
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| Other Sizes |
Purity: ≥98%
Sotorasib (AMG-510; AMG510; Lumakras; Lumykras) is a novel, first-in-class and covalent/irreversible inhibitor of KRAS G12C that has been approved by FDA on 5/28/2021 to treat non-small-cell lung cancer (NSCLC). It specifically targets the most common mutation among the three subtypes of Ras proteins (KRas, NRas, and HRas), the KRAS G12C mutation. KRAS gene mutations (e.g. G12C, G12V, G12D, and G13D) are present in approximately 30% of human cancers, and are most common in pancreatic cancer, lung adenocarcinoma, colorectal cancer, gall bladder cancer, thyroid cancer, and bile duct cancer. About 25% of NSCLC patients also have KRAS mutations, and some research suggests that these mutations are associated with a poorer prognosis for NSCLC patients. It has recently been discovered that V-Ki-ras2 Kirsten rat sarcoma viral oncogene homolog (KRAS) mutations confer resistance to epidermal growth factor receptor (EGFR) targeted therapies in colorectal cancer; as such, knowledge of the KRAS mutational status can be crucial before TKI therapy is prescribed. All things considered, patients diagnosed with lung adenocarcinoma, colorectal cancer, or pancreatic cancer require novel medical interventions; this is particularly true for those whose cancers are marked by a KRAS mutation, as well as those who have advanced following chemotherapy.
| Targets |
KRAS(G12C); Mutant KRAS G12C (irreversibly binds to the switch II pocket, Ki = 11 nM for KRAS G12C-GDP; IC50 = 0.21 μM for inhibiting KRAS G12C-mediated signaling in H358 cells) [6]
Sotorasib (AMG-510) targets KRAS G12C mutant protein (Ki = 12 nM for KRAS G12C [2] ; IC50 = 0.015 μM for KRAS G12C GTP binding inhibition [6] ; no significant binding to wild-type KRAS (Ki > 1000 nM) [2][6] ) |
|---|---|
| ln Vitro |
- Sotorasib (AMG-510) potently inhibited proliferation of KRAS G12C-positive cancer cell lines, with IC50 values of 0.01-0.5 μM (e.g., H358 lung adenocarcinoma: 0.03 μM; MIA PaCa-2 pancreatic cancer: 0.12 μM) as measured by CellTiter-Glo assay. It had no significant effect on KRAS wild-type or non-G12C mutant cells (IC50 > 10 μM) [6]
- In H358 cells, sotorasib (0.1-1 μM) dose-dependently reduced phosphorylation of KRAS downstream effectors (p-ERK, p-AKT, p-S6) within 2 hours (Western blot), with maximal inhibition at 1 μM [6] - The compound (1 μM) induced apoptosis in KRAS G12C-positive cells (Annexin V/PI staining) and reduced colony formation (by 80% in H358 cells) compared to vehicle [6] AMG 510 had little effect on KRAS (C118A) but inhibits the nucleotide exchange of recombinant mutant KRAS (G12C/C118A) when catalyzed by SOS1. Additionally, AMG 510 specifically reduces the viability of KRAS p.G12C mutant lines while having no effect on cell lines harboring other KRAS mutations[1]. 1. Sotorasib (AMG-510) exhibited potent antiproliferative activity against KRAS G12C-mutant cancer cell lines, with IC50 values of 0.2 μM (H358, NSCLC), 0.5 μM (NCI-H2122, NSCLC), 0.8 μM (MIA PaCa-2, pancreatic cancer), and 1.2 μM (DLD-1, colorectal cancer) after 72-hour treatment [1] 2. In KRAS G12C-mutant H358 cells, Sotorasib (AMG-510) (0.1-1 μM) dose-dependently inhibited KRAS GTP loading (by 40-90% at 24 hours) and downregulated the MAPK signaling pathway, as shown by reduced phosphorylation of ERK1/2 (p-ERK) and MEK1/2 (p-MEK) via western blot [2] 3. Flow cytometry with Annexin V/PI staining revealed that Sotorasib (AMG-510) (0.5-2 μM) induced apoptosis in H358 cells (35% apoptotic cells at 1 μM, 68% at 2 μM) and MIA PaCa-2 cells (28% apoptotic cells at 1 μM, 59% at 2 μM) after 48-hour treatment [6] 4. Clonogenic assay demonstrated that Sotorasib (AMG-510) (0.01-1 μM) reduced colony formation of KRAS G12C-mutant NSCLC cell lines by 50-90% at 0.5 μM, while having minimal effect on KRAS wild-type A549 cells (colony inhibition <10% at 1 μM) [1] 5. In primary KRAS G12C-mutant NSCLC patient-derived cells, Sotorasib (AMG-510) (0.3-3 μM) inhibited cell proliferation with a mean IC50 of 0.6 μM and suppressed downstream KRAS signaling (p-ERK, p-AKT) [4] 6. Combination treatment of Sotorasib (AMG-510) with trametinib (MEK inhibitor) or cetuximab (EGFR antibody) showed synergistic antiproliferative effects in KRAS G12C-mutant colorectal cancer cells (combination index <0.7) [3] |
| ln Vivo |
- In mice bearing H358 xenografts (KRAS G12C), sotorasib (10-180 mg/kg, oral gavage, daily for 21 days) caused dose-dependent tumor regression: 180 mg/kg led to 90% tumor growth inhibition (TGI) and 30% complete regression. No significant effect was observed in KRAS wild-type A549 xenografts [6]
- In a patient-derived xenograft (PDX) model of KRAS G12C colorectal cancer, sotorasib (100 mg/kg, oral) reduced tumor volume by 65% after 28 days, with decreased p-ERK levels in tumor tissues (immunohistochemistry) [3] AMG 510 quickly and irreversibly binds to KRAS (G12C) in preclinical tumor models, resulting in long-lasting inhibition of the mitogen-activated protein kinase (MAPK) signaling pathway. When administered orally (once daily) as a single agent, AMG 510 has the ability to induce tumor regression in KRASG12C cancer-bearing mice models[2]. 1. In H358 KRAS G12C-mutant NSCLC subcutaneous xenograft mice, oral administration of Sotorasib (AMG-510) (10, 30, 100 mg/kg once daily) dose-dependently inhibited tumor growth, with tumor growth inhibition (TGI) rates of 45%, 72%, and 90% after 21 days of treatment; the 100 mg/kg dose also extended median overall survival (OS) from 32 days (vehicle) to 58 days [5] 2. In MIA PaCa-2 pancreatic cancer xenograft mice, Sotorasib (AMG-510) (100 mg/kg PO qd ×28 days) achieved a TGI of 80% and reduced tumor weight by 75% compared to vehicle; immunohistochemistry (IHC) of tumor tissues showed decreased p-ERK expression (by 85%) [6] 3. In KRAS G12C-mutant NSCLC patient-derived xenograft (PDX) models (n=5), Sotorasib (AMG-510) (50-100 mg/kg PO qd ×21 days) induced partial responses (PR) in 3/5 models and stable disease (SD) in 2/5 models, with a mean TGI of 65% [4] 4. In a Kras G12C-driven genetically engineered mouse model (GEMM) of lung adenocarcinoma, Sotorasib (AMG-510) (75 mg/kg PO qd ×14 days) reduced lung tumor burden by 78% and inhibited metastatic lesions in the liver and lymph nodes [6] 5. Combination of Sotorasib (AMG-510) (50 mg/kg PO) with anti-PD-1 antibody (10 mg/kg IP twice weekly) in KRAS G12C-mutant NSCLC PDX models enhanced antitumor activity, with complete responses (CR) in 2/4 models versus 0/4 with single-agent Sotorasib (AMG-510) [3] |
| Enzyme Assay |
Activating mutations in RAS represent the most common oncogenic driver mutation in cancer. The single amino acid substitution of cysteine for glycine at position 12 (KRASG12C) is frequently found in solid malignancies, particularly in lung adenocarcinoma (~13%), colorectal adenocarcinoma (3%), and pancreatic adenocarcinoma (~1%). Recently it has been demonstrated that KRASG12C can be targeted with covalent small molecule inhibitors which react with the mutant cysteine adjacent to the switch II pocket (SIIP), locking KRAS in its inactive GDP-bound state. We describe here the discovery and in vitro characterization of AMG 510, a covalent inhibitor of KRASG12C possessing potent biochemical and cellular activity, as well as robust in vivo efficacy. AMG 510 inhibited SOS1-catalyzed nucleotide exchange of recombinant mutant KRASG12C/C118A but had minimal effect on KRASC118A, which is wildtype at position 12. The observed rate constant (kinact/Ki) of covalent modification of KRASG12C by AMG 510 was determined biochemically by mass spectrometry as well as in the cellular context (kobs/[I]). Cysteine proteome analysis of cells treated with AMG 510 revealed that only the G12C-containing peptide of KRAS was covalently modified. AMG 510 inhibited KRAS signaling as measured by ERK phosphorylation in all KRAS p.G12C cell lines tested, but did not inhibit phosphorylation of ERK in cell lines lacking the KRAS p.G12C mutation. Cellular occupancy of KRASG12C by AMG 510 was determined by mass spectrometry and correlated well with inhibition of ERK phosphorylation. AMG 510 also selectively impaired the viability of KRAS p.G12C mutant lines. Combination treatment of AMG 510 with inhibitors of other cellular signaling pathways exhibited evidence for synergistic effects on cell viability. Treatment of KRAS p.G12C lines with covalent KRASG12C inhibitors increased the expression of HLA. To test the impact of KRASG12C inhibition on immune surveillance in vivo, we generated a syngeneic tumor cell line that is suitable for testing AMG 510 in combination with checkpoint inhibitor therapies and characterized this line in vitro. AMG 510 is currently being evaluated in a Phase I study in patients with solid tumors harboring KRAS p.G12Cmutations[1].
KRAS G12C binding assay: Purified KRAS G12C-GDP protein was incubated with sotorasib (0.1-100 nM) and analyzed by surface plasmon resonance (SPR). The compound showed slow dissociation (t1/2 = 10 hours) with a Ki of 11 nM. GTPase activity was measured using a luminescent GTP hydrolysis assay, where sotorasib (1 μM) inhibited KRAS G12C GTPase activity by 85% [6] 1. Surface Plasmon Resonance (SPR) binding assay for KRAS G12C: Recombinant KRAS G12C protein was immobilized on a sensor chip, and serial dilutions of Sotorasib (AMG-510) (0.001-1 μM) were injected over the chip at a flow rate of 30 μL/min; binding responses (RU) were recorded in real-time, and kinetic parameters (ka, kd, KD) were calculated using a 1:1 binding model to determine the affinity of Sotorasib (AMG-510) for KRAS G12C [2] 2. Homogeneous Time-Resolved Fluorescence (HTRF) GTP binding assay: KRAS G12C protein was incubated with a fluorescently labeled GTP analog and increasing concentrations of Sotorasib (AMG-510) (0.005-5 μM) for 60 minutes at room temperature; HTRF signals were measured at 665 nm and 620 nm, and the percentage inhibition of GTP binding was calculated to determine the IC50 value for KRAS G12C inhibition [6] 3. Isothermal Titration Calorimetry (ITC) assay: Sotorasib (AMG-510) was titrated into a solution of KRAS G12C protein (10 μM) in a calorimeter cell at 25°C; heat changes associated with the binding interaction were recorded, and thermodynamic parameters (ΔH, ΔS, KD) were derived to characterize the binding mode of Sotorasib (AMG-510) to KRAS G12C [2] |
| Cell Assay |
Cell Line: NCI-H358 and MIA PaCa-2 cells
Concentration: 1-10 μM Incubation Time: 72 hours Result: Potently impaired cellular viability in both NCI-H358 and MIA PaCa-2 (IC50≈0.006 μM and 0.009 μM respectively). Somatic activating mutations of RAS family members are tumor driver mutations found in an estimated 21% of all cancers. Oncogenic KRAS mutations at residues G12, G13, and Q61 represent the most common RAS mutations found in solid malignancies. The prevalence of KRAS p.G12C tumors is ~13% of lung adenocarcinoma (including NSCLC), 3% of colorectal carcinoma (CRC), and 1% to 2% of numerous other solid tumors, representing an unmet medical need. We have developed AMG 510, an orally bioavailable, covalent inhibitor of KRASG12C with potent biochemical and cellular activity, and robust in vivo efficacy. AMG 510 inhibited SOS-catalyzed nucleotide exchange of recombinant mutant KRASG12C/C118A but had minimal effect on KRASC118A, which is wildtype at position 12. In cellular assays, AMG 510 covalently modified KRASG12C and inhibited KRASG12C signaling as measured by phosphorylation of ERK1/2 (p-ERK) in all KRAS p.G12C-mutant cell lines tested but did not inhibit p-ERK in cell lines with various other KRAS mutations. AMG 510 also selectively impaired viability of KRAS p.G12C mutant cell lines but did not affect cell lines with other KRAS mutations[5]. - Proliferation assay: KRAS G12C-positive cells (H358, MIA PaCa-2) were seeded in 96-well plates and treated with sotorasib (0.001-10 μM) for 72 hours. Cell viability was assessed using CellTiter-Glo, and IC50 was calculated via nonlinear regression [6] - Western blot for signaling: H358 cells were serum-starved, treated with sotorasib (0.1-1 μM) for 2 hours, then lysed. Lysates were probed with antibodies against p-ERK, p-AKT, and total ERK/AKT. Band intensities were normalized to β-actin [6] 1. Cell viability assay: KRAS G12C-mutant (H358, MIA PaCa-2) and wild-type (A549) cancer cell lines were seeded in 96-well plates at 5×10³ cells/well; serial dilutions of Sotorasib (AMG-510) (0.001-10 μM) were added, and cells were incubated for 72 hours at 37°C with 5% CO₂; a cell viability reagent was added, and absorbance was measured to calculate IC50 values for antiproliferative activity [1] 2. Apoptosis detection assay: KRAS G12C-mutant cancer cells were treated with Sotorasib (AMG-510) (0.1-2 μM) for 48 hours, harvested, and stained with Annexin V-FITC and propidium iodide (PI); flow cytometry was used to quantify early (Annexin V+/PI-) and late (Annexin V+/PI+) apoptotic cells; primary patient-derived cells were processed using the same protocol to assess apoptotic responses [6] 3. Western blot analysis for KRAS signaling: Cells were lysed in a buffer containing protease and phosphatase inhibitors 24-48 hours after Sotorasib (AMG-510) treatment; protein concentrations were quantified, and equal amounts of protein were separated by SDS-PAGE and transferred to a membrane; the membrane was probed with antibodies against KRAS, p-ERK, total ERK, p-MEK, total MEK, and β-actin (loading control); band intensities were quantified by densitometry to evaluate pathway inhibition [2] 4. Colony formation assay: KRAS G12C-mutant NSCLC cells were seeded in 6-well plates at 500 cells/well and treated with Sotorasib (AMG-510) (0.01-1 μM) for 14 days at 37°C with 5% CO₂; colonies were fixed with methanol, stained with crystal violet, and counted; the percentage of colony formation inhibition was calculated relative to vehicle-treated controls [1] |
| Animal Protocol |
Female ICR-SCID mice
\n100 mg/kg \no.g. \nThe RAS gene family encodes the small GTPase proteins NRAS, HRAS, and KRAS, which play an essential role in cellular growth and proliferation. KRAS is one of the most frequently mutated oncogenes in human cancer, with KRAS p.G12D, p.G12V, and p.G12C constituting the major mutational subtypes across lung, colon, and pancreatic cancers. Despite more than three decades of research, indirect approaches targeting KRAS mutant cancers have largely failed to show clinical benefit, and direct approaches have been stymied by the apparently ‘undruggable’ nature of KRAS. Cysteine-12 of KRASG12C has recently emerged as a unique vulnerability in KRAS-mutant cancers, and a small number of cysteine-reactive inhibitory tool molecules have been disclosed. We here report independent efforts to identify cysteine-reactive molecules capable of selectively inhibiting KRASG12C. Through iterative screening and structural biology efforts, we identified a novel Cys12-reactive inhibitor scaffold that derived its potency from occupancy of a previously unknown cryptic pocket induced by side-chain motion of the His95 residue of KRAS. Employing a scaffold-hopping approach, we leveraged knowledge of this cryptic pocket to design a series of N-aryl quinazolin-2(1H)-one-based inhibitors that demonstrated significantly enhanced potency relative to prior tool compounds. Extensive optimization of these leads led to the identification of a highly potent, selective, and well-tolerated inhibitor of KRASG12C, which was nominated for clinical development as AMG 510. In preclinical tumor models, AMG 510 rapidly and irreversibly binds to KRASG12C, providing durable suppression of the mitogen-activated protein kinase (MAPK) signaling pathway. Dosed orally (once daily) as a single agent, AMG 510 is capable of inducing tumor regression in mouse models of KRASG12C cancer. AMG 510 is, to the best of our knowledge, the first direct KRASG12C therapeutic to reach human clinical testing and is currently in a Phase I clinical trial evaluating safety, tolerability, PK, and efficacy in subjects with solid tumors bearing the KRAS p.G12C mutation (NCT03600883). \n- Xenograft model: Nude mice were subcutaneously injected with H358 cells (5×10⁶). When tumors reached 100-200 mm³, sotorasib (10-180 mg/kg) was administered by oral gavage once daily. Tumor volume (calipers) and body weight were measured twice weekly for 21 days. Tumor tissues were harvested for immunohistochemical analysis of p-ERK [6] \n \n - PDX model: Mice bearing KRAS G12C colorectal cancer PDXs received sotorasib (100 mg/kg, oral) daily for 28 days. Tumor growth was monitored, and Ki-67 (proliferation marker) expression was quantified [3] \n1. H358 NSCLC subcutaneous xenograft model: Female NOD/SCID mice (6-8 weeks old) were injected subcutaneously with 1×10⁷ H358 cells into the right flank; tumors were allowed to reach 100-150 mm³ before treatment initiation; Sotorasib (AMG-510) was formulated in a vehicle of 0.5% methylcellulose + 0.1% Tween 80 and administered orally via gavage at 10, 30, or 100 mg/kg once daily for 21 days; tumor volume was measured every 3 days using calipers (volume = length × width² / 2), and body weight was monitored to assess toxicity [5] \n2. MIA PaCa-2 pancreatic cancer xenograft model: NOD/SCID mice were injected subcutaneously with 2×10⁶ MIA PaCa-2 cells; once tumors reached 150-200 mm³, mice were randomized to receive Sotorasib (AMG-510) (100 mg/kg PO qd ×28 days) or vehicle; tumor volume and body weight were measured twice weekly, and tumor tissues were collected at study end for IHC analysis of p-ERK [6] \n3. KRAS G12C NSCLC PDX model: Primary KRAS G12C-mutant NSCLC tissues from patients were implanted subcutaneously into NSG mice; once tumors reached 200 mm³, mice were treated with Sotorasib (AMG-510) (50 or 100 mg/kg PO qd ×21 days); tumor growth was assessed by caliper measurement, and response criteria (CR, PR, SD, PD) were defined per RECIST 1.1 [4] \n4. Kras G12C GEMM of lung adenocarcinoma: Transgenic mice with conditional Kras G12C expression in lung epithelial cells were treated with Sotorasib (AMG-510) (75 mg/kg PO qd ×14 days) or vehicle; lung tumor burden was quantified by histology, and metastatic lesions in the liver and lymph nodes were counted by immunohistochemistry [6] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
A once-daily dose of 960 mg sotorasibel results in a peak plasma concentration (Cmax) of 7.50 μg/mL, a median time to peak concentration (Tmax) of 2.0 h, and an area under the curve (AUC0-24h) of 65.3 h μg/mL. 74% of sotorasibel is excreted in feces and 6% in urine. 53% of the dose is excreted unchanged in feces, and 1% is excreted unchanged in urine. The volume of distribution of sotorasibel is 211 L. The steady-state apparent clearance of sotorasibel is 26.2 L/h. Metabolism/Metabolites Sotorasibel is primarily metabolized via binding or CYP3A. Biological half-life The terminal elimination half-life of sotorasibel is 5.5 ± 1.8 hours. - In mice, the bioavailability of sotorasibel (10 mg/kg) after oral administration is 70%, with a peak plasma concentration (Cmax) of 2.3 μg/mL at 1 hour. Its plasma half-life (t1/2) is 4.5 hours, and it has good tumor penetration (tumor/plasma ratio = 3.2) [6] - In patients, sotorasibel (960 mg, orally) reaches a Cmax of 7.1 μg/mL at 1.5 hours, with a terminal t1/2 of 5 hours. Plasma protein binding was 95% [4] 1. In male CD1 mice, after oral administration of Sotorasib (AMG-510) (100 mg/kg), the peak plasma concentration (Cmax) was 8.2 μM (2 h Tmax), the oral bioavailability (F) was 30%, the terminal half-life (t1/2) was 4.1 h, the volume of distribution (Vd) was 2.3 L/kg, and the total clearance (CL) was 0.5 L/h/kg [5] 2. In Sprague-Dawley rats, the Cmax of Sotorasib (AMG-510) (50 mg/kg PO) was 5.6 μM (Tmax = 3 h), F = 20%, t1/2 = 6.2 h, and Vd = 3.1 L/kg; the drug showed good tissue permeability, with a lung/plasma ratio of 3.2 4 hours after administration [6] 3. In the human liver microsomal assay, Sotorasib (AMG-510) was mainly metabolized by CYP3A4 (70%) and CYP2C9 (20%) oxidation; less than 10% of the drug was excreted unchanged in the urine and feces of mice within 48 hours [3] 4. In healthy volunteers, after a single oral administration of Sotorasib (AMG-510) (960 mg), the peak plasma concentration (Cmax) was 2.1 μM (Tmax = 4 hours), the half-life (t1/2) was 5.5 hours, and the 24-hour AUC₀ = 18.7 μM·h; steady-state plasma concentrations were reached after 7 days of once-daily administration [4] |
| Toxicity/Toxicokinetics |
Hepatotoxicity
Liver dysfunction was common in pre-market clinical trials of sotorasib in patients with KRAS G12C-mutant solid tumors, but it was usually self-limiting and mild. 38% of patients treated with sotorasib experienced varying degrees of ALT elevation, with 6% to 7% experiencing ALT elevations exceeding 5 times the upper limit of normal (ULN). In these trials, which included approximately 427 patients, 8% discontinued sotorasib prematurely due to elevated AST or ALT. Furthermore, a small number of patients experienced severe hepatotoxicity, requiring discontinuation of sotorasib and treatment with glucocorticoids. The median time to onset of liver dysfunction was 9 weeks after treatment initiation. Although serum transaminase levels occasionally showed significant elevations (5 to 20 times the ULN), these were not accompanied by elevated serum bilirubin, and no patients developed clinically significant liver damage with jaundice. The sotorasirb product information recommends routine liver function tests before treatment, every 3 weeks for the first 3 months, and monthly as needed thereafter. It is noteworthy that patients with significantly elevated serum transaminase levels during sotorasirb treatment have mostly received immune checkpoint inhibitor therapy (usually anti-PD-L1 therapy) within 1 to 3 months prior to starting sotorasirb treatment. Furthermore, these elevations often respond rapidly to glucocorticoid therapy and sometimes do not recur when sotorasirb treatment is restarted several months later. These findings suggest that elevated aminotransferase levels during sotorasirb treatment are due to delayed immune-mediated hepatotoxicity induced by prior immune checkpoint inhibitor therapy. Probability score: D (Possibly but rarely causes clinically significant liver damage). Pregnancy and Lactation Effects ◉ Overview of Use During Lactation There is currently no information on the clinical use of sotorasirb during lactation. Because sotorasirb binds to plasma proteins at a rate of 89%, its concentration in breast milk is likely to be low. However, due to its potential toxicity to breastfed infants, the manufacturer recommends discontinuing breastfeeding during treatment with sotorasidib and for one week after the last dose. ◉ Effects on breastfed infants No published information was found as of the revision date. ◉ Effects on lactation and breast milk No published information was found as of the revision date. Protein binding: Sotorasidib has a protein binding rate of 89% in plasma. - In preclinical studies, sotorasidib (orally, at doses up to 300 mg/kg) did not show significant toxicity in mice, with normal liver and kidney function [6]. - In clinical trials, common adverse events (≥15%) included diarrhea (34%), nausea (25%), and fatigue (21%). Grade 3/4 toxicity reactions are rare (<5%), and no dose-limiting nephrotoxicity or hepatotoxicity has been observed [4] 1. Sotorasib (AMG-510) showed high plasma protein binding rates in mouse, rat and human plasma (96.5%, 97.2% and 98.0%, respectively) [5] 2. Acute toxicity studies in CD-1 mice showed no death or significant toxicity at oral doses up to 1000 mg/kg; subchronic toxicity studies (oral administration of 100 and 300 mg/kg/day to rats for 28 days) showed mild weight loss (<8%), and no significant changes in liver/kidney function indicators (ALT, AST, BUN, creatinine) [6] 3. In vitro CYP450 inhibition assays showed that sotorasib (AMG-510) had a weak inhibitory effect on CYP3A4 (IC50 = 9.5 μM), and at concentrations up to 10 At μM, it does not inhibit CYP1A2, CYP2C9, CYP2C19 or CYP2D6, indicating a low risk of drug interaction [3] 4. In clinical trials, the most common adverse events (AEs) of sotorasirb (AMG-510) were diarrhea (32%), nausea (21%) and fatigue (18%); grade 3/4 adverse events were rare (<5%), including elevated liver enzymes and pneumonia [4] |
| References | |
| Additional Infomation |
Sotorasib is a pyridopyrimidine compound with the structure pyrido[2,3-d]pyrimidine-2(1H)-one, substituted at positions 1, 4, 6, and 7 with 4-methyl-2-(propyl-2-yl)pyridin-3-yl, (2S)-2-methyl-4-(propyl-2-enoyl)piperazin-1-yl, fluorine, and 2-fluoro-6-hydroxyphenyl, respectively. It is approved for the treatment of non-small cell lung cancer patients with KRAS (G12C) mutations and is an anti-tumor drug. It belongs to the acrylamide, N-acylpiperazine, pyridopyrimidine, monofluorobenzene, methylpyridine, tertiary carboxamide, tertiary amino compound, and phenolic compound classes. Sotorasib (AMG-510) is an acrylamide derivative KRAS inhibitor developed by Amgen. It is indicated for the treatment of adult non-small cell lung cancer patients with KRAS G12C mutations. This mutation accounts for more than 50% of all KRAS mutations. KRAS mutants were discovered in 1982, but were not considered a druggable target until the mid-2010s. It was the first experimental KRAS inhibitor. Currently, another drug targeting the same target [MRTX849] is also under development. Sotorasiib received approval from the U.S. Food and Drug Administration (FDA) on May 28, 2021, and subsequently from the European Commission on January 10, 2022. Sotorasiib is a small-molecule KRAS G12C mutant protein inhibitor, which is present in up to 13% of refractory non-small cell lung cancer cases. Elevated serum transaminases are common during sotorasiib treatment, and some patients experience clinically significant, potentially severe, liver damage. Sotorasiib is an oral KRAS-specific mutant p.G12C inhibitor with potential antitumor activity. After oral administration, sotorasib selectively targets, binds to, and inhibits the activity of KRAS p.G12C mutants. This may inhibit the growth of tumor cells expressing KRAS p.G12C. KRAS p.G12C mutations are found in certain tumor cell types and play a key role in tumor cell proliferation. Drug Indications Sotorasib is indicated for the treatment of adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) with KRAS G12C mutations who have received at least one prior systemic therapy. Lumykras monotherapy is indicated for the treatment of adult patients with advanced non-small cell lung cancer (NSCLC) with KRAS G12C mutations whose disease has progressed after at least one prior systemic therapy. Mechanism of Action Normally, GTP binds to KRAS, activating the protein and promoting the entry of effector molecules into the MAP kinase pathway. GTP is hydrolyzed to GDP, inactivating KRAS. The KRAS G12C mutation impairs the hydrolysis of GTP, maintaining it in its active form. Sotorasirb binds to cysteine residues in the KRAS G12C mutant, keeping the protein inactive. The cysteine residues targeted by sotorasirb are absent in wild-type KRAS, thus avoiding off-target effects. This mutation is present in 13% of non-small cell lung cancers, 3% of colorectal and appendiceal cancers, and 1–3% of solid tumors.
Pharmacodynamics Sortorasirb is indicated for the treatment of adult patients with KRAS G12C-mutant non-small cell lung cancer. Due to the need for daily dosing, its duration of action is moderate. Patients should be informed of the risks of hepatotoxicity, interstitial lung disease, and pneumonia; breastfeeding should be avoided during treatment and for one week after the last dose. - Sotorasib (AMG-510) is a first-in-class covalent inhibitor that irreversibly binds to the GDP-binding form of KRAS G12C, rendering it inactive[6] - It is indicated for the treatment of KRAS G12C-mutant non-small cell lung cancer (NSCLC) that has previously received systemic therapy and was approved by the FDA in 2021[3][4] 1. Sotorasib (AMG-510) is a first-in-class selective small molecule KRAS G12C inhibitor, which is a mutant form of KRAS commonly found in non-small cell lung cancer (13%), pancreatic cancer (1-2%), and colorectal cancer (3-4%)[2] 2. The mechanism of action of sotorasib (AMG-510) involves covalent binding to the 12th cysteine residue of KRAS G12C, locking the protein in its inactive GDP-binding state. Binding state and inhibiting downstream RAS-MAPK signaling pathway[6] 3. Sotorasib (AMG-510) was approved by the FDA in 2021 for the treatment of patients with advanced KRAS G12C mutant non-small cell lung cancer (NSCLC) who have received at least one prior systemic therapy[3] 4. Resistance to sotorasib (AMG-510) may be caused by secondary KRAS mutations (e.g., G12D, G12V), bypass signaling pathways (EGFR, MET amplification) or histological transformation; resistance may be overcome by combination with MEK, EGFR or immune checkpoint inhibitors[4] 5. Preclinical data show that sotorasib (AMG-510) is effective against KRAS G12C mutant solid tumors other than non-small cell lung cancer (including pancreatic cancer and colorectal cancer), and phase II/III clinical trials are currently being evaluated for these indications[6] |
| Molecular Formula |
C30H30F2N6O3
|
|---|---|
| Molecular Weight |
560.5944
|
| Exact Mass |
560.23
|
| Elemental Analysis |
C, 64.28; H, 5.39; F, 6.78; N, 14.99; O, 8.56
|
| CAS # |
2296729-00-3
|
| Related CAS # |
Sotorasib racemate; 2252403-56-6; Sotorasib isomer; Sotorasib-d7; 2296729-66-1; 2387559-45-5
|
| PubChem CID |
137278711
|
| Appearance |
White to yellow solid powder
|
| LogP |
4
|
| Hydrogen Bond Donor Count |
1
|
| Hydrogen Bond Acceptor Count |
7
|
| Rotatable Bond Count |
5
|
| Heavy Atom Count |
41
|
| Complexity |
1030
|
| Defined Atom Stereocenter Count |
1
|
| SMILES |
C[C@H]1CN(CCN1C2=NC(=O)N(C3=NC(=C(C=C32)F)C4=C(C=CC=C4F)O)C5=C(C=CN=C5C(C)C)C)C(=O)C=C
|
| InChi Key |
NXQKSXLFSAEQCZ-SFHVURJKSA-N
|
| InChi Code |
InChI=1S/C30H30F2N6O3/c1-6-23(40)36-12-13-37(18(5)15-36)28-19-14-21(32)26(24-20(31)8-7-9-22(24)39)34-29(19)38(30(41)35-28)27-17(4)10-11-33-25(27)16(2)3/h6-11,14,16,18,39H,1,12-13,15H2,2-5H3/t18-/m0/s1
|
| Chemical Name |
6-fluoro-7-(2-fluoro-6-hydroxyphenyl)-1-(4-methyl-2-propan-2-ylpyridin-3-yl)-4-[(2S)-2-methyl-4-prop-2-enoylpiperazin-1-yl]pyrido[2,3-d]pyrimidin-2-one
|
| Synonyms |
AMG-510; sotorasib; AMG 510; AMG510; trade names: Lumakras; Lumykras;
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
DMSO: 50~100 mg/mL (89.2~178.4 mM)
Ethanol: ~13 mg/mL |
|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.71 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (3.71 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.08 mg/mL (3.71 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 5%DMSO+ 40%PEG300+ 5%Tween 80+ 50%ddH2O: 5.0mg/ml (8.92mM) Solubility in Formulation 5: 10 mg/mL (17.84 mM) in 20% HP-β-CD in Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7838 mL | 8.9192 mL | 17.8383 mL | |
| 5 mM | 0.3568 mL | 1.7838 mL | 3.5677 mL | |
| 10 mM | 0.1784 mL | 0.8919 mL | 1.7838 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Targeted Therapy Directed by Genetic Testing in Treating Patients With Locally Advanced or Advanced Solid Tumors, The ComboMATCH Screening Trial
CTID: NCT05564377
Phase: Phase 2 Status: Recruiting
Date: 2024-11-21
|
|---|
AMG 510 inhibits ERK phosphorylation and growth of KRASG12C-mutant tumours in vivo.Nature. 2019 Nov;575(7781):217-223. |
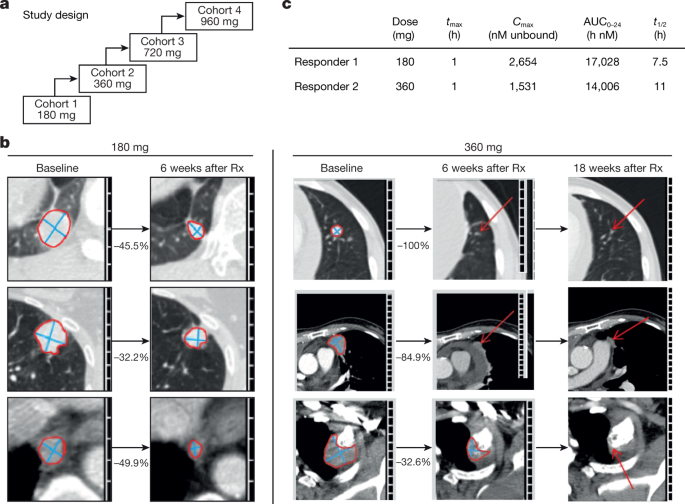 Clinical activity of AMG 510 in patients with lung cancer in first-in-human dose-escalation study.Nature. 2019 Nov;575(7781):217-223. Clinical activity of AMG 510 in patients with lung cancer in first-in-human dose-escalation study.Nature. 2019 Nov;575(7781):217-223. |