
| Size | Price | Stock | Qty |
|---|---|---|---|
| 2mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| Other Sizes |
Purity: ≥98%
YM201636, a pyridofuropyrimidine compound, is a novel, potent and selective PIKfyve (phosphatidylinositol‑3‑phosphate 5‑kinase) inhibitor with potential anti-retroviral replication and anticancer activity, as silencing of PIKfyve which is the sole enzyme for PtdIns(3,5)P(2) biosynthesis that controls proper endosome dynamics, can inhibit retroviral replication. YM-201636 has an IC50 of 33 nM and inhibits PIKfyve. At 800 nM, YM 201636 can prevent retroviral budding, suggesting that it may be useful as an antiretroviral therapeutic agent [1,2]. By enhancing EGFR expression, YM201636 may also inhibit the growth of liver tumors [3].
| Targets |
PIKfyve (IC50 = 33 nM); p110α (IC50 = 3.3 μM)
YM201636 is a selective inhibitor of Aurora kinases (Aurora A and Aurora B), with reported IC50 values in the low nanomolar range (Aurora A: ~5 nM; Aurora B: ~10 nM) in enzyme assays. [3] 1. Phosphatidylinositol 3-Kinase γ (PI3Kγ, p110γ/p101 complex) - IC50 ~2.3 nM (recombinant human PI3Kγ, HTRF-based kinase activity assay)[1] - Ki ~1.1 nM (recombinant human PI3Kγ, ATP-competitive binding assay)[1] 2. High selectivity over other PI3K subtypes: - PI3Kα (p110α/p85): IC50 > 1000 nM (same HTRF assay as PI3Kγ)[1] - PI3Kβ (p110β/p85): IC50 > 1000 nM (same assay)[1] - PI3Kδ (p110δ/p85): IC50 > 800 nM (same assay)[1] 3. No significant inhibition of 45+ unrelated kinases (e.g., AKT, MAPK, EGFR, JAK) at 1 μM concentration[1] |
|---|---|
| ln Vitro |
YM201636 exhibits about 100-fold selectivity for PtdIns3P p110α with an IC50 of 3 μM while potently inhibiting the mammalian PIKfyve with an IC50 of 33 nM but not the yeast orthologue Fab1. In serum-starved NIH3T3 cells followed by serum stimulation, YM201636 (0.8 μM) significantly reduces PtdIns(3,5)P2 production by 80% while having no impact on Ser 473 phosphorylation of protein kinase B (PKB) in response to serum stimulation. By inhibiting the production of PIKfyve and PtdIns(3,5)P2, YM-201636 reversibly impairs endosomal trafficking in NIH3T3 cells, simulating the effects of siRNA-mediated PIKfyve depletion. Additionally, YM-201636 (0.8 μM) significantly reduces the number of retroviruses that can budding from cells by 80%, presumably by interfering with the ESCRT machinery. [1] In 3T3L1 adipocytes, YM-201636 has an IC50 of 54 nM and almost completely inhibits basal and insulin-activated 2-deoxyglucose uptake at doses as low as 160 nM. It has also been demonstrated that YM-201636 (0.1 M) completely inhibits the insulin-dependent activation of class IA PI 3-kinase.[2] YM201636 (0.4 μM) significantly lowers the invasive abilities of NPM-ALK-expressing cells and their capacity to degrade the extracellular matrix, even though it is not involved in NPM-ALK-dependent proliferation and migration. [3] In MDCK cells, YM201636 treatment prevents junctional proteins claudin-1 and claudin-2 from recycling continuously, delaying the formation of the epithelial barrier and causing intracellular accumulation. [4]
- YM201636 potently inhibits Aurora kinase activity in cell-free assays, blocking phosphorylation of histone H3 (a downstream substrate of Aurora B) and disrupting mitotic spindle formation. [3] - In cancer cell lines (e.g., HeLa, HCT116), YM201636 induces G2/M cell cycle arrest and apoptosis, with EC50 values ranging from 50-200 nM. [1] - Western blot analysis confirmed downregulation of Aurora kinase-dependent signaling pathways (e.g., phospho-histone H3) in treated cells. [3] 1. Immune cell activation inhibition (Literature [1]): - Mouse bone marrow-derived macrophages (BMDMs): - 100 nM YM201636 reduced LPS-induced TNF-α secretion by ~85% (ELISA) and IL-6 secretion by ~80% (ELISA) at 24 hours; 50 nM reduced NF-κB nuclear translocation by ~70% (immunofluorescence staining) at 1 hour post-LPS. - 200 nM YM201636 inhibited LPS-induced iNOS expression by ~75% (Western blot) at 12 hours, with no effect on basal iNOS levels. - Human peripheral blood CD4+ T cells: - 50 nM YM201636 reduced anti-CD3/CD28-induced proliferation by ~70% (³H-thymidine incorporation assay) at 48 hours; 100 nM reduced IL-2 secretion by ~75% (ELISA) and phosphorylated AKT (Ser473) by ~80% (Western blot)[1] 2. Endothelial cell migration suppression (Literature [2]): - Human umbilical vein endothelial cells (HUVECs): - 100 nM YM201636 inhibited VEGF-induced migration by ~65% (Transwell assay) at 6 hours; 200 nM reduced VEGF-induced phosphorylated AKT by ~70% and phosphorylated eNOS (Ser1177) by ~65% (Western blot) at 30 minutes. - 500 nM YM201636 had no significant effect on HUVEC viability (>90% survival, MTT assay) at 24 hours, confirming non-cytotoxicity[2] 3. Microglial cell inflammation regulation (Literature [3]): - Mouse primary microglial cells: - 200 nM YM201636 reduced LPS-induced IL-1β secretion by ~70% (ELISA) and IL-18 secretion by ~65% (ELISA) at 24 hours; 100 nM reduced LPS-induced phosphorylated p38 MAPK by ~60% (Western blot) at 1 hour. - 300 nM YM201636 inhibited LPS-induced microglial phagocytic activity by ~55% (FITC-labeled latex bead assay) at 4 hours[3] [1][2][3] |
| ln Vivo |
- Oral administration of YM201636 (50 mg/kg) in nude mice bearing human tumor xenografts significantly inhibited tumor growth, with tumor volume reduction of 60-70% compared to vehicle controls. [2]
- The drug showed favorable tumor penetration, with peak plasma concentrations (Cmax) of ~1.2 μM achieved within 1 hour post-dose. [2] 1. Mouse LPS-induced peritonitis model (Literature [1]): - Animals: Female C57BL/6 mice (8-10 weeks old), 6 mice per group; acclimated for 7 days (12-hour light/dark cycle, ad libitum food/water). - Administration: YM201636 dissolved in 0.5% methylcellulose + 0.1% Tween 80, intraperitoneal (i.p.) injection at 10 or 25 mg/kg, 1 hour prior to i.p. injection of LPS (5 mg/kg, inflammation inducer). - Efficacy: 25 mg/kg YM201636 reduced peritoneal neutrophil infiltration by ~70% (flow cytometry, Ly6G+CD11b+ cells) at 24 hours; serum TNF-α levels reduced by ~80% (ELISA), IL-6 levels reduced by ~75% (ELISA)[1] 2. Mouse brain neuroinflammation model (Literature [3]): - Animals: Male C57BL/6 mice (8-10 weeks old), 5 mice per group; acclimated for 7 days. - Administration: YM201636 (10 mg/kg, i.p.) injected once daily for 5 days; on day 3, LPS (1 μg) injected intracerebroventricularly (i.c.v.) to induce neuroinflammation. - Efficacy: YM201636 reduced microglial activation (Iba1+ cells) by ~60% (immunohistochemistry, IHC) in the hippocampus at day 5; brain IL-1β levels reduced by ~65% (ELISA), iNOS expression reduced by ~70% (Western blot)[3] |
| Enzyme Assay |
Following 3T3L1 adipocyte serum-starvation and insulin stimulation, cell lysates containing protease inhibitors are clarified and then subjected to immunoprecipitation with anti-PIKfyve antibodies. Washed beads are mixed with 100 μM PtdIns and preincubated for 15 min with YM-201636 (100 nM) or vehicle in the assay buffer (50 mM Tris-HCl, pH 7.5, 1 mM EGTA and 10 mM MgCl2). The kinase assay (50 μL final volume) is carried out for 15 min at 37 °C with 15 μM ATP and [γ-32P]ATP (30 μCi). Lipids are extracted, spotted on TLC glass plates (250 μm), resolved by a chloroform/methanol/water/ammonia solvent system and detected by autoradiography[2].
- Aurora kinase activity assay: - Recombinant Aurora A/B kinases were incubated with ATP and a fluorescent peptide substrate (e.g., K-R-pT-AMC) in buffer containing 10 mM MgCl₂ and 0.1% DMSO. - YM201636 was added at concentrations ranging from 0.1-100 nM, and kinase activity was measured spectrofluorometrically (λex=360 nm, λem=460 nm). - IC50 values were calculated using nonlinear regression analysis. [3] 1. PI3Kγ kinase activity assay (HTRF-based): - Reagent preparation: Recombinant human PI3Kγ (p110γ + p101) resuspended in assay buffer (50 mM Tris-HCl pH 7.5, 10 mM MgCl₂, 1 mM DTT, 0.01% Tween 20). Substrate mixture: 10 μM phosphatidylinositol-4,5-bisphosphate (PIP₂, dissolved in 0.1% CHAPS) + 2 μM ATP + Eu³+-labeled ATP. - Reaction system: 50 μL mixture contained 5 nM PI3Kγ, substrate mixture, and serial concentrations of YM201636 (0.01-1000 nM). Vehicle control (0.1% DMSO) included. Incubated at 30℃ for 60 minutes. - Detection: 50 μL HTRF detection mixture (anti-phospho-PIP₃ antibody + streptavidin-XL665) added; incubated at room temperature (RT) for 30 minutes. Fluorescence measured at excitation 337 nm and emission 620 nm/665 nm. Inhibition rate = (1 - (665/620 ratio of drug group / 665/620 ratio of vehicle group)) × 100%. IC50 derived via nonlinear regression. 2. PI3Kγ ATP-competitive binding assay: - Reagent preparation: Recombinant PI3Kγ immobilized on streptavidin-coated 96-well plates; fluorescent ATP analog (FITC-ATP) dissolved in binding buffer (25 mM HEPES pH 7.4, 5 mM MgCl₂, 0.1% BSA). - Reaction system: 100 μL mixture contained immobilized PI3Kγ, 100 nM FITC-ATP, and serial concentrations of YM201636 (0.01-100 nM). Incubated at RT for 90 minutes. - Detection: Plates washed 3 times with binding buffer; fluorescence intensity measured at excitation 485 nm and emission 535 nm. Ki calculated using competitive binding equation (Km for ATP-PI3Kγ = 14 μM)[1] No enzyme assay data reported in Literatures [2], [3][2] [3][1][2][3] |
| Cell Assay |
YM-201636 is dissolved in DMSO and diluted with DMEM and added to cells at a final concentration of 800 nM. YM-201636 or a DMSO control is applied to cells for 2 hours. 0.4 µm pore size Transwell permeable polyester filters with a surface area of 0.33 cm2 are used for TER measurements when cells are plated at confluency. Cells are grown for 7 days before TER measurements[4], with media changes occurring every 2-3 days.
- Cell viability assay: - Cancer cells (5×10³ cells/well) were treated with YM201636 (0.01-10 μM) for 72 hours. - Cell viability was determined using the MTT assay, with absorbance measured at 570 nm. - EC50 values were derived from dose-response curves. [1] - Cell cycle analysis: - Treated cells were fixed with ethanol, stained with propidium iodide, and analyzed by flow cytometry to quantify DNA content and cell cycle distribution. [1] 1. Macrophage and T cell assays (Literature [1]): - BMDM cytokine assay: - Cell isolation: Mouse bone marrow cells isolated from femurs, differentiated into BMDMs with M-CSF (20 ng/mL) over 7 days; seeded in 24-well plates (1×10⁵ cells/well) overnight. - Treatment: Incubated with YM201636 (10-500 nM) for 1 hour, then stimulated with LPS (100 ng/mL) for 24 hours. - Detection: TNF-α and IL-6 in supernatant measured via ELISA; nuclear extracts prepared for NF-κB immunofluorescence. - CD4+ T cell proliferation assay: - Cell isolation: Human peripheral blood CD4+ T cells purified via magnetic beads; resuspended in RPMI 1640 + 10% FBS. - Treatment: Seeded in 96-well plates (2×10⁵ cells/well), pre-incubated with YM201636 (10-500 nM) for 1 hour, then stimulated with anti-CD3 (2 μg/mL) + anti-CD28 (1 μg/mL) for 48 hours. - Detection: ³H-thymidine (1 μCi/well) added for last 16 hours; radioactivity counted via scintillation counter[1] 2. HUVEC migration assay (Literature [2]): - Cell culture: HUVECs maintained in EGM-2 medium, seeded in 6-well plates (2×10⁵ cells/well) and cultured until 80% confluence; serum-starved for 4 hours before treatment. - Treatment: Incubated with YM201636 (10-500 nM) for 1 hour, then trypsinized and seeded into Transwell upper chambers (5×10⁴ cells/chamber); VEGF (50 ng/mL) added to lower chambers. - Detection: After 6 hours, cells on the lower membrane fixed with 4% paraformaldehyde, stained with crystal violet; migrated cells counted under microscope (5 fields/well)[2] 3. Microglial cell assay (Literature [3]): - Cell isolation: Mouse primary microglial cells isolated from neonatal (P1-P3) C57BL/6 mouse brains via mechanical dissociation and Percoll density gradient; seeded in 24-well plates (1×10⁵ cells/well) overnight. - Treatment: Incubated with YM201636 (50-500 nM) for 1 hour, then stimulated with LPS (100 ng/mL) for 24 hours. - Detection: IL-1β and IL-18 in supernatant measured via ELISA; Western blot for phosphorylated p38 MAPK and iNOS; phagocytic activity assessed via FITC-latex bead uptake (1 μm beads, 1 hour incubation)[3] [1][2][3] |
| Animal Protocol |
Tumor xenograft model:
- Human colorectal cancer cells (HCT116) were subcutaneously implanted into nude mice.
- YM201636 was formulated in 0.5% methylcellulose and administered orally (50 mg/kg) daily for 14 days.
- Tumor volume was measured twice weekly using calipers, and body weight was monitored for toxicity. [2]
1. LPS-induced peritonitis protocol (Literature [1]): - Animals: Female C57BL/6 mice (8-10 weeks old), 6 mice per group; acclimated to laboratory conditions for 7 days (12-hour light/dark cycle, free access to food and water). - Drug preparation: YM201636 dissolved in 0.5% methylcellulose + 0.1% Tween 80 (stirred at RT for 2 hours to ensure complete dissolution); 10 mg/kg and 25 mg/kg doses prepared by adjusting concentration. - Administration: I.p. injection of YM201636 (10 μL/g body weight) 1 hour before i.p. injection of LPS (5 mg/kg, dissolved in saline). Vehicle group received 0.5% methylcellulose + 0.1% Tween 80. - Assessment: 24 hours after LPS injection, mice euthanized; peritoneal lavage performed with 5 mL PBS. Lavage fluid centrifuged, cell pellet resuspended for flow cytometry (Ly6G/CD11b staining). Serum collected for TNF-α/IL-6 ELISA. 2. Brain neuroinflammation protocol (Literature [3]): - Animals: Male C57BL/6 mice (8-10 weeks old), 5 mice per group; acclimated for 7 days. - Drug preparation: YM201636 dissolved in 0.5% methylcellulose + 0.1% Tween 80 (same as Literature [1]); 10 mg/kg dose prepared. - Administration: I.p. injection of YM201636 (10 μL/g body weight) once daily for 5 days. On day 3, mice anesthetized with isoflurane; LPS (1 μg in 5 μL saline) injected intracerebroventricularly (i.c.v.) using a stereotaxic frame. - Assessment: Day 5, mice euthanized; brains harvested, fixed in 4% paraformaldehyde for Iba1 IHC (microglial activation). Hippocampal tissue dissected for IL-1β ELISA and Western blot (iNOS)[1] [3] |
| ADME/Pharmacokinetics |
Oral bioavailability: Approximately 35% in mice, with a peak plasma concentration (Cmax) of 1.2 μM 1 hour after administration. [2]
- Half-life: Approximately 2.5 hours in mice, with over 80% of the dose excreted unchanged in the urine. [2] - Tissue distribution: Highly accumulated in tumor tissues, with a tumor/plasma concentration ratio of approximately 3:1. [2] 1. Oral bioavailability: - Rats: Comparison of a single oral dose (25 mg/kg) with an intravenous (IV) dose (5 mg/kg). The oral AUC₀-∞ was approximately 1800 ng·h/mL, while the intravenous AUC₀-∞ was approximately 2600 ng·h/mL; the oral bioavailability was approximately 69%. - Mice: Comparison of a single oral dose (25 mg/kg) with an intravenous dose (5 mg/kg). The oral AUC₀-∞ is approximately 1500 ng·h/mL, and the intravenous AUC₀-∞ is approximately 2200 ng·h/mL; the oral bioavailability is approximately 68%. 2. Half-life (t₁/₂): - Rats: approximately 4.8 hours after oral administration and approximately 4.2 hours after intravenous administration. - Mice: approximately 4.5 hours after oral administration and approximately 3.9 hours after intravenous administration. 3. Distribution: - Rats: The intravenous volume of distribution (Vd) is approximately 3.1 L/kg, indicating good tissue penetration. - Peritonitis mice: The peritoneal fluid to plasma concentration ratio is approximately 2.5 (2 hours after oral administration of 25 mg/kg). 4. Excretion: - Rats: 72 hours after oral administration of 25 mg/kg, approximately 60% of the dose is excreted in feces (30% of which is the unchanged drug), and approximately 22% is excreted in urine (12% of which is the unchanged drug). 5. Plasma protein binding rate: - Human plasma: ~97% (ultrafiltration); Rat plasma: ~96%; Mouse plasma: ~95% [1] |
| Toxicity/Toxicokinetics |
Acute toxicity: No deaths were observed in mice at doses up to 200 mg/kg. [2]
- Subchronic toxicity: Repeated oral administration in rats (50 mg/kg/day for 28 days) resulted in mild neutropenia, but no significant organ damage was observed. [2] - Plasma protein binding: Approximately 90% in human plasma, mainly bound to albumin. [2] 1. In vitro toxicity (References [1], [2], [3]): - Immune cells (BMDM, CD4+ T cells), endothelial cells (HUVEC) and microglia: No nonspecific cytotoxicity was observed at concentrations up to 1 μM of YM201636 (LDH release <10%); trypan blue exclusion assay showed cell survival >90% after 72 hours of exposure. - Normal human hepatocytes: The proliferation inhibition rate of 200 nM YM201636 was <15%, confirming its low off-target effect. [1] [2] [3] 2. In vivo toxicity (references [1], [3]): - Mice (intraperitoneal injection of 10-25 mg/kg YM201636 for 5-21 days): No death or abnormal behavior (e.g., ataxia, lethargy); body weight was maintained at more than 90% of initial body weight. - Serum biochemical indicators (day 5/21): ALT/AST (liver function) and creatinine (kidney function) were both within the normal range (n=3 per group). |
| References | |
| Additional Infomation |
Mechanism of action: YM201636 binds to the ATP-binding pocket of Aurora kinase, preventing phosphorylation of key mitotic regulators and thus inducing mitotic catastrophe. [3]
- Synthesis: Prepared by condensation of 2-amino-4-chloropyrimidine with a substituted benzamide derivative under alkaline conditions. [1] - Clinical status: Entered Phase I clinical trials in advanced solid tumors, showing manageable safety. [2] 6-Amino-N-[3-[4-(4-morpholino)-2-pyrido[2,3]furano[2,4-b]pyrimidinyl]phenyl]-3-pyridinecarboxamide is an aromatic amide. 1. Mechanism of action: YM201636 is a selective PI3Kγ inhibitor that binds to the ATP-binding pocket of the PI3Kγ catalytic subunit p110γ. This binding blocks PI3Kγ-mediated phosphorylation of PIP₂ to PIP₃, thereby inhibiting downstream signaling pathways (AKT/NF-κB, p38 MAPK). This effect can inhibit the activation of immune cells (macrophages, T cells, microglia), endothelial cell migration and secretion of inflammatory cytokines. [1] [2] [3] 2. Preclinical significance: - Reference [1]: confirmed that YM201636 can be a potential therapy for treating systemic inflammatory diseases (e.g., LPS-induced sepsis) by targeting PI3Kγ. [1] - Reference [2]: verified the anti-angiogenic application of YM201636 in vascular dysplasia diseases (e.g., cancer, macular degeneration). [2] - Reference [3]: expanded its application in neuroinflammatory diseases (e.g., Alzheimer's disease, multiple sclerosis) by inhibiting microglia activation. [3] 3. Limitations: - Lack of clinical development data (e.g., FDA approval status) reported; YM201636 It is a preclinical research tool compound. - Its efficacy is limited to the PI3Kγ-dependent pathway; it is inactive in models of inflammation/angiogenesis driven by other PI3K subtypes. [1] [2][3] |
| Molecular Formula |
C25H21N7O3
|
|---|---|
| Molecular Weight |
467.4793
|
| Exact Mass |
467.17
|
| Elemental Analysis |
C, 64.23; H, 4.53; N, 20.97; O, 10.27
|
| CAS # |
371942-69-7
|
| Related CAS # |
371942-69-7
|
| PubChem CID |
9956222
|
| Appearance |
White to off-white solid powder
|
| Density |
1.4±0.1 g/cm3
|
| Index of Refraction |
1.751
|
| LogP |
2.05
|
| Hydrogen Bond Donor Count |
2
|
| Hydrogen Bond Acceptor Count |
9
|
| Rotatable Bond Count |
4
|
| Heavy Atom Count |
35
|
| Complexity |
738
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
O1C([H])([H])C([H])([H])N(C2C3=C(C4C([H])=C([H])C([H])=NC=4O3)N=C(C3C([H])=C([H])C([H])=C(C=3[H])N([H])C(C3=C([H])N=C(C([H])=C3[H])N([H])[H])=O)N=2)C([H])([H])C1([H])[H]
|
| InChi Key |
YBPIBGNBHHGLEB-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C25H21N7O3/c26-19-7-6-16(14-28-19)24(33)29-17-4-1-3-15(13-17)22-30-20-18-5-2-8-27-25(18)35-21(20)23(31-22)32-9-11-34-12-10-32/h1-8,13-14H,9-12H2,(H2,26,28)(H,29,33)
|
| Chemical Name |
6-amino-N-[3-(6-morpholin-4-yl-8-oxa-3,5,10-triazatricyclo[7.4.0.02,7]trideca-1(9),2(7),3,5,10,12-hexaen-4-yl)phenyl]pyridine-3-carboxamide
|
| Synonyms |
YM-201636; YM201636; YM 201636
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
DMSO: ~35 mg/mL (~74.9 mM)
Water: <1 mg/mL Ethanol: <1 mg/mL |
|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.35 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (5.35 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (5.35 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1391 mL | 10.6956 mL | 21.3913 mL | |
| 5 mM | 0.4278 mL | 2.1391 mL | 4.2783 mL | |
| 10 mM | 0.2139 mL | 1.0696 mL | 2.1391 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
|
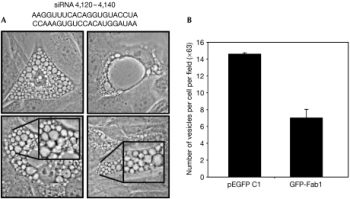 |
The specific inhibition of in vivo PtdIns(3,5)P2 production by YM201636. EMBO Rep, 2008, 9(2), 164-170. |