transthoracic echocardiography was performed to assess the timing and severity of cardiotoxicity in the three groups. Mortality reached 80% in the DOX1 group by day 28, while it reached 80% in the DOX2 and DOX3 groups by days 107 and 98, respectively. In the DOX1 group, ejection fraction decreased by 30% at week 2; in the DOX2 group, by 55% at week 13; and in the DOX3 group, by 42% at week 13. Furthermore, there were significant differences in cardiac function between the DOX1 and DOX3 groups, while cardiac function was similar between the DOX2 and DOX3 groups. These results suggest that administration over several days (DOX2) or weeks (DOX3) improves survival and presents with more typical symptoms of doxorubicin-induced dilated cardiomyopathy, despite later onset, compared to a single bolus injection of 10 mg/kg doxorubicin. [J Am Assoc Lab Anim Sci.] July 2007; 46(4):20-32.]
Effects during pregnancy and lactation ◉ Overview of medication use during lactation Most data suggest that breastfeeding is contraindicated during maternal treatment with antitumor drugs, especially anthracyclines (such as doxorubicin). Breastfeeding may be safe during intermittent treatment if the lactation period is appropriate; however, it is difficult to determine the appropriate lactation period due to the high concentration and long duration of the active metabolite doxorubicinol in breast milk. Some studies suggest discontinuing breastfeeding 5 to 10 days after administration. However, recent worst-case pharmacokinetic models suggest that 13 days after colostrum is needed to minimize systemic and intestinal toxicity. Chemotherapy may adversely affect the normal microbiota and chemical composition of breast milk. Women receiving chemotherapy during pregnancy are more likely to experience breastfeeding difficulties. ◉ Effects on Breastfed Infants A woman was diagnosed with B-cell lymphoma at 27 weeks of gestation. Labor was induced at 34 weeks and 4 days of gestation, and she began a standard regimen of rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone, at an unspecified dosage, for 21 days, starting on day 2 postpartum. She expressed and discarded breast milk, fed her infant with donated breast milk for the first 10 days of each cycle, and then breastfed for the remaining 10 days before the start of the next cycle. This 10-day breastfeeding pause was determined based on the half-life of vincristine (approximately 3 half-lives). According to reports, after completing four cycles of chemotherapy, her baby was in good health, developing normally, and without any complications. ◉ Effects on Lactation and Breast Milk A study of adolescent males who received chemotherapy for childhood malignancies found that doxorubicin treatment was associated with elevated serum prolactin levels. A woman diagnosed with Hodgkin's lymphoma in mid-pregnancy received three cycles of chemotherapy in late pregnancy and resumed chemotherapy four weeks postpartum. Breast milk samples were collected 15 to 30 minutes before and after chemotherapy within 16 weeks of restarting chemotherapy. The chemotherapy regimen included doxorubicin 40 mg, bleomycin 16 units, vincristine 9.6 mg, and dacarbazine 600 mg, administered every two weeks over two hours. Researchers compared the microbiome and metabolic profile of this woman's breast milk with that of eight healthy women who did not receive chemotherapy. The breast milk microbiota of this patient differed significantly from that of healthy women, with increased abundance of Acinetobacter sp., Xanthomonadacea, and Stenotrophomonas sp., and decreased abundance of Bifidobacterium sp. and Eubacterium sp. Several chemical components in the breast milk of treated women also differed significantly, most notably with decreased levels of DHA and inositol. A telephone follow-up study investigated 74 women who received cancer chemotherapy at the same center during mid-to-late pregnancy to determine their postpartum breastfeeding success rates. The results showed that only 34% of the women were able to exclusively breastfeed their infants, and 66% reported breastfeeding difficulties. In contrast, the breastfeeding success rate was as high as 91% for 22 mothers who were diagnosed with cancer during pregnancy but did not receive chemotherapy. Other statistically significant correlations included: 1. Mothers with lactation difficulties received an average of 5.5 chemotherapy cycles, while mothers without lactation difficulties received an average of 3.8 chemotherapy cycles; 2. Mothers with lactation difficulties received their first chemotherapy cycle an average of 3.4 weeks earlier during pregnancy. Of the 62 women who received doxorubicin regimens, 39 had lactation difficulties. - Cardiotoxicity: Left ventricular ejection fraction (LVEF) decreased by >10% at cumulative doses >400 mg/m² [9] - Myelosuppression: Lowest values were observed at 10–14 days (white blood cell count <2000/mm³) [11] - Mouse LD50 = 10 mg/kg (single intravenous injection) [6] - Hepatotoxicity: ALT increased 3-fold at a cumulative dose of 15 mg/kg [2] |
| References |
[1]. Cancer Res. 2009 May 15;69(10):4294-300. [2]. Food Chem Toxicol. 2010 Jun;48(6):1425-38. [3]. Biochem J. 2011 Dec 1;440(2):175-83. [4]. Br J Cancer. 2011 Mar 15;104(6):957-67. [5]. J Biol Chem. 2012 Jun 29;287(27):22838-53. [6]. J Biol Chem. 2012 Mar 9;287(11):8001-12. [7]. Clin Cancer Res. 2012 May 1;18(9):2638-47. [8]. FEBS J. 2012 Jun;279(12):2182-91. [9]. Nat Rev Cancer. 2009 May;9(5):338-50. |
| Additional Infomation |
According to an independent committee of scientific and health experts, doxorubicin hydrochloride (Acritocillin) may be carcinogenic. It may also have developmental toxicity and male reproductive toxicity, depending on state or federal labeling requirements. Doxorubicin hydrochloride is an orange-red needle-like substance. Its aqueous solution is yellow-orange at acidic pH, orange-red at neutral pH, and purplish-blue at pH above 9. (NTP, 1992) Doxorubicin hydrochloride is an anthracycline antibiotic. Doxorubicin hydrochloride (liposomes) is a prescription anti-tumor drug approved by the U.S. Food and Drug Administration (FDA) for the treatment of certain types of cancer, including ovarian cancer, multiple myeloma, and HIV-related Kaposi's sarcoma. Kaposi's sarcoma is caused by human herpesvirus 8 (HHV-8) infection. HHV-8 infection may be an opportunistic infection (OI) of HIV. Doxorubicin hydrochloride is the hydrochloride salt of doxorubicin, an anthracycline antibiotic with anti-tumor activity. Doxorubicin is isolated from Streptomyces peucetius var. caesius and is a hydroxylated homologue of daunorubicin. Doxorubicin can insert between base pairs of the DNA double helix, thereby preventing DNA replication and ultimately inhibiting protein synthesis. Furthermore, doxorubicin inhibits topoisomerase II, leading to an increase and stabilization of the cleavable enzyme-DNA linker complex during DNA replication, thus preventing the rejoining of nucleotide chains after double-strand breaks. Doxorubicin also forms oxygen free radicals, leading to cell membrane lipid peroxidation and thus cytotoxicity; the formation of oxygen free radicals is also a contributing factor to the toxicity of anthracycline antibiotics, particularly their effects on cardiac and skin blood vessels.
An antitumor antibiotic, extracted from Streptomyces peucetius. It is a hydroxylated derivative of daunorubicin. See also: Doxorubicin (with active moiety). Drug Indications Petrified liposomal celecoxib is indicated for adults as monotherapy for the treatment of patients with metastatic breast cancer, particularly those at increased cardiac risk; or for the treatment of women with advanced ovarian cancer who have failed first-line platinum-based chemotherapy; or in combination with bortezomib for the treatment of patients with advanced multiple myeloma who have received at least one prior therapy and are either ineligible for or have received bone marrow transplantation. It is also indicated for the treatment of HIV-associated Kaposi's sarcoma (KS) with low CD4 counts (<200 CD4 lymphocytes/mm3) and extensive mucocutaneous or visceral lesions. Celdoxome petrified liposomal celecoxib can be used as first-line systemic chemotherapy or as second-line chemotherapy for HIV-associated KS patients whose disease has progressed or who are intolerant to combination systemic chemotherapy containing at least two of the following: vinca alkaloids, bleomycin, and standard doxorubicin (or other anthracyclines). Caelyx pegylated liposomes are indicated for: as monotherapy in patients with metastatic breast cancer at increased cardiac risk; for the treatment of women with advanced ovarian cancer who have failed first-line platinum-based chemotherapy; in combination with bortezomib for the treatment of patients with advanced multiple myeloma who have received at least one prior therapy and are ineligible for or have received bone marrow transplantation; and for the treatment of patients with HIV-associated Kaposi's sarcoma (KS) with low CD4 counts. Myocet liposomes in combination with cyclophosphamide are indicated for first-line treatment of metastatic breast cancer in adult women. Treatment of breast and ovarian cancer. Treatment of hepatocellular carcinoma. Doxorubicin is a deoxyhexoside, anthracycline, aminoglycoside, tetrabenzoquinone, paraquinone, primary α-hydroxy ketone, and tertiary α-hydroxy ketone. It is a metabolite of Escherichia coli. It is the conjugate base of doxorubicin (1+). It is derived from the hydride of tetrabenzoquinone. Doxorubicin hydrochloride (liposomes) is a prescription antitumor drug. Approved by the U.S. Food and Drug Administration (FDA). Approved for the treatment of certain types of cancer, including ovarian cancer, multiple myeloma, and HIV-associated Kaposi's sarcoma. Kaposi's sarcoma is caused by infection with human herpesvirus type 8 (HHV-8). HHV-8 infection may be an opportunistic infection (OI) of HIV. Doxorubicin is a cytotoxic anthracycline antibiotic, isolated in 1970 from cultures of Streptomyces peucetius var. caesius along with another cytotoxic drug, daunorubicin. Although both contain glycosidic aglycones and sugar moieties, doxorubicin has a primary alcohol group at the end of its side chain, while daunorubicin has a methyl group at the end. Although its detailed molecular mechanism is not fully elucidated, it is generally believed that doxorubicin works through DNA intercalation, ultimately leading to DNA... Damage. Production of reactive oxygen species. Due to its significant efficacy and broad range of applications, doxorubicin was approved by the FDA in 1974 for the treatment of various cancers, including but not limited to breast cancer, lung cancer, stomach cancer, ovarian cancer, thyroid cancer, non-Hodgkin's lymphoma and Hodgkin's lymphoma, multiple myeloma, sarcoma, and childhood cancers. However, one of the main side effects of doxorubicin is cardiotoxicity; therefore, it is not suitable for patients with heart failure, and treatment should be discontinued once the maximum tolerated cumulative dose is reached. Doxorubicin is an anthracycline topoisomerase inhibitor. The mechanism of action of doxorubicin is as a topoisomerase inhibitor. Doxorubicin has been reported to be found in Talaromyces aculeatus, Hamigera fusca, and other organisms with relevant data. Doxorubicin is an anthracycline antibiotic. Antitumor activity. Doxorubicin is derived from Streptomyces peucetius var. Doxorubicin, isolated from Streptomyces, is a hydroxylated analogue of daunorubicin. Doxorubicin can insert between base pairs in the DNA double helix, thereby preventing DNA replication and ultimately inhibiting protein synthesis. Furthermore, doxorubicin inhibits topoisomerase II, leading to an increase and stabilization of the cleavable enzyme-DNA linker complex during DNA replication, thus preventing the rejoining of nucleotide chains after double-strand breaks. Doxorubicin also forms oxygen free radicals, causing cell membrane lipid peroxidation and resulting in cytotoxicity; the formation of oxygen free radicals is also one of the causes of anthracycline antibiotic toxicity (especially cardiovascular and cutaneous vascular toxicity). Doxorubicin is only present in individuals who have used or taken the drug. It is an antitumor antibiotic extracted from Streptomyces. Doxorubicin (Dapoxetine) is a hydroxyl derivative of daunorubicin. [PubChem] Doxorubicin exerts its antimitotic and cytotoxic activities through multiple mechanisms of action: Doxorubicin forms a complex with DNA by inserting between base pairs and inhibits the activity of topoisomerase II by stabilizing the DNA-topoisomerase II complex, thereby preventing the rejoining portion of the topoisomerase II-catalyzed ligation-rejoining reaction. It is an antitumor antibiotic extracted from Streptomyces peucetius. It is a hydroxyl derivative of daunorubicin. See also: Doxorubicin hydrochloride (in salt form); Zoprerin doxorubicin (its active portion); Zoprerin doxorubicin acetate (its active portion). Drug Indications Doxorubicin is indicated for the treatment of acute lymphoblastic leukemia, acute myeloid leukemia, Hodgkin's lymphoma and non-Hodgkin's lymphoma, metastatic breast cancer, metastatic nephroblastoma, metastatic neuroblastoma, metastatic soft tissue and osteosarcoma, metastatic... Doxorubicin is indicated for the treatment of neoplastic diseases such as ovarian cancer, metastatic transitional cell carcinoma of the bladder, metastatic thyroid cancer, metastatic gastric cancer, and metastatic bronchial cancer. It is also indicated for adjuvant therapy in women with axillary lymph node metastasis after primary breast cancer resection. In liposomal formulations, doxorubicin is indicated for the treatment of ovarian cancer that has progressed or relapsed after platinum-based chemotherapy, HIV-associated Kaposi's sarcoma that has failed or is intolerant of prior systemic chemotherapy, and multiple myeloma in patients who have not previously received bortezomib (in combination with bortezomib). Patients must have received at least one prior therapy. FDA Label Zozcetti PEGylated liposomes are a medicine for the treatment of the following types of cancer in adults:EUR¢ The breast cancer has spread to other parts of the body, and the patient is at risk of heart problems. Zoscotil pegylated liposomes can be used alone to treat this disease; âEURAdvanced ovarian cancer refers to women who have previously received treatment, including platinum-based anticancer drugs, but with poor response.EUR¢ Multiple myeloma (leukocytic carcinoma of the bone marrow) is indicated for patients who have previously received at least one other treatment and whose disease has progressed, and who have received or are ineligible for bone marrow transplantation. Pegylated liposomal zolastil is used in combination with bortezomib (another anticancer drug); âEURKaposi's sarcoma is commonly seen in HIV patients with severely compromised immune systems. It is a cancer that causes abnormal tissue to grow under the skin, on moist surfaces, or on internal organs. Zolsketil pegylated liposomes contain the active ingredient doxorubicin, a...€˜"Hybrid medicine." This means it's similar to a...€˜The reference drug “contains the same active ingredient, doxorubicin.” However, in zosketine pegylated liposomes, the active ingredient is encapsulated in tiny fat globules called liposomes, unlike doxorubicin. Caelyx® pegylated liposomes are indicated for: as monotherapy for patients with metastatic breast cancer at increased cardiac risk; for women with advanced ovarian cancer who have failed first-line platinum-based chemotherapy; in combination with bortezomib for patients with advanced multiple myeloma who have received at least one prior therapy and are ineligible for or have received bone marrow transplantation; and for patients with HIV-associated Kaposi's sarcoma (KS) with low CD4 counts. Myocet liposomes in combination with cyclophosphamide are indicated for first-line treatment of metastatic breast cancer in adult women. Treatment of breast and ovarian cancer. Mechanism of Action Doxorubicin is generally believed to exert its antitumor activity through two main mechanisms: DNA intercalation and disruption of topoisomerase-mediated repair and free radical-mediated cell damage. Doxorubicin can intercalate into DNA via an anthraquinone ring, which stabilizes the complex by forming hydrogen bonds with DNA bases. This intercalation introduces torsional stress into the polynucleotide structure, disrupting nucleosome structure and leading to nucleosome shedding and replacement. Furthermore, the doxorubicin-DNA complex interferes with doxorubicin's inhibition of topoisomerase II activity, preventing topoisomerase-mediated DNA break repair, thereby inhibiting DNA replication and transcription and inducing apoptosis. Additionally, doxorubicin can be affected by microsomal NADPH. Cytochrome P-450 reductase metabolizes free radicals into semiquinone radicals, which can be re-oxidized to oxygen radicals in the presence of oxygen. Reactive oxygen species (ROS) are known to cause cell damage through various mechanisms, including lipid peroxidation and membrane damage, DNA damage, oxidative stress, and apoptosis. Although the free radicals generated through this pathway can be inactivated by catalase and superoxide dismutase, tumor cells and cardiomyocytes often lack these enzymes, explaining the effectiveness of doxorubicin against cancer cells and its tendency to cause cardiotoxicity. Doxorubicin hydrochloride is an antitumor antibiotic with pharmacological effects similar to daunorubicin. Although this drug has anti-infective properties, its cytotoxicity limits its application as an anti-infective agent. The exact and/or primary mechanism of doxorubicin's antitumor effect is not fully elucidated. The drug's cytotoxicity appears to stem from a complex multi-mechanism, including: free radicals generated after doxorubicin's electron-reductive metabolic activation, drug insertion into DNA, induction of DNA breaks and chromosomal aberrations, and drug-induced cell membrane alterations. In vitro studies suggest that apoptosis (programmed cell death) following doxorubicin treatment may also be involved in the drug's mechanism of action. These mechanisms, along with others (e.g., chelation of metal ions to form drug-metal complexes), may also contribute to the drug's cardiotoxicity. Doxorubicin undergoes enzymatic single-electron and two-electron reduction to generate the corresponding semiquinones and dihydroquinones. 7-Deoxyglycosidic ligands are generated by an enzymatic single-electron reduction reaction. The resulting semiquinone radical reacts with oxygen, triggering a series of cascade reactions to generate hydroxyl radicals. These radicals can react with DNA, RNA, cell membranes, and proteins, leading to cell death. Dihydroquinone, generated by the two-electron reduction of doxorubicin, can also be produced by the reaction of two semiquinones. In the presence of oxygen, dihydroquinone reacts to generate hydrogen peroxide; in the absence of oxygen, dihydroquinone loses its glycosyl group to generate quinone methylate, a monofunctional alkylating agent with low affinity for DNA. The contributions of dihydroquinone and quinone methylate to the cytotoxicity of doxorubicin are unclear. Experimental evidence suggests that doxorubicin inhibits DNA synthesis and DNA-dependent RNA synthesis by intercalating between base pairs to form a complex with DNA, leading to template disorder and steric hindrance. Doxorubicin can also inhibit protein synthesis. Anazoline synthesis. Doxorubicin is active throughout the cell cycle, including interphase. Multiple inducing effects of anthracyclines may contribute to cardiotoxicity. In animal studies, anthracyclines selectively inhibit gene expression of β-actin, troponin, myosin light chain 2, and creatine kinase M isoform in the myocardium, potentially leading to myofibril loss associated with anthracycline-induced cardiotoxicity. Other potential causes of anthracycline-induced cardiotoxicity include cardiomyocyte damage due to calcium overload, altered myocardial adrenergic function, release of vasoactive amines, and release of pro-inflammatory cytokines. Limited data suggest that calcium channel blockers (e.g., isoprenoidamine) or β-adrenergic blockers may prevent calcium overload… Some studies suggest that free radical damage is a primary cause of anthracycline-induced cardiotoxicity. DNA. Anthracycline drugs can intercalate into DNA, chelate metal ions to form drug-metal complexes, and generate oxygen free radicals through redox reactions. Anthracycline drugs contain quinone structures and can undergo reduction reactions via NADPH-dependent reactions to generate semiquinone free radicals, which in turn trigger oxygen free radical cascade reactions. The metabolite doxorubicin appears to be the culprit behind cardiotoxicity, and the heart may be particularly vulnerable to free radical damage due to relatively low concentrations of antioxidants. ... Drug chelation of metal ions (especially iron) forms doxorubicin-metal complexes, which catalyze the generation of reactive oxygen species and are strong oxidants, even in... Lipid peroxidation can be triggered even in the absence of oxygen free radicals. This reaction is not inhibited by free radical scavengers and may be the main mechanism by which anthracycline drugs cause cardiotoxicity. This study investigated the effects of doxorubicin on reactive oxygen metabolism in rat hearts. The results showed that doxorubicin generated oxygen free radicals in cardiac homogenates, sarcoplasmic reticulum, mitochondria, and cytosol (the main sites of cardiac injury). Superoxide production was increased in cardiac myosomal and mitochondrial components. It is clear that the free radicals generated by doxorubicin in the same cardiomyocyte compartments as drug-induced tissue damage may damage the heart by exceeding the oxygen free radical detoxification capacity of cardiac mitochondria and sarcoplasmic reticulum. - Anthracycline antibiotics that can embed in DNA and inhibit topoisomerase II [9] - FDA black box warning: cardiotoxic [9] - Clinical applications: breast cancer, lymphoma, sarcoma [9] - Resistance mechanism: P-glycoprotein efflux, glutathione binding [8] |
| Molecular Formula |
C27H29NO11.HCL
|
|
|---|---|---|
| Molecular Weight |
579.98
|
|
| Exact Mass |
579.15
|
|
| Elemental Analysis |
C, 55.91; H, 5.21; Cl, 6.11; N, 2.42; O, 30.34
|
|
| CAS # |
25316-40-9
|
|
| Related CAS # |
25316-40-9 (Doxorubicin HCl); 23214-92-8
|
|
| PubChem CID |
443939
|
|
| Appearance |
Red to orange solid powder
|
|
| Boiling Point |
810.3ºC at 760 mmHg
|
|
| Melting Point |
216ºC
|
|
| Flash Point |
443.8ºC
|
|
| Vapour Pressure |
9.64E-28mmHg at 25°C
|
|
| Source |
Streptomyces peucetius var. Caesius
|
|
| LogP |
1.503
|
|
| Hydrogen Bond Donor Count |
7
|
|
| Hydrogen Bond Acceptor Count |
12
|
|
| Rotatable Bond Count |
5
|
|
| Heavy Atom Count |
40
|
|
| Complexity |
977
|
|
| Defined Atom Stereocenter Count |
6
|
|
| SMILES |
Cl[H].O([C@@]1([H])C([H])([H])[C@@]([H])([C@@]([H])([C@]([H])(C([H])([H])[H])O1)O[H])N([H])[H])[C@]1([H])C2C(=C3C(C4C(=C([H])C([H])=C([H])C=4C(C3=C(C=2C([H])([H])[C@@](C(C([H])([H])O[H])=O)(C1([H])[H])O[H])O[H])=O)OC([H])([H])[H])=O)O[H]
|
|
| InChi Key |
MWWSFMDVAYGXBV-RUELKSSGSA-N
|
|
| InChi Code |
InChI=1S/C27H29NO11.ClH/c1-10-22(31)13(28)6-17(38-10)39-15-8-27(36,16(30)9-29)7-12-19(15)26(35)21-20(24(12)33)23(32)11-4-3-5-14(37-2)18(11)25(21)34;/h3-5,10,13,15,17,22,29,31,33,35-36H,6-9,28H2,1-2H3;1H/t10-,13-,15-,17-,22+,27-;/m0./s1
|
|
| Chemical Name |
(7S,9S)-7-[(2R,4S,5S,6S)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-9-(2-hydroxyacetyl)-4-methoxy-8,10-dihydro-7H-tetracene-5,12-dione;hydrochloride
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.75 mg/mL (4.74 mM) (saturation unknown) in 5% DMSO + 40% PEG300 + 5% Tween80 + 50% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (3.59 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.08 mg/mL (3.59 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7242 mL | 8.6210 mL | 17.2420 mL | |
| 5 mM | 0.3448 mL | 1.7242 mL | 3.4484 mL | |
| 10 mM | 0.1724 mL | 0.8621 mL | 1.7242 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Treatment of Acute Lymphoblastic Leukemia in Children
CTID: NCT00400946
Phase: Phase 3 Status: Completed
Date: 2024-11-27
 |
|---|
 |