
| Size | Price | Stock | Qty |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g | |||
| 5g | |||
| Other Sizes |
Purity: ≥98%
Asenapine maleate (Org5222; Org-5222; HSDB-8061; HSDB8061; Saphris and Sycrest), an atypical antipsychotic, is a potent and high-affinity antagonist of serotonin, norepinephrine, dopamine and histamine receptors. Asenapine has been authorized for use as an atypical antipsychotic in the treatment of acute mania brought on by bipolar disorders and schizophrenia. Based on initial data, it appears to have negligible side effects related to anticholinergic and cardiovascular systems, along with negligible weight gain. The FDA gave it their approval in August 2009.
| Targets |
sPLA2 ( Ki = 2.5 nM ); 5-HT2A Receptor ( Ki = 0.06 nM ); 5-HT2C Receptor ( Ki = 0.03 nM ); 5-HT7 Receptor ( Ki = 0.13 nM ); D2 Receptor ( Ki = 1.3 nM ); D3 Receptor ( Ki = 0.42 nM ); D4 Receptor ( Ki = 1.1 nM )
Dopamine D2 receptor (Ki = 0.4 nM); Dopamine D3 receptor (Ki = 0.8 nM) [2] Serotonin 5-HT1A receptor (Ki = 2.6 nM); 5-HT2A receptor (Ki = 0.1 nM); 5-HT2B receptor (Ki = 0.5 nM); 5-HT2C receptor (Ki = 0.2 nM); 5-HT6 receptor (Ki = 0.6 nM); 5-HT7 receptor (Ki = 0.9 nM) [2] Adrenergic α1 receptor (Ki = 3.2 nM); α2 receptor (Ki = 4.8 nM) [2] Histamine H1 receptor (Ki = 1.3 nM) [2] |
|---|---|
| ln Vitro |
Asenapine has a higher affinity for 5-HT2C, 5-HT2A, 5-HT2B, 5-HT7, 5-HT6, α2B, and D3 receptors than it does for D2 receptors. This suggests that these targets will be more strongly engaged at therapeutic doses. At 5-HT1A (7.4), 5-HT1B (8.1), 5-HT2A (9.0), 5-HT2B (9.3), 5-HT2C (9.0), 5-HT6 (8.0), 5-HT7 (8.5), D2 (9.1), D3 (9.1), α2A (7.3), α2B (8.3), α2C (6.8), and H1 (8.4) receptors, asenapine exhibits potent antagonist (pKB) behavior[2].
Asenapine maleate (Org 5222 maleate) exhibited high-affinity binding to multiple central nervous system receptors. It acted as a competitive antagonist at dopamine D2/D3 receptors, inhibiting dopamine-induced intracellular calcium mobilization with an IC50 of 0.7 nM for D2 [2] At serotonin receptors, it functioned as a partial agonist at 5-HT1A (EC50 = 3.1 nM) and a potent antagonist at 5-HT2A/2B/2C/6/7 receptors, blocking 5-HT-induced receptor activation by >80% at 1 μM [2] It inhibited voltage-gated calcium channels in cultured cortical neurons (IC50 = 5.2 μM), reducing calcium influx and subsequent neuronal excitability [2] No significant affinity for dopamine D1/D4/D5, serotonin 5-HT3, or GABA receptors was observed at concentrations up to 10 μM [2] |
| ln Vivo |
Asenapine is an atypical antipsychotic that is being used to treat bipolar I disorder and schizophrenia. Compared to other agents, asenapine may have a better therapeutic effect on anxiety symptoms in rats[3]. In the defensive marble burying test and the EPM, asenapine exhibits anxiolytic-like effects in mice[4].
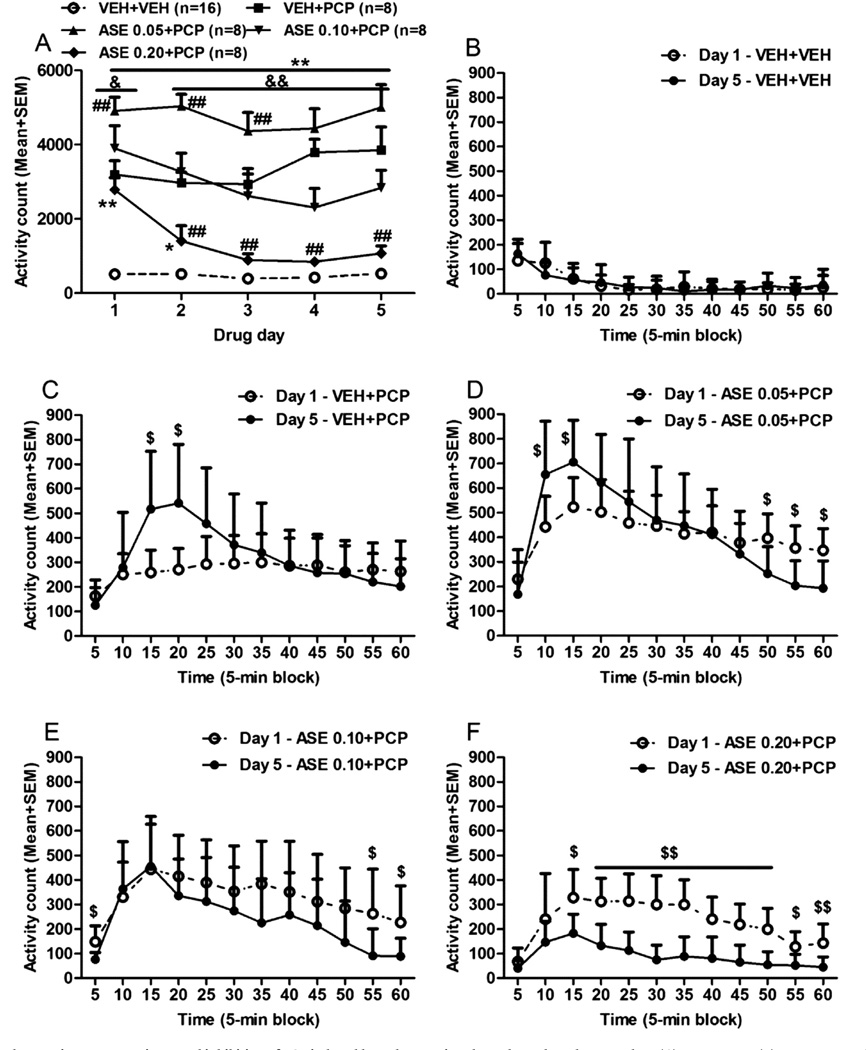
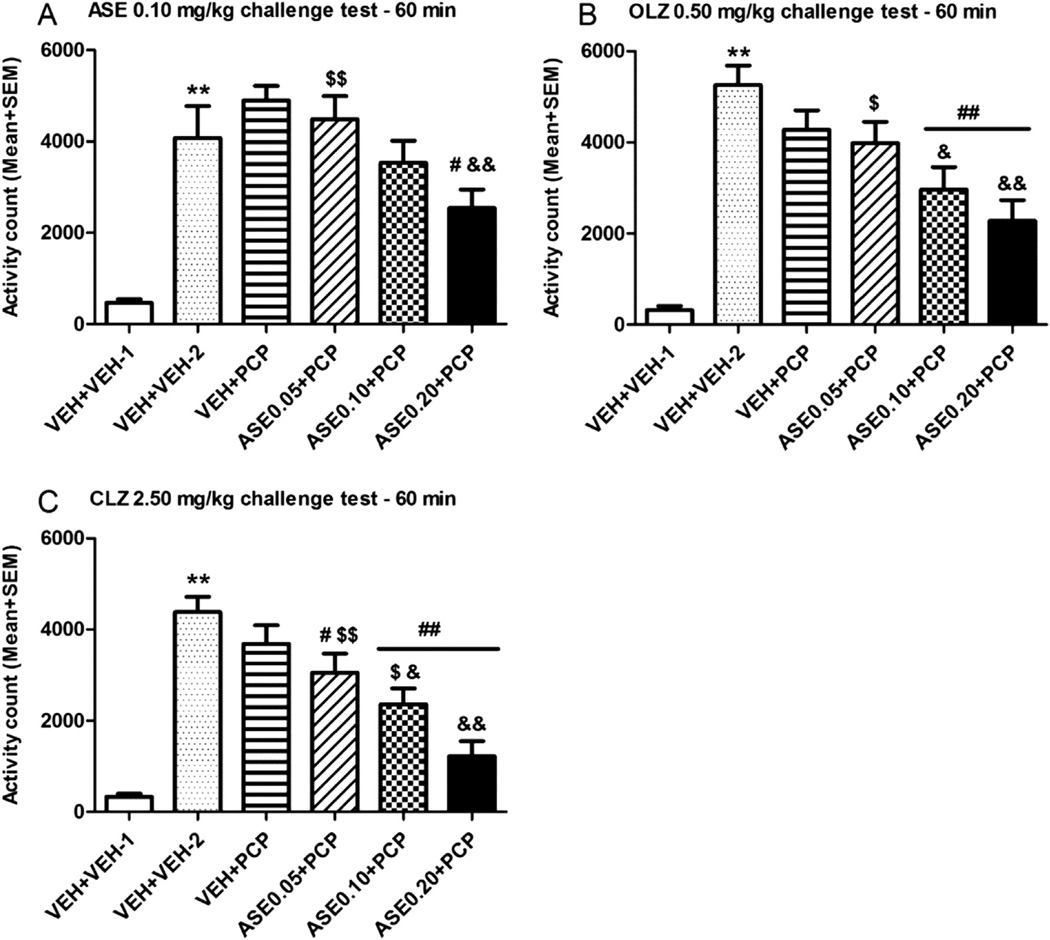
In rats subjected to conditioned fear stress (CFS) model, subcutaneous administration of Asenapine maleate (Org 5222 maleate) (0.3, 1, 3 mg/kg) dose-dependently reduced anxiety-related freezing behavior. At 3 mg/kg, freezing time was decreased by ~45% compared to vehicle, with effects lasting up to 6 hours. It also normalized CFS-induced increases in corticosterone levels [3] In mice, repeated oral administration of Asenapine maleate (Org 5222 maleate) (0.1, 0.3, 1 mg/kg/day for 7 days) exhibited anxiolytic-like effects in multiple tests. In the elevated plus-maze, it increased open arm entries by ~30% and time spent in open arms by ~35% at 1 mg/kg. In the light-dark box test, it increased time in the light compartment by ~40%, and in the marble-burying test, it reduced burying behavior by ~25% [4] In preclinical models of schizophrenia (e.g., PCP-induced hyperlocomotion), Asenapine maleate (Org 5222 maleate) (0.5-2 mg/kg, sc) dose-dependently reversed hyperlocomotion, with an ED50 of ~0.8 mg/kg [1] |
| Enzyme Assay |
Asenapine has a higher affinity for 5-HT2C, 5-HT2A, 5-HT2B, 5-HT7, 5-HT6, α2B, and D3 receptors than it does for D2 receptors. This suggests that these targets will be more strongly engaged at therapeutic doses. At 5-HT1A (7.4), 5-HT1B (8.1), 5-HT2A (9.0), 5-HT2B (9.3), 5-HT2C (9.0), 5-HT6 (8.0), 5-HT7 (8.5), D2 (9.1), D3 (9.1), α2A (7.3), α2B (8.3), α2C (6.8), and H1 (8.4) receptors, asenapine exhibits potent antagonist (pKB) behavior.
Radioligand binding assay for central receptors: Prepare membrane homogenates from human embryonic kidney (HEK) cells transfected with target receptors (D2, 5-HT2A, etc.) or rat brain tissues. Incubate homogenates with subtype-specific [3H]-labeled ligands and various concentrations of Asenapine maleate (Org 5222 maleate) (0.01-100 nM) at 25°C for 90 minutes. Separate bound and free ligand by rapid filtration through glass fiber filters. Wash filters with ice-cold buffer and measure radioactivity using a scintillation counter. Calculate Ki values from competition binding curves [2] Calcium mobilization assay for D2 receptor function: Seed HEK-D2 cells in 96-well plates and culture until confluent. Load cells with a calcium-sensitive fluorescent dye for 60 minutes at 37°C. Preincubate cells with Asenapine maleate (Org 5222 maleate) (0.1-10 nM) for 30 minutes, then stimulate with dopamine (10 μM). Record fluorescent intensity changes in real time using a microplate reader and calculate IC50 [2] |
| Cell Assay |
Cortical neuron calcium influx assay: Isolate cortical neurons from embryonic rat brains, seed in poly-D-lysine-coated plates, and culture in neurobasal medium for 7-10 days. Load neurons with a calcium-sensitive fluorescent dye for 45 minutes at 37°C. Treat neurons with Asenapine maleate (Org 5222 maleate) (1-10 μM) for 20 minutes, then stimulate with potassium chloride (50 mM) to induce calcium influx. Record fluorescent intensity using a confocal microscope and quantify calcium concentration changes [2]
|
| Animal Protocol |
Mice: 0.1 or 0.3mg/kg; Rats: 1 mL/kg, injections Rats: Asenapine maleate is given in a volume of 1 mL/kg body weight, suspended in 10% hydroxypropyl-β-cyclodextrin. Rats are individually trained to fear by putting them in a Skinner box with an electrical foot shock. Thirty minutes prior to the assessment of freezing behavior, animals receive an intraperitoneal (i.p.) injection of asenapine, clozapine, olanzapine, buspirone, or SB242084. Mice: The male ICR mice undergo repeated injections of asenapine at doses of 0.1 or 0.3 mg/kg. Following treatment, the mice are subjected to a battery of anxiety-related behavioral tests, such as the elevated plus-maze (EPM), defensive marble burying, open-field test, and hyponeophagia test.
Rat conditioned fear stress (CFS) model: Adult male rats are subjected to paired tone-shock conditioning (tone: 80 dB, 20 seconds; shock: 0.8 mA, 2 seconds) for 5 days to establish the CFS model. One day after conditioning, rats are randomly divided into vehicle and treatment groups. Asenapine maleate (Org 5222 maleate) is dissolved in saline and administered subcutaneously at 0.3, 1, or 3 mg/kg 30 minutes before the fear recall test. Freezing behavior (immobility) is recorded for 10 minutes during tone presentation, and blood samples are collected to measure corticosterone levels [3] Mouse anxiety test battery: Adult male mice are randomly assigned to vehicle and treatment groups. Asenapine maleate (Org 5222 maleate) is suspended in 0.5% methylcellulose and administered orally at 0.1, 0.3, or 1 mg/kg/day for 7 consecutive days. On day 8, mice are subjected to elevated plus-maze (5-minute test, record open/closed arm entries and time), light-dark box (10-minute test, record light compartment time), and marble-burying test (30-minute test, record number of buried marbles) [4] |
| ADME/Pharmacokinetics |
Oral absorption: Asenapine maleate (Org 5222 maleate) has low oral bioavailability (about 1%) due to extensive first-pass metabolism, but its bioavailability can reach about 35% when administered sublingually[1]. Distribution: The drug is widely distributed in tissues, with a volume of distribution (Vdss) of about 20 L/kg in the human body. It has good brain permeability, with a brain/plasma concentration ratio of about 2.3[1]. Metabolism: The drug is mainly metabolized in the liver by cytochrome P450 1A2 and 3A4, producing inactive metabolites (e.g., N-desmethylasenapine)[1]. Excretion: The elimination half-life (t1/2) in the human body is about 24 hours. Approximately 70% of the dose is excreted in feces, 20% in urine, and less than 5% is excreted unchanged.[1]
Plasma protein binding rate:Asenapine maleate (Org 5222 maleate) has a plasma protein binding rate of approximately 95% in the human body.[1] |
| Toxicity/Toxicokinetics |
Common adverse reactions in humans include dry mouth, dizziness, drowsiness, and weight gain, which are mild to moderate and dose-dependent [1]. Subchronic toxicity studies in rats (28 days) at oral doses of 1, 5, and 20 mg/kg/day showed no significant hepatotoxicity or nephrotoxicity. Mild sedation was observed at high doses (20 mg/kg/day) [1]. It inhibits cytochrome P450 2D6, potentially increasing plasma concentrations of 2D6 substrates such as fluoxetine [1].
|
| References |
|
| Additional Infomation |
(S,S)-Asenapine maleate is a maleate formed by the combination of equimolar amounts of (S,S)-asenapine and maleic acid. It contains (S,S)-asenapine (1+). It is the enantiomer of (R,R)-asenapine maleate. See also: Asenapine (with active moiety).
Drug indications Sycrest is indicated for the treatment of moderate to severe manic episodes associated with bipolar I disorder in adults. Treatment of bipolar I disorder Asenapine maleate (Org 5222 maleate) is a second-generation (atypical) antipsychotic with unique multi-receptor binding properties [1][2]. Its mechanism of action includes balancing central neurotransmission by antagonizing dopamine D2/D3 and serotonin 5-HT2A/2C receptors and partially activating 5-HT1A receptors, thereby exerting antipsychotic and anxiolytic effects[2][3]. Clinical indications: For the treatment of adult schizophrenia and bipolar I disorder (manic or mixed episodes). [1] The drug has a rapid onset of action (within 1-2 weeks) and is effective for both positive and negative symptoms of schizophrenia. [1] Anxiety-like effects observed in preclinical models suggest that the drug may have potential application value in the treatment of mental illness combined with anxiety. [3][4] |
| Molecular Formula |
C21H20CLNO5
|
|---|---|
| Molecular Weight |
401.84
|
| Exact Mass |
401.103
|
| Elemental Analysis |
C, 62.77; H, 5.02; Cl, 8.82; N, 3.49; O, 19.91
|
| CAS # |
85650-56-2
|
| Related CAS # |
Asenapine; 65576-45-6; Asenapine hydrochloride; 1412458-61-7
|
| PubChem CID |
6917875
|
| Appearance |
White to off-white solid powder
|
| Boiling Point |
357.9ºC at 760 mmHg
|
| Melting Point |
141-145°
|
| Flash Point |
170.2ºC
|
| LogP |
3.908
|
| Hydrogen Bond Donor Count |
2
|
| Hydrogen Bond Acceptor Count |
6
|
| Rotatable Bond Count |
2
|
| Heavy Atom Count |
28
|
| Complexity |
482
|
| Defined Atom Stereocenter Count |
2
|
| SMILES |
ClC1C([H])=C([H])C2=C(C=1[H])[C@@]1([H])C([H])([H])N(C([H])([H])[H])C([H])([H])[C@]1([H])C1=C([H])C([H])=C([H])C([H])=C1O2
|
| InChi Key |
GMDCDXMAFMEDAG-CHHFXETESA-N
|
| InChi Code |
InChI=1S/C17H16ClNO.C4H4O4/c1-19-9-14-12-4-2-3-5-16(12)20-17-7-6-11(18)8-13(17)15(14)10-19;5-3(6)1-2-4(7)8/h2-8,14-15H,9-10H2,1H3;1-2H,(H,5,6)(H,7,8)/b;2-1-/t14-,15-;/m1./s1
|
| Chemical Name |
(Z)-but-2-enedioic acid;(2S,6S)-9-chloro-4-methyl-13-oxa-4-azatetracyclo[12.4.0.02,6.07,12]octadeca-1(18),7(12),8,10,14,16-hexaene
|
| Synonyms |
HSDB 8061; Org5222; Org-5222; HSDB-8061; Org 5222; HSDB 8061; Org 5222; HSDB8061; Asenapine; Asenapine maleate; trade names: Saphris; Sycrest
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.22 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.22 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (6.22 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 1.2 mg/mL (2.99 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4886 mL | 12.4428 mL | 24.8855 mL | |
| 5 mM | 0.4977 mL | 2.4886 mL | 4.9771 mL | |
| 10 mM | 0.2489 mL | 1.2443 mL | 2.4886 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT01460290 | Completed | Drug: Asenapine | Bipolar Disorder | University Hospitals Cleveland Medical Center |
October 2011 | Phase 4 |
| NCT01400113 | Completed | Drug: Asenapine Drug: Placebo |
Radiation Injuries | Unity Health Toronto | April 2012 | Phase 4 |
| NCT01349907 | Completed | Drug: Asenapine Drug: Rescue medication |
Bipolar Disorder | Organon and Co | June 16, 2011 | Phase 3 |
| NCT01395992 | Completed | Drug: asenapine | Bipolar I Disorder | Forest Laboratories | April 2012 | Phase 3 |
| NCT01396291 | Completed | Drug: asenapine Drug: placebo |
Bipolar 1 Disorder | Forest Laboratories | December 2011 | Phase 3 |
|
|---|
 |
 |