
| Size | Price | Stock | Qty |
|---|---|---|---|
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| 5g | |||
| Other Sizes |
Purity: ≥98%
Atomoxetine HCl (formerly LY139603; LY-139603; Tomoxetine; Strattera; Tomoxetina; Tomoxetinum), the hydrochloride salt of Atomoxetine, is a potent and selective norepinephrine (NE) transporter/reuptate inhibitor that has been used for treating attention-deficit hyperactivity disorder (ADHD). It has a Ki of 5 nM for norepinephrine (NE) transporter/reuptate inhibition, and it has 290- and 15-fold lower affinity for human 5-HT and DA transporters, respectively. The treatment of ADHD with atomoxetine has received approval.
| Targets |
Norepinephrine (NE) transporter ( Ki = 5 nM ); 5-HT ( Ki = 77 nM )
Norepinephrine transporter (NET) (Ki: 0.8 nM in rat brain membranes, Ki: 0.5 nM in human recombinant NET); no significant binding to serotonin transporter (SERT, Ki > 1000 nM) or dopamine transporter (DAT, Ki > 500 nM) [5] - Voltage-gated sodium channels (VGSC) subtypes: Nav1.2 (IC50: 3.2 μM), Nav1.6 (IC50: 4.5 μM) (state-dependent inhibition); no significant inhibition on Nav1.1 or Nav1.5 (IC50 > 20 μM) [2] |
|---|---|
| ln Vitro |
In vitro activity: Atomoxetine is a selective norepinephrine reuptake inhibitor that binds to serotonin and dopamine transporters at a Ki of 5 nM as opposed to 77 and 1451 nM. [1]
Atomoxetine Hydrochloride is the hydrochloride salt of atomoxetine, a phenoxy-3-propylamine derivative and selective non-stimulant, norepinephrine reuptake inhibitor with cognitive-enhancing activity. Although its precise mechanism of action is unknown, atomoxetine appears to selectively inhibit the pre-synaptic norepinephrine transporter, resulting in inhibition of the presynaptic reabsorption of norepinephrine and prolongation of norepinephrine activity in the synaptic cleft. The effect on cognitive brain function may result in improved attention and decreased impulsivity and activity levels.
NET inhibition in rat brain synaptosomes: Atomoxetine HCl dose-dependently inhibited [³H]norepinephrine (NE) uptake, with an IC50 of 1.2 nM; at 10 nM, it blocked >90% of NE uptake, while having no effect on [³H]serotonin (5-HT) or [³H]dopamine (DA) uptake (uptake assay) [5] - VGSC block in HEK293 cells: - State-dependent inhibition: In HEK293 cells expressing human Nav1.2, Atomoxetine HCl (1–10 μM) showed stronger inhibition of inactivated VGSCs (IC50: 3.2 μM) than resting VGSCs (IC50: 18 μM) (patch-clamp electrophysiology) [2] - Use-dependent inhibition: At 5 μM, Atomoxetine HCl increased inhibition from ~20% (1st stimulus) to ~65% (10th stimulus) in Nav1.6-expressing cells (10 Hz stimulation) [2] - Extracellular neurotransmitter modulation in rat prefrontal cortex (PFC) slices: 1 μM Atomoxetine HCl increased extracellular NE levels by ~250% and DA levels by ~120% (microdialysis coupled with HPLC); no effect on 5-HT levels [3] |
| ln Vivo |
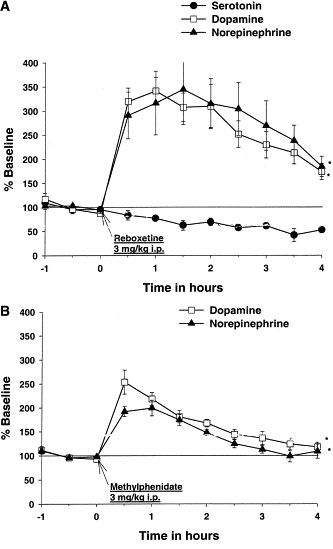
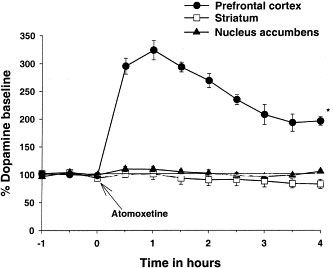
Atomoxetine triples the extracellular (EX) levels of NE in the prefrontal cortex (PFC) in microdialysis studies, but it has no effect on the levels of 5-HTEX. Additionally, atomoxetine triples the concentration of DAEX in PFC while leaving DAEX unchanged in the striatum or nucleus accumbens. Atomoxetine raises Fos in PFC by a factor of 3.7, but not in the nucleus accumbens or striatum.[1] In animal models of depression, atomoxetine exhibits activity by selectively inhibiting the presynaptic uptake of norepinephrine in adrenergic neurons.[2][3]
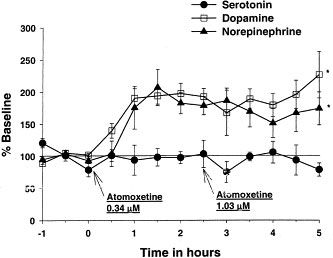
Tomoxetine (0.3-3 mg/kg; i.p.; 0-4 hours; male Sprague-Dawley rats) increases extracellular norepinephrine and dopamine 3-fold and increases intracellular Expression of Fos[1]. Tomoxetine (0.1-5 mg/kg; intraperitoneal injection and oral administration; for 14 days; spontaneously hypertensive rats) can improve ADHD-related behaviors in rats [3].
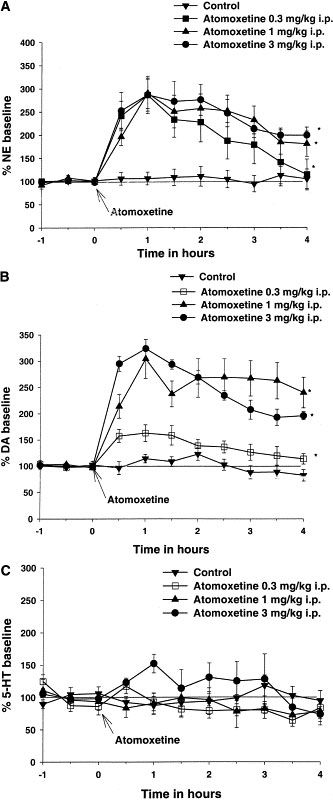
Atomoxetine (ATX) is a commonly used non-stimulant treatment for Attention deficit hyperactivity disorder (ADHD). It primarily acts to increase noradrenalin levels; however, at higher doses it can increase dopamine levels. To date there has been no investigations into the effects of orally-administered ATX in the most commonly used model of ADHD, the spontaneously hypertensive rat (SHR). The aim of this study was to describe the effects of doses thought to be selective (0.15 mg/kg) and non-selective (0.3 mg/kg) for noradrenalin on behavioural measures in the SHR. Firstly, we examined the effects of acute and chronic ATX on locomotor activity including sensitisation and cross-sensitisation to amphetamine. Secondly, we measured drug effects on impulsivity using a T-maze delay discounting paradigm. We found no effect of ATX on locomotor activity and no evidence for sensitisation or cross-sensitisation. Furthermore, there were no differences in T-maze performance, indicating no effects on impulsivity at these doses. The absence of behavioural sensitisation supports previous claims of superior safety relative to psychostimulants for the doses administered. There was also no effect on impulsivity; however, we suggest that was confounded by stress specific to SHRs. Implications for future studies, behavioural assessment of SHRs and their use as a model of ADHD are discussed[1]. Locomotor activity and impulsivity in spontaneously hypertensive rats (SHR, ADHD model) [1]: - Locomotion: Intraperitoneal (i.p.) injection of Atomoxetine HCl at 0.3 mg/kg, 1 mg/kg, and 3 mg/kg reduced locomotor activity in SHR by ~15%, ~35%, and ~50%, respectively (open field test); no effect on locomotion in normotensive Wistar-Kyoto (WKY) rats [1] - Impulsivity: 1 mg/kg and 3 mg/kg Atomoxetine HCl (i.p.) increased correct response rate by ~20% and ~30%, respectively, and reduced impulsive errors by ~25% and ~40% in the 5-choice serial reaction time task (5-CSRTT) [1] - Extracellular NE/DA elevation in rat PFC [3]: - I.p. administration of Atomoxetine HCl at 1 mg/kg, 3 mg/kg, and 10 mg/kg increased extracellular NE levels by ~180%, ~320%, and ~450%, and DA levels by ~90%, ~150%, and ~220%, respectively (in vivo microdialysis); peak effects occurred at 60–90 minutes post-injection [3] - Antidepressant-like activity in mice [4]: - Forced Swim Test (FST): Oral Atomoxetine HCl at 10 mg/kg and 20 mg/kg reduced immobility time by ~30% and ~45%, respectively; no effect on locomotor activity (control for FST) [4] |
| Enzyme Assay |
Atomoxetine, a neuroactive drug, is approved for the treatment of attention-deficit/hyperactivity disorder (ADHD). It is primarily known as a high affinity blocker of the noradrenaline transporter, whereby its application leads to an increased level of the corresponding neurotransmitter in different brain regions. However, the concentrations used to obtain clinical effects are much higher than those which are required to block the transporter system. Thus, off-target effects are likely to occur. In this way, we previously identified atomoxetine as blocker of NMDA receptors. As many psychotropic drugs give rise to sudden death of cardiac origin, we now tested the hypothesis whether atomoxetine also interacts with voltage-gated sodium channels of heart muscle type in clinically relevant concentrations. Electrophysiological experiments were performed by means of the patch-clamp technique at human heart muscle sodium channels (hNav1.5) heterogeneously expressed in human embryonic kidney cells. Atomoxetine inhibited sodium channels in a state- and use-dependent manner. Atomoxetine had only a weak affinity for the resting state of the hNav1.5 (Kr: ∼ 120 µM). The efficacy of atomoxetine strongly increased with membrane depolarization, indicating that the inactivated state is an important target. A hallmark of this drug was its slow interaction. By use of different experimental settings, we concluded that the interaction occurs with the slow inactivated state as well as by slow kinetics with the fast-inactivated state. Half-maximal effective concentrations (2-3 µM) were well within the concentration range found in plasma of treated patients. Atomoxetine also interacted with the open channel. However, the interaction was not fast enough to accelerate the time constant of fast inactivation. Nevertheless, when using the inactivation-deficient hNav1.5_I408W_L409C_A410W mutant, we found that the persistent late current was blocked half maximal at about 3 µM atomoxetine. The interaction most probably occurred via the local anesthetic binding site. Atomoxetine inhibited sodium channels at a similar concentration as it is used for the treatment of ADHD. Due to its slow interaction and by inhibiting the late current, it potentially exerts antiarrhythmic properties[2].
NET binding assay (rat brain membranes) [5]: - Rat cerebral cortex membranes were prepared and mixed with [³H]nisoxetine (NET-selective ligand, final concentration: 1 nM) and Atomoxetine HCl (0.01 nM–100 nM) in binding buffer (50 mM Tris-HCl pH 7.4, 120 mM NaCl, 5 mM KCl, 0.1% BSA). The mixture was incubated at 25°C for 90 minutes, then filtered through glass fiber filters (pre-soaked in 0.5% polyethyleneimine) to separate bound and free ligand. Filters were washed 3 times with ice-cold binding buffer, and radioactivity was measured via liquid scintillation counter. Ki values were calculated using the Cheng-Prusoff equation [5] - VGSC current recording (patch-clamp) [2]: - HEK293 cells were transfected with human Nav1.2 or Nav1.6 cDNA and cultured in DMEM + 10% FBS at 37°C, 5% CO2. Whole-cell patch-clamp recordings were performed using an extracellular solution (140 mM NaCl, 5 mM KCl, 2 mM CaCl2, 1 mM MgCl2, 10 mM HEPES) and intracellular solution (140 mM CsF, 10 mM CsCl, 10 mM HEPES, 1 mM EGTA). Atomoxetine HCl (0.1 μM–30 μM) was added to the extracellular solution, and VGSC currents were evoked by voltage steps (from -120 mV to 0 mV, 50 ms duration). Inactivation curves were generated by pre-pulsing to voltages from -120 mV to 0 mV (10 mV increments) before the test pulse; use-dependent inhibition was assessed via 10 Hz stimulus trains [2] |
| Cell Assay |
The selective norepinephrine (NE) transporter inhibitor atomoxetine (formerly called tomoxetine or LY139603) has been shown to alleviate symptoms in Attention Deficit/Hyperactivity Disorder (ADHD). We investigated the mechanism of action of atomoxetine in ADHD by evaluating the interaction of atomoxetine with monoamine transporters, the effects on extracellular levels of monoamines, and the expression of the neuronal activity marker Fos in brain regions. Atomoxetine inhibited binding of radioligands to clonal cell lines transfected with human NE, serotonin (5-HT) and dopamine (DA) transporters with dissociation constants (K(i)) values of 5, 77 and 1451 nM, respectively, demonstrating selectivity for NE transporters. In microdialysis studies, atomoxetine increased extracellular (EX) levels of NE in prefrontal cortex (PFC) 3-fold, but did not alter 5-HT(EX) levels. Atomoxetine also increased DA(EX) concentrations in PFC 3-fold, but did not alter DA(EX) in striatum or nucleus accumbens. In contrast, the psychostimulant methylphenidate, which is used in ADHD therapy, increased NE(EX) and DA(EX) equally in PFC, but also increased DA(EX) in the striatum and nucleus accumbens to the same level. The expression of the neuronal activity marker Fos was increased 3.7-fold in PFC by atomoxetine administration, but was not increased in the striatum or nucleus accumbens, consistent with the regional distribution of increased DA(EX). We hypothesize that the atomoxetine-induced increase of catecholamines in PFC, a region involved in attention and memory, mediates the therapeutic effects of atomoxetine in ADHD. In contrast to methylphenidate, atomoxetine did not increase DA in striatum or nucleus accumbens, suggesting it would not have motoric or drug abuse liabilities[3].
HEK293 cell VGSC expression and patch-clamp assay (detailed in Enzyme Assay section) [2] - Rat PFC slice microdialysis assay [3]: - Coronal PFC slices (300 μm thick) were prepared from male Sprague-Dawley rats and maintained in oxygenated (95% O2/5% CO2) artificial cerebrospinal fluid (ACSF: 124 mM NaCl, 3 mM KCl, 1.25 mM KH2PO4, 2 mM CaCl2, 1 mM MgSO4, 10 mM glucose, 26 mM NaHCO3) at 32°C. A microdialysis probe (2 mm membrane length) was inserted into the slice, and ACSF was perfused at 1 μL/min. After 60 minutes of equilibration, Atomoxetine HCl (0.1 μM–10 μM) was added to the perfusate. Dialysates were collected every 20 minutes and analyzed for NE, DA, and 5-HT via HPLC with electrochemical detection [3] |
| Animal Protocol |
SHR locomotor and impulsivity studies [1]:
- Male SHR and WKY rats (8–10 weeks old, 250–300 g) were group-housed (4 per cage) under 12 h light/dark cycle. For open field test: Rats were acclimated to the arena (40×40×30 cm) for 30 minutes, then Atomoxetine HCl (dissolved in saline + 0.1% DMSO) was administered i.p. at 0.3 mg/kg, 1 mg/kg, 3 mg/kg (n=8/group). Locomotor activity (total distance traveled) was recorded for 60 minutes post-injection via video tracking. For 5-CSRTT: Rats were trained to respond to light cues for food rewards; after training, Atomoxetine HCl (1 mg/kg, 3 mg/kg i.p.) was administered 30 minutes before testing, and correct responses/impulsive errors were recorded [1] - Rat in vivo microdialysis [3]: - Male Sprague-Dawley rats (250–300 g) were anesthetized with sodium pentobarbital (50 mg/kg i.p.), and a guide cannula was implanted into the medial PFC (coordinates: AP +3.2 mm, ML ±0.8 mm, DV -2.5 mm relative to bregma). After 7 days of recovery, a microdialysis probe (2 mm membrane) was inserted into the guide cannula, and ACSF (95% O2/5% CO2 bubbled) was perfused at 1 μL/min. After 2 hours of equilibration, Atomoxetine HCl (1 mg/kg, 3 mg/kg, 10 mg/kg i.p., n=6/group) was administered, and dialysates were collected every 20 minutes for 4 hours. NE/DA/5-HT levels were quantified via HPLC [3] - Mouse FST [4]: - Male ICR mice (20–25 g) were fasted for 12 hours before testing. Atomoxetine HCl (dissolved in 0.5% methylcellulose) was administered orally at 10 mg/kg, 20 mg/kg (n=10/group) 60 minutes before FST. Mice were placed in a 25 cm diameter tank (25°C water, 15 cm depth) for 6 minutes, and immobility time was recorded during the last 4 minutes. Locomotor activity was measured in an open field (30×30 cm) for 30 minutes post-FST [4] - ADME animal studies [4]: - Rats (male Sprague-Dawley, 250 g) and beagle dogs (male, 10 kg) were administered Atomoxetine HCl via oral gavage (10 mg/kg) or intravenous injection (2 mg/kg). Blood samples were collected at 0.25, 0.5, 1, 2, 4, 6, 8, 12, 24 hours post-dosing. Plasma drug concentrations were measured via HPLC, and PK parameters (t1/2, Cmax, F) were calculated [4] |
| ADME/Pharmacokinetics |
Absorption, Distribution, and Excretion
Absorption The pharmacokinetic characteristics of atormoxetine are highly dependent on individual cytochrome P450 2D6 gene polymorphisms. A significant proportion of the population (up to 10% of Caucasians, 2% of African Americans, and 1% of Asians) are poor metabolizers (PMs) of CYP2D6-metabolized drugs. Compared to individuals with normal CYP2D6 activity, these individuals have reduced activity in this metabolic pathway, resulting in a 10-fold higher AUC, a 5-fold higher peak plasma concentration, and a slower elimination rate (plasma half-life of 21.6 hours). After oral administration, atormoxetine is rapidly absorbed, with an absolute bioavailability of approximately 63% in vigorous metabolizers (EMs) and approximately 94% in poor metabolizers (PMs). The mean maximum plasma concentration (Cmax) is reached approximately 1 to 2 hours after administration, with a maximum concentration of 350 ng/ml and an AUC of 2 mcg·h/ml. Elimination Pathway Atomoxetine is primarily excreted as 4-hydroxyatoromoxetine-O-glucuronide, mainly in the urine (over 80% of the administered dose) and a small amount in the feces (less than 17% of the administered dose). Only a small amount (less than 3%) of atoromoxetine is excreted unchanged, indicating extensive biotransformation. Volume of Distribution The volume of distribution of orally administered atoromoxetine is 1.6–2.6 L/kg. The steady-state volume of distribution of intravenously administered atoromoxetine is approximately 0.85 L/kg. Clearance The clearance of atoromoxetine depends on individual CYP2D6 gene polymorphism and ranges from 0.27–0.67 Lh/kg. Steady-state volume of distribution (intravenous administration): 8.5 L/kg. Atomoxetine is primarily distributed in body fluids; after standardization to body weight, the volume of distribution is similar among patients of different body weights. After oral administration, atormoxetine is rapidly absorbed, with an absolute bioavailability of approximately 63% in fast metabolizers and approximately 94% in slow metabolizers. Maximum plasma concentration (Cmax) is reached approximately 1 to 2 hours after administration. /Breast Milk/ Aatormoxetine and/or its metabolites are distributed into the milk of rats; it is unknown whether the drug is distributed into human breast milk. View More... Aatormoxetine has high water solubility and biomembrane permeability, and is rapidly and completely absorbed after oral administration. Its absolute oral bioavailability ranges from 63% to 94%, mainly affected by the degree of first-pass metabolism. Systemic clearance of atormoxetine involves three oxidative metabolic pathways: aromatic ring hydroxylation, benzylic hydroxylation, and N-demethylation. Aromatic ring hydroxylation of atormoxetine yields its major oxidative metabolite, 4-hydroxyatormoxetine, which is subsequently glucuronidated and excreted in the urine. The formation of 4-hydroxyatormoxetine is primarily mediated by the polymorphically expressed cytochrome P450 (CYP) 2D6 enzyme. This results in two individual groups: those with a high capacity to metabolize atormoxetine (CYP2D6 fast metabolizers) and those with a low capacity to metabolize atormoxetine (CYP2D6 slow metabolizers). Oral bioavailability and clearance of atormoxetine are influenced by CYP2D6 activity; however, plasma pharmacokinetic parameters are predictable for both fast and slow metabolizers. Following a single oral dose, atormoxetine reaches peak plasma concentrations approximately 1–2 hours after administration. In rapid metabolizers, the plasma half-life of atormoxetine was 5.2 hours, while in slow metabolizers it was 21.6 hours. The systemic plasma clearance of atormoxetine in rapid and slow metabolizers was 0.35 L/h/kg and 0.03 L/h/kg, respectively. Correspondingly, the mean steady-state plasma concentration in slow metabolizers was approximately 10 times that in rapid metabolizers. After multiple doses, plasma accumulation of atormoxetine occurred in slow metabolizers, while almost no accumulation occurred in rapid metabolizers. The volume of distribution was 0.85 L/kg, indicating that atormoxetine was distributed in systemic water in both rapid and slow metabolizers. Atomoxetine had a high binding rate to plasma albumin (approximately 99%). Although the steady-state concentration of atormoxetine was higher in slow metabolizers than in rapid metabolizers after the same mg/kg/day dose, the frequency and severity of adverse events were similar regardless of CYP2D6 phenotype. Atomoxetine administration does not inhibit or induce the clearance of other drugs metabolized by CYP enzymes. In rapid metabolizers, potent and selective CYP2D6 inhibitors reduce the clearance of atormoxetine; however, in slow metabolizers, the use of CYP inhibitors has no effect on the steady-state plasma concentration of atormoxetine. PMID:15910008Metabolism/Metabolites Atormoxetine is primarily biotransformed via the cytochrome P450 2D6 (CYP2D6) enzymatic pathway. Plasma concentrations of atormoxetine are higher in individuals with reduced CYP2D6 metabolic pathway activity (also known as slow metabolizers or PM) than in individuals with normal CYP2D6 metabolic pathway activity (also known as rapid metabolizers or EM). For slow metabolizers, the steady-state AUC of atormoxetine is approximately 10 times that of rapid metabolizers, and the Cmax is approximately 5 times that of rapid metabolizers. Regardless of CYP2D6 status, the major oxidative metabolite is 4-hydroxyatormoxetine, which is rapidly glucuroninated. 4-Hydroxyatormoxetine, as an inhibitor of the norepinephrine transporter, has potency comparable to atormoxetine, but its plasma concentration is much lower (1% of atormoxetine concentration in fast metabolizers and 0.1% in slow metabolizers). In individuals lacking CYP2D6 activity, 4-hydroxyatormoxetine remains the major metabolite, but it is generated by several other cytochrome P450 enzymes at a slower rate. Another minor metabolite, N-desmethylatormoxetine, is generated by CYP2C19 and other cytochrome P450 enzymes, but its pharmacological activity is much lower than that of atormoxetine, and its plasma concentration is also lower (5% of atormoxetine concentration in fast metabolizers and 45% in slow metabolizers). Atomoxetine is primarily metabolized via the CYP2D6 enzymatic pathway. Individuals with reduced activity in this pathway (slow metabolizers) have higher plasma concentrations of atormoxetine than individuals with normal activity in this pathway (fast metabolizers). For slow metabolizers, the AUC of atormoxetine is approximately 10 times that of fast metabolizers, and the Css,max is approximately 5 times that of fast metabolizers. Laboratory methods are currently available to identify CYP2D6 slow metabolizers. Regardless of CYP2D6 status, the main oxidative metabolite is 4-hydroxyatormoxetine, which is glucuroninated. 4-Hydroxyatormoxetine is as potent as norepinephrine transporter inhibitor as atormoxetine, but its plasma concentration is much lower (1% of atormoxetine concentration in fast metabolizers (EM) and 0.1% in slow metabolizers (PM)). 4-Hydroxyatormoxetine is primarily produced by CYP2D6, but in slow metabolizers, several other cytochrome P450 enzymes can also produce 4-hydroxyatormoxetine at a slower rate. N-Desmethylatormoxetine is produced by CYP2C19 and other cytochrome P450 enzymes, but its pharmacological activity is much lower than that of atormoxetine, and its plasma concentration is also lower (5% of atormoxetine concentration in the fast metabolizer and 45% in the slow metabolizer). It has been confirmed that polymorphic cytochrome P450 2D6 (CYP2D6) plays an important role in the pharmacokinetics of atormoxetine hydrochloride [(-)-N-methyl-γ-(2-methylphenoxy)amphenicol hydrochloride; LY139603] after single and multiple doses. This study evaluated the effects of CYP2D6 polymorphism on the overall distribution and metabolism of 20 mg of N-desmethylatormoxetine after administration to subjects with CYP2D6 fast metabolizers (EM; n = 4) and slow metabolizers (PM; n = 3) under steady-state conditions. Atomoxetine is well absorbed in the gastrointestinal tract and is primarily eliminated through metabolism, with most radioactive material excreted in the urine. In rapid metabolizers, most of the radioactive dose is eliminated within 24 hours, while in slow metabolizers, most of the dose is eliminated within 72 hours. The biotransformation of atormoxetine is similar in all subjects, involving aromatic ring hydroxylation, benzylic oxidation, and N-demethylation; no CYP2D6 phenotype-specific metabolites were found. The major oxidative metabolite of atormoxetine is 4-hydroxyatormoxetine, which subsequently binds to glucuronic acid to form 4-hydroxyatormoxetine-O-glucuronide. Due to the lack of CYP2D6 activity, the systemic exposure time to radioactive material was prolonged in PM subjects (half-life t1/2 = 62 hours) compared to EM subjects (t1/2 = 18 hours). In EM subjects, atormoxetine (t1/2 = 5 h) and 4-hydroxyatormoxetine-O-glucuronide (t1/2 = 7 h) were the major circulating drugs, while in PM subjects, atormoxetine (t1/2 = 20 h) and N-desmethylatormoxetine (t1/2 = 33 h) were the major circulating drugs. Although differences were observed in the excretion and relative concentration of metabolites, the main difference between EM and PM subjects was the rate of bioconversion of atormoxetine to 4-hydroxyatormoxetine. PMID: 12485958 Atormoxetine is primarily excreted as 4-hydroxyatormoxetine-O-glucuronide, mainly in the urine (more than 80% of the dose) and a small amount in the feces (less than 17% of the dose). Only a small fraction of the Strattera dose is excreted unchanged as atormoxetine (less than 3% of the dose), indicating extensive biotransformation. Atomoxetine is primarily metabolized to 4-hydroxyatormoxetine via the CYP2D6 pathway. 4-Hydroxyatormoxetine is comparable in potency to atormoxetine as a norepinephrine transporter inhibitor, but its plasma concentrations are significantly lower (1% of atormoxetine in fast metabolizers and 0.1% in slow metabolizers). Half-life: 5 hours. Biological half-life: The reported half-life depends on individual CYP2D6 gene polymorphisms and ranges from 3 to 5.6 hours. The plasma elimination half-life in normal (fast metabolizers) metabolizers is approximately 5 hours. Slow metabolizers (7% of whites and 2% of blacks) have much higher plasma drug concentrations, with a plasma elimination half-life of 24 hours. The mean apparent plasma clearance of atormoxetine after oral administration in fast metabolizers in adults is 0.35 L/hr/kg, with a mean half-life of 5.2 hours. In slow metabolizers (PM), the mean apparent plasma clearance is 0.03 L/hr/kg, with a mean half-life of 21.6 hours. The AUC of atormoxetine in slow metabolizers is approximately 10 times that in fast metabolizers, and the Css,max is approximately 5 times that in fast metabolizers. In fast metabolizers, the elimination half-life of 4-hydroxyatormoxetine is similar to that of N-desmethylatormoxetine (6 to 8 hours), while in slow metabolizers, the half-life of N-desmethylatormoxetine is much longer (34 to 40 hours). …In fast metabolizers, the plasma half-life of atormoxetine is 5.2 hours, while in slow metabolizers, the plasma half-life of atormoxetine is 21.6 hours. PMID: 15910008 …… Twenty-one patients with extensive cytochrome P450 2D6 metabolites participated in these single-dose and steady-state pharmacokinetic evaluations. Atormoxetine is rapidly absorbed, reaching peak plasma concentrations 1 to 2 hours after administration. The mean half-life after a single dose and at steady state was 3.12 hours and 3.28 hours, respectively. ... Rat pharmacokinetics[4]: - Oral administration (10 mg/kg): Cmax = 85 ng/mL, Tmax = 1.5 h, t1/2 = 3.2 h, oral bioavailability (F) = 75%, CL = 15 mL/min/kg, Vd = 4.8 L/kg [4] - Intravenous administration (2 mg/kg): Cmax = 62 ng/mL, t1/2 = 2.8 h, CL = 16 mL/min/kg [4] - Canine pharmacokinetics[4]: - Oral administration (10 mg/kg): Cmax = 68 ng/mL, Tmax = 2 h, t1/2 = 4.5 h, F = 68% [4] - Human pharmacokinetics[4]: - Healthy volunteers (n=12) were given oral administration (40 mg): Cmax = 80 ng/mL, Tmax = 1.8 h, t1/2 = 5.2 h, F = 60% (strong metabolizers, normal CYP2D6); t1/2 = 21 h for weak CYP2D6 metabolizers [4] - Metabolism: Atomoxetine hydrochloride is mainly metabolized by CYP2D6 in the human body (accounting for about 70% of the metabolism); the main metabolite is 4-hydroxyatoromoxetine (inactive). In individuals with weak CYP2D6 metabolism, metabolism mainly occurs via CYP1A2 (approximately 40%) and CYP2C19 (approximately 30%) [4] - Excretion: Approximately 80% of the dose is excreted in the urine within 48 hours (10% as the original drug and 70% as metabolites); approximately 15% is excreted in the feces [4] |
| Toxicity/Toxicokinetics |
Toxicity Overview
Identification and Use: Atomoxetine (brand name: Strattera) is indicated for the treatment of attention deficit hyperactivity disorder (ADHD). Human Exposure and Toxicity: In short-term studies in children or adolescents with ADHD, atomoxetine increased the risk of suicidal ideation. Symptoms associated with acute or chronic atomoxetine overdose include gastrointestinal symptoms, somnolence, dizziness, tremor, behavioral abnormalities, hyperactivity, agitation, and signs and symptoms consistent with mild to moderate sympathetic nervous system activation (e.g., tachycardia, elevated blood pressure, dilated pupils, dry mouth). Less commonly, QT interval prolongation and altered mental status, including disorientation and hallucinations, have been reported. Atomoxetine may cause clinically significant hepatotoxicity through metabolic-specific reactions or by inducing autoimmune hepatitis. There have been reports of death resulting from mixed overdose of atomoxetine (Strattera) and at least one other drug. Sudden death, stroke, and myocardial infarction have been reported in children and adults with structural heart abnormalities or other serious heart conditions. Animal studies: The estimated median lethal dose (LD50) of atomoxetine hydrochloride in animals via oral administration is: 25 mg/kg for cats, >37.5 mg/kg for dogs, and 0.190 mg/kg for rats and mice. Prodromal symptoms of toxicity following a single oral dose of atomoxetine in animals include mydriasis and decreased pupillary light reflex, mucus in stool, salivation, vomiting, ataxia, tremor, myoclonus, and convulsions. Chronic toxicity studies were conducted in adult rats and dogs for up to one year. No major target organ toxicity was observed in dogs at oral doses up to 16 mg/kg/day, or in rats with atomoxetine supplementation in their diet at a time-weighted average dose up to 47 mg/kg/day. These doses are 4–5 times the maximum recommended daily oral dose for adults. Mild hepatotoxicity was observed in male rats at a time-weighted average dose ≥ 14 mg/kg/day, manifested as mottled pallor of the liver, increased relative liver weight, hepatocyte vacuolation, and a slight increase in serum ALT levels. No hepatotoxicity was observed in dogs. Dogs exhibited clinical symptoms such as dilated pupils, decreased pupillary light reflex, vomiting, and tremors, but these symptoms were most mild at daily doses ≥ 8 mg/kg in adult dogs. During organogenesis, oral administration of atormoxetine hydrochloride to rabbits and rats at doses up to 100 mg/kg/day and 150 mg/kg/day (13 times the maximum recommended daily oral dose for adults) did not reveal evidence of drug-related teratogenicity or fetal growth retardation. In a rat fertility study, decreased pup weight and survival were observed, primarily occurring in the first week postpartum, following a time-weighted average dose of atormoxetine ingested by the mother of 23 mg/kg/day or higher. Atomoxetine hydrochloride showed negative results in a series of genotoxicity studies, including the Ames test (reverse point mutation assay), in vitro mouse lymphoma assay, Chinese hamster ovary cell chromosomal aberration assay, rat hepatocyte unplanned DNA synthesis assay, and mouse in vivo micronucleus assay. However, a slight increase in the proportion of bichromosomes was observed in Chinese hamster ovary cells, suggesting internal duplication (chromosomal number abnormality). In rats and mice, no carcinogenicity was observed after two consecutive years of dietary administration of atormoxetine hydrochloride at a weighted average dose of up to 47 and 458 mg/kg/day, respectively. The exact mechanism of action of atormoxetine in treating attention deficit hyperactivity disorder (ADHD) is unclear, but in vitro studies suggest its action may be related to selective inhibition of the presynaptic norepinephrine transporter. Atormoxetine appears to have low affinity for other norepinephrine receptors or other neurotransmitter transporters or receptors. Hepatotoxicity: A small percentage of patients (approximately 0.5%) have experienced elevated serum transaminases after taking atormoxetine. More importantly, several cases of clinically significant acute liver injury due to atormoxetine use have been reported. The injury typically occurs within 3 to 12 weeks of starting treatment. Typical serum enzyme elevations are hepatocellular, with significantly elevated serum transaminase levels (usually exceeding 20 times the upper limit of normal), clinically resembling acute viral hepatitis. Most cases are self-limiting, but there have been reports of acute liver failure, sometimes requiring emergency liver transplantation. No immune hypersensitivity features were observed, but several patients with acute liver injury had antinuclear antibodies, and at least one patient had other features similar to autoimmune hepatitis (characteristic liver histology and high levels of immunoglobulins in serum). Probability Score: C (Possibly the cause of clinically significant liver injury). View MoreEffects during Pregnancy and Lactation◉ Overview of Use During Lactation There is currently no publicly available experience with the use of atormoxetine during lactation, but the manufacturer reports that no serious adverse reactions occurred in two breastfed infants. If a mother of an older infant needs to take atormoxetine, this is not a reason to stop breastfeeding, but until more data is available, it is recommended to prioritize other medications, especially when breastfeeding newborns or premature infants. Breastfed infants should be monitored for excessive sedation. ◉ Effects on Breastfed Infants A review article reported that Eli Lilly and Co., the manufacturer of atormoxetine, received two reports that two infants slept longer than usual after their mothers took atormoxetine and breastfed. Neither infant experienced any serious adverse events. The article did not provide information on dosage, duration of maternal use, infant age, or extent of breastfeeding. ◉ Effects on Lactation and Breast Milk No relevant published information was found as of the revision date. ◈ What is Atormoxetine? Atormoxetine is a medication approved for the treatment of attention deficit hyperactivity disorder (ADHD). It belongs to the norepinephrine reuptake inhibitor class of drugs. Atomoxetine is marketed as Strattera®. Sometimes, when people find out they are pregnant, they consider changing how they take the medication or stopping it altogether. However, it is essential to talk to your healthcare provider before changing how you take this medication. Your healthcare provider can discuss with you the benefits of treating your condition and the risks of not treating it during pregnancy. ◈ I am taking atormoxetine, and my doctor told me I have a slow metabolism. What does this mean for my pregnancy? Some people metabolize atormoxetine more slowly than others. People with slower metabolisms may have higher concentrations of the drug in their blood. It is currently unclear whether this will have a different effect on pregnancy compared to people who metabolize the drug more quickly. ◈ I am taking atormoxetine. Will it make it harder for me to get pregnant? There are currently no human studies to determine whether atormoxetine makes it harder to get pregnant. Animal studies have not shown any changes in fertility. ◈ Does taking atormoxetine increase the risk of miscarriage? Miscarriage is common and can occur in any pregnancy for many reasons. There are currently no studies confirming whether atormoxetine increases the risk of miscarriage. ◈ Does taking atormoxetine increase the risk of birth defects? There is a 3-5% risk of birth defects in every pregnancy, known as background risk. Research on the use of atormoxetine during pregnancy is insufficient. Four human studies did not find an increased risk of birth defects. Most of these studies used prescription databases to find patients who had taken atormoxetine during pregnancy. However, this cannot determine whether a patient took atormoxetine during pregnancy. Animal studies showed that at commonly used human doses, atormoxetine does not increase the risk of birth defects. However, if the dose of atormoxetine is higher than the human therapeutic dose, there is an increased risk of birth defects. It is currently unclear whether this information applies to individuals with lower metabolic capacity. ◈ Does taking atormoxetine during pregnancy increase the risk of other pregnancy-related problems? It is currently unclear whether atormoxetine causes other pregnancy-related problems. A study of 453 pregnant women who took atormoxetine during the first 20 weeks of pregnancy showed that atormoxetine did not increase the risk of placental abruption (the placenta detaching from the uterine wall before the onset of labor), small for gestational age, preterm birth (delivery before 37 weeks of gestation), or preeclampsia (dangerous high blood pressure). ◈ Does taking atormoxetine during pregnancy cause long-term behavioral or learning problems in children? There are currently no studies confirming whether atormoxetine causes behavioral or learning problems in children. ◈ Breastfeeding while taking atormoxetine: There are currently no studies on taking atormoxetine while breastfeeding. If you are breastfeeding and taking this medication, and you suspect your infant is experiencing any symptoms (such as excessive sedation), contact your child's healthcare provider. Be sure to consult your healthcare provider about all breastfeeding-related questions. ◈ If men take atormoxetine, does it affect fertility (the ability to impregnate a partner) or increase the risk of birth defects in the fetus? Currently, no studies have explored whether atormoxetine affects male fertility or increases the risk of birth defects in the fetus. Generally, exposure to the drug by the father or sperm donor is unlikely to increase the risk of pregnancy. For more information, please refer to the "Paternal Drug Exposure" information sheet on the MotherToBaby website: https://mothertobaby.org/fact-sheets/paternal-exposures-pregnancy/. Route of Exposure Atormoxetine is rapidly absorbed after oral administration, with an absolute bioavailability of approximately 63% for rapid metabolizers (EM) and approximately 94% for slow metabolizers (PM). Drugs that increase gastric pH (such as magnesium hydroxide/aluminum hydroxide, omeprazole) do not affect the bioavailability of atormoxetine. Food has little effect on absorption. Symptoms The most common symptoms of acute and chronic overdose include drowsiness, agitation, hyperactivity, behavioral abnormalities, and gastrointestinal symptoms. Treatment Maintain a clear airway. Monitoring of heart rate and vital signs is recommended, along with appropriate symptomatic and supportive care. Gastric lavage performed shortly after administration may be effective. Activated charcoal helps limit drug absorption. Due to the high protein binding of atormoxetine, dialysis may be ineffective in treating overdose. (L1712) L1712: RxList: The Internet Drug Index (2009). http://www.rxlist.com/ Drug Interactions Atomoxetine should be used with caution in patients receiving systemic (oral or intravenous) salbutamol (or other β2-receptor agonists) therapy, as salbutamol may enhance its cardiovascular effects, leading to increased heart rate and blood pressure. Salbutamol (600 mcg, intravenously, over 2 hours) can cause increased heart rate and blood pressure. Atomoxetine (60 mg, twice daily for 5 days) can enhance these effects, and is most pronounced after the first combined use of salbutamol and atormoxetine. However, in another study, no such effects on heart rate and blood pressure were observed in 21 healthy Asian subjects who were excluded due to poor metabolic capacity after combined use of inhaled doses of salbutamol (200-800 mcg) and atormoxetine (80 mg, once daily for 5 days). The manufacturer states that atormoxetine is contraindicated in patients currently receiving or recently (within two weeks) receiving monoamine oxidase (MAO) inhibitors. Furthermore, at least two weeks should elapse after discontinuing atormoxetine before initiating MAO inhibitors. Severe and potentially fatal reactions (including high fever, muscle rigidity, myoclonus, autonomic dysfunction with rapid fluctuations in vital signs, and altered mental status, such as extreme agitation, which may develop into delirium and coma) have been reported in patients concurrently taking other medications that affect monoamine concentrations in the brain. Protein binding At therapeutic concentrations, 98.7% of plasma atormoxetine is bound to proteins, of which 97.5% is bound to albumin, followed by α-1-acid glycoprotein and immunoglobulin G. Acute toxicity[4]: - Oral LD50 in mice = 350 mg/kg; oral LD50 in rats = 280 mg/kg. Acute toxicity symptoms included sedation, ataxia and decreased activity, which were completely resolved within 24 hours at doses below the LD50[4]. - Subacute toxicity in rats (28 days, oral 10 mg/kg, 30 mg/kg): No significant changes were observed in body weight, food intake or hematological parameters (white blood cells, red blood cells, platelets). Serum ALT/AST levels were slightly elevated (approximately 15%) at a dose of 30 mg/kg, but remained within the normal range; no histopathological lesions were observed in the liver/kidneys [4] - Plasma protein binding: 98% in human plasma (balanced dialysis), 97% in rat plasma, and 96% in canine plasma; binding was not affected by pH (6.5–8.0) or plasma concentration (10–1000 ng/mL) [4] - Drug interactions [4]: - Co-administration with CYP2D6 inhibitors (e.g., paroxetine) increased plasma concentrations of atormoxetine hydrochloride by approximately 3-fold in fast metabolizers [4] |
| References |
|
| Additional Infomation |
Atomoxetine hydrochloride is the hydrochloride salt of atormoxetine. It is an antidepressant and an adrenergic reuptake inhibitor. It contains atormoxetine. Atomoxetine hydrochloride is the hydrochloride salt of atormoxetine, a phenoxy-3-propylamine derivative, a selective non-excitatory norepinephrine reuptake inhibitor with cognitive-enhancing effects. Although its exact mechanism of action is not fully understood, atormoxetine appears to selectively inhibit presynaptic norepinephrine transporters, thereby inhibiting the reuptake of presynaptic norepinephrine and prolonging the activity of norepinephrine in the synaptic cleft. Effects on cognitive brain function may result in improved attention, impulsivity, and reduced activity levels. A propylamine derivative, a selective adrenergic reuptake inhibitor, used to treat attention deficit hyperactivity disorder. See also: Atomoxetine (containing the active ingredient).
Drug Indications Treatment of Attention Deficit Hyperactivity Disorder (ADHD) Atomoxetine hydrochloride (formerly known as atomoxetine) is a selective norepinephrine reuptake inhibitor (SNRI) that was initially developed as an antidepressant but was later approved for the treatment of attention deficit/hyperactivity disorder (ADHD) in children, adolescents and adults [1][3] -Mechanism of action in ADHD: By inhibiting NET, Atomoxetine hydrochloride increases extracellular NE levels in the prefrontal cortex (PFC), thereby enhancing attention. Executive functions mediated by the prefrontal cortex (PFC) (attention, impulse control, working memory) are also affected. It can also moderately increase dopamine (DA) levels in the PFC (through norepinephrine (NE)-induced dopamine transporter (DAT) inhibition), which helps improve the efficacy of attention deficit hyperactivity disorder (ADHD) [3] - Clinical dosage: The recommended oral dose for ADHD is 0.5–1.4 mg/kg/day (once daily or twice daily); dose adjustment is required for those with weaker CYP2D6 metabolism (maximum dose 40 mg/day) [4] - Literature [2] points out a new off-target effect: state/use-dependent voltage-gated sodium channel (VGSC) inhibition, which may lead to mild side effects (e.g., dizziness, palpitations) but is not directly related to the efficacy of ADHD [2] - Unlike stimulant ADHD drugs (e.g. methylphenidate), atormoxetine hydrochloride has no abuse potential (because it does not inhibit DAT) and is listed as a non-controlled substance [3][4] |
| Molecular Formula |
C17H22CLNO
|
|
|---|---|---|
| Molecular Weight |
291.82
|
|
| Exact Mass |
291.138
|
|
| Elemental Analysis |
C, 69.97; H, 7.60; Cl, 12.15; N, 4.80; O, 5.48
|
|
| CAS # |
82248-59-7
|
|
| Related CAS # |
Atomoxetine; 83015-26-3; Atomoxetine-d5 hydrochloride
|
|
| PubChem CID |
54840
|
|
| Appearance |
White to off-white solid powder
|
|
| Boiling Point |
389ºC at 760 mmHg
|
|
| Melting Point |
167-169ºC
|
|
| Flash Point |
164.1ºC
|
|
| LogP |
4.917
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
2
|
|
| Rotatable Bond Count |
6
|
|
| Heavy Atom Count |
20
|
|
| Complexity |
237
|
|
| Defined Atom Stereocenter Count |
1
|
|
| SMILES |
[C@H](C1C=CC=CC=1)(CCNC)OC1C=CC=CC=1C.Cl
|
|
| InChi Key |
LUCXVPAZUDVVBT-UNTBIKODSA-N
|
|
| InChi Code |
InChI=1S/C17H21NO.ClH/c1-14-8-6-7-11-16(14)19-17(12-13-18-2)15-9-4-3-5-10-15;/h3-11,17-18H,12-13H2,1-2H3;1H/t17-;/m1./s1
|
|
| Chemical Name |
(3R)-N-methyl-3-(2-methylphenoxy)-3-phenylpropan-1-amine;hydrochloride
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.57 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.57 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (8.57 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 8.33 mg/mL (28.54 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication (<60°C). |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4268 mL | 17.1338 mL | 34.2677 mL | |
| 5 mM | 0.6854 mL | 3.4268 mL | 6.8535 mL | |
| 10 mM | 0.3427 mL | 1.7134 mL | 3.4268 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Comparison of Lisdexamfetamine Dimesylate With Atomoxetine HCl in Attention-Deficit/Hyperactivity Disorder (ADHD) Subjects With an Inadequate Response to Methylphenidate
CTID: NCT01106430
Phase: Phase 3 Status: Completed
Date: 2021-06-11
 |
|---|
  |
 |