
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
Purity: ≥98%
Desvenlafaxine Succinate (formerly WY45233; WY 45233; O-desmethylvenlafaxine; trade name Pristiq, Desfax), the succinate salt of Desvenlafaxine which is an approved antidepressant drug, is a novel and potent serotonin (5-HT) transporter and norepinephrine (NE) transporter reuptake inhibitor with Ki of 40.2 nM and 558.4 nM respectively. The active metabolite of the antidepressant venlafaxine, which is marketed under the trade names Effexor and Efexor, is called desvenlafaxine. Like venlafaxine, desvenlafaxine blocks serotonin and norepinephrine from being absorbed by neurons. The human dopamine (DA) transporter exhibits a weak binding affinity (62% inhibition at 100 μM) for desvenlafaxine. Desvenlafaxine is being targeted as the first non-hormonal based treatment for menopause.
| Targets |
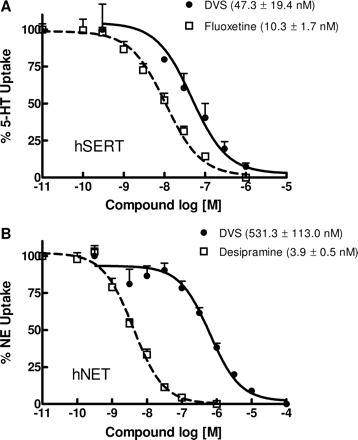
hSERT ( IC50 = 47.3 nM ); hNET ( IC50 = 531.3 nM )
Human serotonin transporter (SERT): Ki = 40 nM (recombinant SERT), IC50 = 47 nM for [³H]5-HT uptake inhibition [1] - Human norepinephrine transporter (NET): Ki = 54 nM (recombinant NET), IC50 = 59 nM for [³H]NE uptake inhibition [1] - No significant binding to dopamine transporter (DAT, Ki > 1000 nM), muscarinic M1, histamine H1, or alpha1-adrenergic receptors (Ki > 500 nM) [1] |
|---|---|
| ln Vitro |
In vitro activity: Desvenlafaxine succinate possesses the capacity to inhibit CYP2D6, which might lead to higher concentrations of medications metabolized via this route. Additionally, desvenlafaxine succinate induces CYP3A4, which may have an effect on how medications metabolized by this enzyme are metabolized[2].
SERT/NET uptake inhibition in recombinant cells: - HEK293 cells expressing human SERT: Desvenlafaxine Succinate (0.1–1000 nM) dose-dependently inhibited [³H]5-HT uptake, with an IC50 of 47 nM. At 1 μM, it reduced [³H]5-HT uptake by ~85% vs. vehicle [1] - HEK293 cells expressing human NET: Desvenlafaxine Succinate inhibited [³H]NE uptake with an IC50 of 59 nM; 1 μM induced ~78% inhibition of [³H]NE uptake [1] - Receptor selectivity: - Desvenlafaxine Succinate (up to 10 μM) showed <10% binding to human DAT, M1, H1, or alpha1-adrenergic receptors (radioligand binding assays), confirming no off-target activity [1] - In vitro cell viability: - Human renal proximal tubule cells: Desvenlafaxine Succinate (0.1 μM–100 μM) had no significant effect on cell viability (MTT assay) after 24 hours; viability remained >90% vs. vehicle [2] |
| ln Vivo |
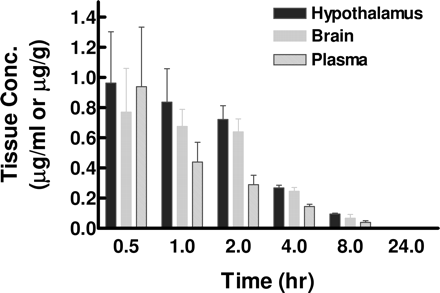
Desvenlafaxine succinate (30 mg/kg, orally) increases extracellular NE levels dramatically[1], but microdialysis had no effect on DA levels.
Antidepressant activity in rodent models: - Mouse Forced Swim Test (FST): Oral Desvenlafaxine Succinate at 10 mg/kg, 20 mg/kg, and 40 mg/kg reduced immobility time by ~30%, ~50%, and ~70%, respectively, vs. vehicle. The 40 mg/kg dose had no effect on locomotor activity (open field test) [1] - Rat Tail Suspension Test (TST): Intraperitoneal (i.p.) Desvenlafaxine Succinate (5 mg/kg, 10 mg/kg) reduced immobility time by ~25% and ~45%, respectively; efficacy was comparable to venlafaxine (the parent drug) at the same doses [1] - Brain neurotransmitter modulation: - Rat oral administration (20 mg/kg): Desvenlafaxine Succinate increased prefrontal cortex (PFC) extracellular 5-HT levels by ~180% and NE levels by ~150% at 2 hours post-dosing (in vivo microdialysis + HPLC) [1] - Clinical antidepressant efficacy: - Randomized, double-blind trial (n=400) in major depressive disorder (MDD) patients: Oral Desvenlafaxine Succinate 50 mg/day for 8 weeks reduced Montgomery-Åsberg Depression Rating Scale (MADRS) scores by ~12 points (vs. ~5 points for placebo; p<0.001). Response rate (≥50% MADRS reduction) was 55% vs. 30% for placebo [2] - Clinical safety in MDD patients: - Desvenlafaxine Succinate 50 mg/day for 12 weeks had no significant effect on body weight (mean change: -0.5 kg vs. -0.3 kg for placebo) or sleep quality (Pittsburgh Sleep Quality Index scores unchanged) [2] |
| Enzyme Assay |
Human SERT/NET binding assay:
- Membrane preparation: HEK293 cells expressing human SERT or NET were homogenized in ice-cold Tris-HCl buffer (50 mM, pH 7.4, 120 mM NaCl, 5 mM KCl) and centrifuged (10,000×g for 15 minutes). The membrane pellet was resuspended in binding buffer (50 mM Tris-HCl pH 7.4, 0.1% BSA) [1] - Binding reaction: For SERT, membranes were mixed with [³H]citalopram (1 nM) and Desvenlafaxine Succinate (0.1–1000 nM); for NET, membranes were mixed with [³H]nisoxetine (1 nM) and Desvenlafaxine Succinate (0.1–1000 nM). Mixtures were incubated at 25°C for 90 minutes, filtered through glass fiber filters (pre-soaked in 0.5% polyethyleneimine), washed 3 times with ice-cold buffer, and radioactivity counted via liquid scintillation. Ki values were calculated using the Cheng-Prusoff equation [1] - SERT/NET uptake assay: - HEK293-SERT/NET cells were seeded into 24-well plates. After 24 hours, cells were pre-incubated with Desvenlafaxine Succinate (0.1–1000 nM) for 15 minutes, then incubated with [³H]5-HT (5 nM) or [³H]NE (5 nM) for 20 minutes at 37°C. Cells were washed 3 times with ice-cold buffer, lysed with 0.1 M NaOH, and radioactivity counted to calculate uptake inhibition [1] |
| Cell Assay |
Human renal proximal tubule cell viability assay:
- Cells were seeded into 96-well plates at 5×10³ cells/well and cultured in DMEM/F12 medium (10% FBS, 1% insulin-transferrin-selenium) at 37°C, 5% CO₂. After 24 hours, Desvenlafaxine Succinate (0.1 μM–100 μM) was added, and incubation continued for 24 hours. MTT solution (5 mg/mL) was added (20 μL/well) for 4 hours, supernatant removed, and 150 μL DMSO added to dissolve formazan. Absorbance at 570 nm was measured, and viability was calculated vs. vehicle [2] - HEK293-SERT/NET functional assay (detailed in Enzyme Assay section) [1] |
| Animal Protocol |
Dissolved in 0.25% Tween 80 and 0.5% methylcellulose; 30 mg/kg; oral gavage
Male Sprague-Dawley rats Mouse FST and open field test: - Male ICR mice (20–25 g) were divided into 4 groups (n=8/group): vehicle (0.5% methylcellulose, oral), Desvenlafaxine Succinate 10 mg/kg, 20 mg/kg, 40 mg/kg (oral). Dosing was performed 60 minutes before FST. Mice were placed in a 25 cm diameter tank (25°C water, 15 cm depth) for 6 minutes, and immobility time was recorded during the last 4 minutes. For open field test, mice were placed in a 30×30 cm arena for 30 minutes, and total distance traveled was measured via video tracking [1] - Rat in vivo microdialysis: - Male Sprague-Dawley rats (250–300 g) were anesthetized, and a microdialysis probe was implanted into the PFC. After 24 hours of recovery, Desvenlafaxine Succinate (20 mg/kg, oral) or vehicle was administered. Dialysates were collected every 20 minutes for 4 hours, and 5-HT/NE levels were analyzed via HPLC with electrochemical detection [1] - Rat TST: - Male Wistar rats (220–250 g) were divided into 3 groups (n=6/group): vehicle (saline, i.p.), Desvenlafaxine Succinate 5 mg/kg, 10 mg/kg (i.p.). Dosing was performed 30 minutes before TST. Rats were suspended by the tail (50 cm above the floor) for 6 minutes, and immobility time was recorded during the last 4 minutes [1] |
| ADME/Pharmacokinetics |
Human pharmacokinetics: - Oral administration (50 mg): The peak plasma concentration (Cmax) in healthy volunteers (n=12) was 320 ng/mL, the time to peak concentration (Tmax) was 5.5 hours, the elimination half-life (t₁/₂) was 11 hours, and the oral bioavailability (F) was 80% (food had no effect on absorption) [1][2] - Steady-state: After oral administration of 50 mg daily for 7 consecutive days, the steady-state peak plasma concentration (Cmax) was 640 ng/mL (cumulative 2-fold) [2] - Rat pharmacokinetics: - Oral administration (20 mg/kg): The peak plasma concentration (Cmax) was 450 ng/mL, the time to peak concentration (Tmax) was 2 hours, the elimination half-life (t₁/₂) was 4.5 hours, and F = 75% [1] - Intravenous injection (5 mg/kg: Half-life (t₁/₂) is 4.2 hours, clearance (CL) is 14 mL/min/kg, volume of distribution (Vd) = 4.0 L/kg [1]
- Metabolism: - Desvenlafaxine succinate is primarily metabolized in the liver via glucuronidation (approximately 50%) and CYP3A4-mediated oxidation (approximately 30%); the major metabolites are inactive (e.g., desvenlafaxine O-glucuronide, SERT/NET Ki value >1000 nM) [1] - Excretion: - Approximately 70% of the dose is excreted in the urine within 48 hours (45% as unchanged drug, 25% as metabolites); approximately 15% is excreted in the feces [1] |
| Toxicity/Toxicokinetics |
Acute toxicity: - Oral LD50 in mice = 1200 mg/kg; oral LD50 in rats = 1000 mg/kg. Acute symptoms (500 mg/kg in rats) included transient sedation and decreased activity, which recovered completely within 24 hours [1] - Subacute toxicity (28 days): - Oral administration of desvenlafaxine succinate (10 mg/kg, 30 mg/kg, 100 mg/kg/day) to rats showed no significant changes in body weight, food intake, or serum ALT/AST (liver) and creatinine/BUN (kidney) levels. No lesions were found in the histopathological examination of the brain, liver and kidney tissues [1] - Adverse clinical reactions: - Common side effects (incidence 5%–15%): nausea (10%), dizziness (8%), headache (7%) and dry mouth (5%); all side effects were mild to moderate and subsided within 1–2 weeks after treatment [2] - Plasma protein binding rate: - 30% in human plasma (balanced dialysis) and 28% in rat plasma; the binding rate was not concentration-dependent (10–1000 ng/mL) [1] - Drug interactions: - No significant interactions were observed with paroxetine (CYP2D6 inhibitor) or ketoconazole (CYP3A4 inhibitor) in healthy volunteers; plasma concentration of desvenlafaxine succinate changed by <15% [2]
|
| References | |
| Additional Infomation |
Desvenlafaxine is a cyclohexanol and phenol derivative and a metabolite of venlafaxine. It is a serotonin and norepinephrine reuptake inhibitor (SNRI) used as an antidepressant. See also: Desvenlafaxine succinate (note moved to). Desvenlafaxine succinate (WY 45233; O-desmethylvenlafaxine) is the major active metabolite of venlafaxine, a serotonin-norepinephrine reuptake inhibitor (SNRI) approved for the treatment of major depressive disorder (MDD), generalized anxiety disorder (GAD), and social anxiety disorder (SAD) [1][2].
- Mechanism of action: Succinate desvenlafaxine inhibits the reuptake of serotonin (5-HT) and norepinephrine (NE) mediated by serotonin transporter (SERT) and norepinephrine transporter (NET) in presynaptic neurons. Neurons, increasing the level of extracellular neurotransmitters in the brain—this enhances monoaminergic neurotransmission, thereby exerting its antidepressant and anti-anxiety effects[1] - Clinical advantages compared to venlafaxine: - Longer half-life (11 hours, compared to 3-4 hours for venlafaxine), allowing for once-daily dosing[2] - No significant first-pass metabolism (F=80%, compared to 45% for venlafaxine), reducing individual variability in plasma concentrations[2] - Literature indicates that desenavenlafaxine succinate is classified as a "me too" drug (structure/mechanism similar to existing SNRIs), but provides practical benefits for patient compliance due to the simplified dosing method[2] |
| Molecular Formula |
C20H31NO6
|
|
|---|---|---|
| Molecular Weight |
381.46
|
|
| Exact Mass |
381.215
|
|
| CAS # |
448904-47-0
|
|
| Related CAS # |
Desvenlafaxine; 93413-62-8; Desvenlafaxine succinate hydrate; 386750-22-7; Desvenlafaxine fumarate; 93414-04-1
|
|
| PubChem CID |
9800068
|
|
| Appearance |
Typically exists as solid at room temperature
|
|
| LogP |
2.668
|
|
| Hydrogen Bond Donor Count |
4
|
|
| Hydrogen Bond Acceptor Count |
7
|
|
| Rotatable Bond Count |
7
|
|
| Heavy Atom Count |
27
|
|
| Complexity |
359
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
CN(C)CC(C1=CC=C(C=C1)O)C2(CCCCC2)O.C(CC(=O)O)C(=O)O
|
|
| InChi Key |
ORUUBRMVQCKYHB-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C16H25NO2.C4H6O4/c1-17(2)12-15(13-6-8-14(18)9-7-13)16(19)10-4-3-5-11-16;5-3(6)1-2-4(7)8/h6-9,15,18-19H,3-5,10-12H2,1-2H3;1-2H2,(H,5,6)(H,7,8)
|
|
| Chemical Name |
butanedioic acid;4-[2-(dimethylamino)-1-(1-hydroxycyclohexyl)ethyl]phenol
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
|
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6215 mL | 13.1075 mL | 26.2151 mL | |
| 5 mM | 0.5243 mL | 2.6215 mL | 5.2430 mL | |
| 10 mM | 0.2622 mL | 1.3108 mL | 2.6215 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT00818155 | Completed | Drug: desvenlafaxine succinate SR Other: Placebo |
Healthy | Wyeth is now a wholly owned subsidiary of Pfizer |
January 2009 | Phase 1 |
| NCT01948895 | Completed | Drug: Desvenlafaxine | Dysthymic Disorder | Centre for Addiction and Mental Health |
August 2012 | Not Applicable |
| NCT01309542 | Completed | Drug: Desvenlafaxine Succinate | Major Depressive Disorder | Pfizer | August 2003 | Phase 3 |
| NCT00887224 | Completed | Drug: Placebo Drug: Desvenlafaxine succinate sustained release 50 mg |
Major Depressive Disorder | Pfizer | June 2009 | Phase 3 |
| NCT00683800 | Completed | Drug: Placebo Drug: desvenlafaxine succinate (DVS) SR |
Vasomotor Symptoms | Pfizer | June 2008 | Phase 3 |
|
|---|
|
  |