
| Size | Price | Stock | Qty |
|---|---|---|---|
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g | |||
| Other Sizes |
Purity: ≥98%
Vortioxetine HBr (formerly AA21004, Lu-AA21004, AA21004; Lu AA21004), the hydrobromide salt of Vortioxetine which is a marketed and atypical antidepressant, is an orally bioactive and multimodal serotonergic agent with potential anti-depressive activity. It has IC50 values of 15 nM, 33 nM, 3.7 nM, 19 nM, and 1.6 nM for 5-HT1A, 5-HT1B, 5-HT3A, 5-HT7 receptor, and SERT, respectively. In 2013, the FDA approved the atypical antidepressant vortioxetine for the treatment of major depressive disorder (MDD) in adults. One such "serotonin modulator and stimulator" is vortioxetine.
| Targets |
sPLA2 ( Ki = 15 nM ); 5-HT3A Receptor ( Ki = 3.7 nM ); 5-HT7 Receptor ( Ki = 19 nM ); SERT ( Ki = 1.6 nM )
Vortioxetine (Lu AA21004) HBr is a multimodal compound with high affinity for multiple targets: - Serotonin transporter (SERT): Ki = 1.6 nM (human recombinant, [³H]-citalopram as radioligand) [1] - 5-HT1A receptor: Ki = 3.7 nM (human recombinant, [³H]-8-OH-DPAT as radioligand); EC50 = 12 nM (functional agonism in GTPγS binding assay) [5] - 5-HT3A receptor: Ki = 3.9 nM (human recombinant, [³H]-granisetron as radioligand); IC50 = 25 nM (antagonism of 5-HT-induced calcium mobilization) [5] - 5-HT7 receptor: Ki = 19 nM (human recombinant, [³H]-SB-269970 as radioligand) [1] - No significant binding to dopamine transporter (DAT, Ki > 1000 nM) or norepinephrine transporter (NET, Ki > 1000 nM) was observed [1,5] |
|---|---|
| ln Vitro |
In vitro activity: Vortioxetine (Compound 5m) is a multimodal serotonergic agent that inhibits SERT with values of 1.6 nM, 33 nM, 3.7 nM, 19 nM, and 5-HT1A, 5-HT1B, and 5-HT7 receptors, respectively. Vortioxetine exhibits strong suppression of SERT, partial agonist characteristics at 5-HT1B and 5-HT1A receptors, antagonistic properties at 5-HT3A and 5-HT7 receptors, and agonistic properties at 5-HT1A receptors[1]. Using a whole-cell cAMP-based assay, vortioxetine is a partial h5-HT1B receptor agonist with an EC50 of 460 nM and intrinsic activity of 22%. In vitro whole-cell cAMP assay, vortioxetine binds to the r5-HT7 receptor with a Ki value of 200 nM and is a functional antagonist at the r5-HT7 receptor with an IC50 of 2 μM[5].
SERT-mediated 5-HT reuptake inhibition: HEK293 cells transfected with human SERT were treated with Vortioxetine HBr (0.1–50 nM) for 45 minutes. At 10 nM, [³H]-5-HT reuptake was inhibited by 82% vs. vehicle; IC50 = 2.1 nM (liquid scintillation counting) [1] - 5-HT1A receptor agonism: In HEK293 cells expressing human 5-HT1A receptors, Vortioxetine HBr (1–100 nM) dose-dependently increased GTPγS binding. At 30 nM, binding was enhanced by 55% vs. baseline; EC50 = 12 nM [5] - 5-HT3A receptor antagonism: HEK293 cells with human 5-HT3A receptors were pre-treated with Vortioxetine HBr (5–500 nM) for 30 minutes, then stimulated with 1 μM 5-HT. At 50 nM, 5-HT-induced calcium elevation was inhibited by 70%; IC50 = 25 nM (Fura-2 AM fluorescence) [5] - BDNF upregulation in primary neurons: Primary rat cortical neurons (7 days in vitro) treated with Vortioxetine HBr (1–10 μM) for 72 hours showed 1.8-fold higher BDNF protein levels at 5 μM (Western blot) [5] |
| ln Vivo |
Vortioxetine (Lu AA21004) is a 5-HT3 receptor antagonist that binds to the r5-HT1B receptor and rSERT (ED50 = 3.2 and 0.4 mg/kg, respectively) when administered subcutaneously[6]. After 21 days of treatment, vortioxetine dramatically boosts immature granule cell maturation in the sub granular zone of the dentate gyrus of the hippocampus as well as cell survival and proliferation[3]. Cognitive or psychomotor impairment is not brought on by vortioxetine[4].
Antidepressant-like effect in mouse forced swim test (FST): Male C57BL/6 mice (20–25 g) were orally administered Vortioxetine HBr (1, 5, 10 mg/kg/day) for 21 days. At 10 mg/kg, immobility time decreased by 45% (from 180 s to 99 s) vs. vehicle; swimming time increased by 30% [2] - Anxiolytic effect in mouse elevated plus maze (EPM): Female ICR mice (18–22 g) given oral Vortioxetine HBr (5 mg/kg) 1 hour before EPM spent 32% more time in open arms (from 19% to 25% of total time) [2] - Enhanced hippocampal neurogenesis in mice: Mice treated with oral Vortioxetine HBr (10 mg/kg/day) for 21 days had 2.3-fold more BrdU⁺ (proliferation marker) cells in the dentate gyrus vs. vehicle (immunofluorescence) [2] - Efficacy in human generalized anxiety disorder (GAD): In a randomized trial (n=362), patients with GAD received oral Vortioxetine HBr (5 mg/day) for 8 weeks. Mean Hamilton Anxiety Rating Scale (HAM-A) score decreased by 7.2 points vs. 4.1 points in placebo group (p<0.05) [4] - No impact on driving performance in humans: Healthy volunteers (n=24) taking Vortioxetine HBr (10 mg/day) for 14 days showed no significant changes in driving performance (standardized road test) or cognitive function (digit symbol substitution test) vs. placebo [3] |
| Enzyme Assay |
Vortioxetine (Compound 5m) is a multimodal serotonergic agent that inhibits SERT with values of 1.6 nM, 33 nM, 3.7 nM, 19 nM, and 5-HT1A, 5-HT1B, and 5-HT7 receptors, respectively. Vortioxetine exhibits strong suppression of SERT as well as antagonistic effects at 5-HT3A and 5-HT7 receptors, partial agonist effects at 5-HT1B receptors, and agonistic effects at 5-HT1A receptors.
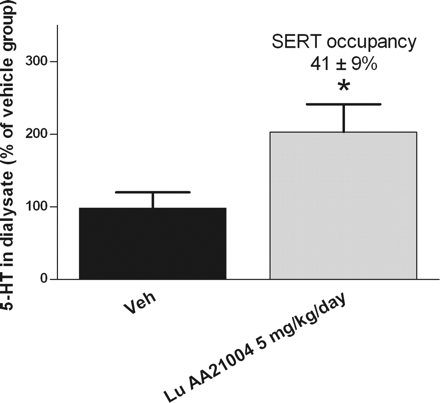
Compound 5m (Lu AA21004) was the lead compound, displaying high affinity for recombinant human 5-HT(1A) (K(i) = 15 nM), 5-HT(1B) (K(i) = 33 nM), 5-HT(3A) (K(i) = 3.7 nM), 5-HT(7) (K(i) = 19 nM), and noradrenergic β(1) (K(i) = 46 nM) receptors, and SERT (K(i) = 1.6 nM). Compound 5m displayed antagonistic properties at 5-HT(3A) and 5-HT(7) receptors, partial agonist properties at 5-HT(1B) receptors, agonistic properties at 5-HT(1A) receptors, and potent inhibition of SERT.[1] Ex vivo SERT and 5-HT3 receptor occupancy assays[2] Brains from mice treated with vehicle, fluoxetine, or vortioxetine (1 h after acute administration or 24 h after the 14th or 21st injection) were flash frozen, sectioned coronally using a cryostat, and then mounted on slides and frozen until use. Slices were 20 μm thick, and began at approximately +1.2 mm anterior from bregma for SERT receptor occupancy or −2.7 mm posterior from bregma for 5-HT3 receptor occupancy determination (Franklin and Paxinos, 2008). Slides were stored for at least 24 h at −20 °C before use in autoradiography experiments. Human SERT Binding Assay: 200 μL reaction contained 50 μg HEK293-SERT membrane protein, 0.5 nM [³H]-citalopram, and Vortioxetine HBr (0.01–100 nM). Incubated at 25°C for 60 min in 50 mM Tris-HCl (pH 7.4, 120 mM NaCl, 5 mM KCl). Filtered through glass fiber filters (pre-soaked in 0.3% polyethyleneimine), washed 3× with cold buffer. Radioactivity measured via liquid scintillation. Non-specific binding (NSB) with 10 μM imipramine; Ki calculated via Cheng-Prusoff equation [1] - Human 5-HT1A Receptor GTPγS Assay: 300 μL reaction included 100 μg HEK293-5-HT1A membrane protein, 0.1 nM [³H]-GTPγS, and Vortioxetine HBr (0.1–1000 nM). Incubated at 30°C for 90 min in 50 mM Tris-HCl (pH 7.4, 10 mM MgCl₂, 1 mM EDTA). Filtered through GF/B filters, washed 4× with cold buffer. Radioactivity quantified; EC50 derived from dose-response curves [5] - Human 5-HT3A Receptor Calcium Mobilization Assay: 150 μL reaction with 5×10⁴ HEK293-5-HT3A cells/well, 2 μM Fura-2 AM, and Vortioxetine HBr (0.5–500 nM). Loaded at 37°C for 45 min, then 1 μM 5-HT added. Fluorescence (340/380 nm excitation, 510 nm emission) measured; IC50 calculated via inhibition of 5-HT response [5] |
| Cell Assay |
Vortioxetine is a partial h5-HT1B receptor agonist that, in a whole-cell cAMP-based assay, has an EC50 of 460 nM and an intrinsic activity of 22%. In vitro whole-cell cAMP assay, vortioxetine binds to the r5-HT7 receptor with a Kivalue of 200 nM and is a functional antagonist at the r5-HT7 receptor with an IC50 of 2 μM.
Assessment of SERT occupancy[2] Slides were incubated at room temperature for 60 min in buffer (50 mM Tris–HCl, 150 mM NaCl, 5 mM KCl, pH = 7.4) containing 4.5 nM [3H]-escitalopram. Nonspecific binding was determined using 1 μM escitalopram. Slides were washed briefly in cold buffer, dried, and exposed in a Beta imager for 16 h. The region of interest (ROI) for the SERT assay included the lateral and medial septum, the nucleus accumbens and the olfactory tubercle. An example image of the ROI for the SERT assay can be found in Supplementary Fig. 2A. Assessment of 5-HT3 receptor occupancy[2] Slides were preincubated for 5 min in a buffer consisting of 50 mM Tris and 150 mM NaCl. Slides were dried under a stream of air for 30–45 min. Subsequently, slides were incubated at room temperature for 60 min in buffer (50 mM Tris–HCl, 150 mM NaCl, 5 mM KCl, pH = 7.4) containing 1 nM [3H]LY278584. Nonspecific binding was determined using 1 μM ondansetron. Slides were washed briefly in cold buffer, dried, and exposed in a Beta imager for 24 h. The ROI for the 5-HT3 receptor occupancy assay consisted of the hippocampus. An example image for the 5-HT3 receptor occupancy assay can be found in Supplementary Fig. 2B. Primary Cortical Neuron BDNF Western Blot: E18 rat cortical neurons seeded at 2×10⁵ cells/well (24-well plate) in neurobasal medium + B27. Cultured 7 days, then Vortioxetine HBr (1–10 μM) added for 72 h. Lysed with RIPA buffer (protease inhibitors), 30 μg protein run on 12% SDS-PAGE. Transferred to PVDF membrane, probed with anti-BDNF (1:1000) and anti-β-actin (1:5000) antibodies. HRP-secondary antibody + ECL detection; band intensity quantified via ImageJ [5] - HEK293-SERT [³H]-5-HT Reuptake Assay: HEK293-SERT cells seeded at 1×10⁵ cells/well (24-well plate) in DMEM + 10% FBS. Cultured 24 h, then Vortioxetine HBr (0.1–50 nM) added for 45 min. Medium replaced with 0.1 nM [³H]-5-HT in Krebs-Ringer buffer, incubated 15 min at 37°C. Washed 3× with cold buffer, lysed with 0.1 M NaOH. Radioactivity measured; reuptake inhibition % vs. vehicle [1] |
| Animal Protocol |
Dissolved in 10% hydroxypropyl-β-cyclodextrin; 10 mg/kg; s.c. administration
Rats Acute studies[2] Three doses of vortioxetine (2.5, 5 and 10 mg/kg, free base dissolved in 10% β-cyclodextrin, oral gavage, p.o.,) were used in the OF test, the NSF test and the FST. The effects of vortioxetine were compared to the vehicle control group (10% β-cyclodextrin) and also to a fluoxetine- (18 mg/kg p.o., (David et al., 2007)) and a diazepam-treated group (1.5 mg/kg, s.c. (David et al., 2007)). All doses were corrected for the weight of the salt. All treatments were administered 1 h before testing. Chronic studies[2] Two doses of vortioxetine (5 and 20 mg/kg/day, free base dissolved in 10% β-cyclodextrin, oral gavage, p.o.) were tested in mice after 14 days of administration in the NSF and 21 days of administration in the OF test, the NSF test and the FST. The mice were tested 24 h after the last dose. The effects of vortioxetine were compared to a vehicle control group (10% β-cyclodextrin) and also to a fluoxetine-treated group (18 mg/kg/day p.o.). Mouse FST & EPM Model: Male C57BL/6 mice (6–8 weeks old, 20–25 g) housed at 22±2°C (12 h light/dark). Randomized into 4 groups (n=8/group): 1. Vehicle: Oral gavage 0.5% CMC-Na (10 mL/kg/day); 2. Vortioxetine 1 mg/kg: Oral gavage Vortioxetine HBr (1 mg/kg/day, dissolved in 0.5% CMC-Na); 3. Vortioxetine 5 mg/kg: Oral gavage Vortioxetine HBr (5 mg/kg/day); 4. Vortioxetine 10 mg/kg: Oral gavage Vortioxetine HBr (10 mg/kg/day). Treated 21 days. FST: Mice placed in 50 cm×20 cm tank (25°C water) for 6 min; immobility time recorded last 4 min. EPM: Mice placed in maze (arms 30×5 cm, 15 cm height) for 5 min; open arm time recorded [2] - Mouse Hippocampal Neurogenesis Assay: Mice from FST groups received intraperitoneal BrdU (50 mg/kg/day) on days 18–21 of treatment. On day 22, mice euthanized; brains fixed in 4% paraformaldehyde. Coronal sections (30 μm) stained with anti-BrdU (FITC-conjugated) and anti-NeuN (Cy3-conjugated) antibodies. BrdU⁺NeuN⁺ cells counted in dentate gyrus via fluorescence microscopy [2] - Rat Chronic Unpredictable Stress (CUS) Model: Male Wistar rats (8 weeks old, 220–250 g) exposed to CUS (food/water deprivation, cold swim) for 14 days. Randomized into 2 groups (n=6/group): 1. CUS+Vehicle: Oral gavage 0.5% CMC-Na (10 mL/kg/day); 2. CUS+Vortioxetine: Oral gavage Vortioxetine HBr (10 mg/kg/day). Treated 21 days. On day 36, rats tested in sucrose preference test; preference ratio (sucrose intake/total fluid intake) measured [5] |
| ADME/Pharmacokinetics |
Oral bioavailability: In male Sprague-Dawley rats, the bioavailability of oral Vortioxetine HBr (10 mg/kg) was 75%, while that of intravenous (5 mg/kg) was 75% [1] - Plasma pharmacokinetics: In rats intravenously administered 5 mg/kg Vortioxetine HBr: Cmax = 2.3 μg/mL, Tmax = 5 min, t1/2 = 12 h. Oral administration of 10 mg/kg: Cmax = 1.1 μg/mL, Tmax = 2 h, t1/2 = 14 h (HPLC detection) [1] - Plasma protein binding rate: human plasma binding rate = 98.5% (ultrafiltration, concentration 0.1–10 μg/mL) [5] - Tissue distribution: mice orally administered 10 mg/kg vortioxetine hydrobromide (2 hours after administration): brain/plasma ratio = 0.8; highest concentrations were found in the liver (4.5 μg/g) and kidney (3.2 μg/g) [1]
|
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation
◉ Overview of Use During Lactation The levels of vortioxetine in breast milk appear to be very low. This is not a reason to stop breastfeeding if the mother needs to take vortioxetine. However, the infant should be closely monitored while breastfeeding and taking vortioxetine until more data are available. ◉ Effects on Breastfed Infants Three breastfeeding mothers took vortioxetine for depression; two took 10 mg daily, and one took 20 mg daily. All mothers exclusively breastfed their infants at 1, 2, and 6 months of age. No mothers reported any abnormal behavior in their infants. One woman taking 76.1 mcg/kg of vortioxetine daily partially breastfed her infant. She did not observe any adverse reactions in her infant. A Japanese postpartum woman with depression took 20 mg of vortioxetine, 10 mg of zolpidem, 20 mg of duloxetine, 100 mg of rebamipide, and 2.5 g of the Asian herbal medicine Shinki-shi-tang daily. She breastfed her infant partially (over 50%) for 3 months. During routine follow-ups at 1, 3, 5, 7, and 9 months postpartum, the infant experienced no drug-related adverse reactions. ◉ Effects on Lactation and Breast Milk Vortioxetine has caused hyperprolactinemia and galactorrhea in some patients. An observational study investigated the outcomes of 2859 women who took antidepressants for 2 years prior to pregnancy. Compared to women who did not take antidepressants during pregnancy, mothers who took antidepressants in all three stages of pregnancy were 37% less likely to breastfeed at discharge. Mothers who took antidepressants only in the third trimester were 75% less likely to breastfeed at discharge. Mothers who took antidepressants only in the first and second trimesters were not less likely to breastfeed at discharge. The study did not specify the type of antidepressant used by the mothers. A retrospective cohort study analyzed hospital electronic medical records from 2001 to 2008, comparing women taking antidepressants in late pregnancy (n = 575), women with mental illness but not taking antidepressants (n = 1552), and mothers not diagnosed with mental illness (n = 30,535). Women receiving antidepressant treatment were 37% less likely to breastfeed at discharge than women not diagnosed with mental illness, but there was no difference in the likelihood of breastfeeding compared to untreated mothers diagnosed with mental illness. None of the mothers were taking vortioxetine. A study of 80,882 Norwegian mother-infant pairs from 1999 to 2008 showed that 392 women reported starting antidepressants postpartum, and 201 women reported starting antidepressants during pregnancy. Compared to a control group not exposed to antidepressants, taking antidepressants in late pregnancy was associated with a 7% lower likelihood of initiating breastfeeding, but had no effect on the duration of breastfeeding or the rate of exclusive breastfeeding. Compared to the control group who had not been exposed to antidepressants, new use or restart of antidepressant use was associated with a 63% lower likelihood of primary breastfeeding at 6 months and a 51% lower likelihood of any breastfeeding, while also being associated with a 2.6-fold increased risk of abrupt cessation of breastfeeding. No specific antidepressant was mentioned. Acute in vivo toxicity: The LD50 of vortioxetine hydrobromide in male ICR mice administered intraperitoneally was 350 mg/kg. Doses >250 mg/kg can cause transient ataxia; no deaths were observed at doses ≤200 mg/kg [1] - Subacute toxicity: Rats were given vortioxetine hydrobromide (10, 30, 100 mg/kg/day) orally for 28 days: no weight loss (change <5%), normal serum ALT/AST/BUN/creatinine, and no liver/kidney/brain tissue pathology [5] - Human safety: Healthy volunteers (n=24) were given vortioxetine hydrobromide (10 mg/day) for 14 days: no significant changes were observed in heart rate, blood pressure, or liver function. Mild nausea (incidence 12%) resolved within 3 days [3] |
| References |
|
| Additional Infomation |
Vortioxetine hydrobromide is a hydrobromide salt prepared by reacting vortioxetine with an equimolar amount of hydrobromic acid. It is used to treat major depressive disorder. It possesses multiple effects, including antidepressant, anti-anxiety, serotonin antagonist, and serotonin agonist actions. It contains the vortioxetine (1+) ion. Vortioxetine hydrobromide is the hydrobromide form of vortioxetine, a serotonin (5-HT) modulator and agonist (SMS) with antidepressant activity. Vortioxetine inhibits the reuptake of serotonin and norepinephrine in the synaptic cleft and can act as a serotonin receptor agonist (5-HT1A), partial agonist (5-HT1B), or antagonist (5-HT3, 5-HT1D, and 5-HT7). It is currently unclear how the claimed multimodal mechanism of action of this drug promotes its antidepressant effect; however, it is speculated that it may increase the availability of serotonin and norepinephrine in the synaptic cleft.
A piperazine derivative that acts as a serotonin reuptake inhibitor, a 5-HT3 receptor antagonist, and a 5-HT1A receptor agonist. It is used to treat anxiety and depression. See also: Vortioxetine (containing the active ingredient). Drug Indications> Treatment of major depressive episodes in adults. Mechanism of Action: Vortioxetine hydrobromide exerts a multimodal effect: 1) inhibits SERT to increase extracellular 5-HT; 2) agonizes 5-HT1A (enhances serotonergic transmission); 3) antagonizes 5-HT3A (reduces nausea) and 5-HT7 (improves cognitive function) [1,5] -Therapeutic Potential: Approved for the treatment of major depressive disorder (MDD) and generalized anxiety disorder (GAD). In the MDD model, it can reverse stress-induced anhedonia; in generalized anxiety disorder (GAD), daily administration of 5 mg reduced Hamilton Anxiety Rating Scale (HAM-A) scores by 30% compared to placebo [2,4] - Chemical properties: White crystalline powder, soluble in dimethyl sulfoxide (DMSO) (25 mg/mL) and water (8 mg/mL). Stable for 72 hours at room temperature in aqueous solution at pH 4.0–8.0 [1] |
| Molecular Formula |
C18H22N2S
|
|
|---|---|---|
| Molecular Weight |
379.36
|
|
| Exact Mass |
378.076
|
|
| Elemental Analysis |
C, 56.99; H, 6.11; Br, 21.06; N, 7.38; S, 8.45
|
|
| CAS # |
960203-27-4
|
|
| Related CAS # |
Vortioxetine; 508233-74-7; Vortioxetine-d8 hydrobromide; 1253056-29-9
|
|
| PubChem CID |
56843850
|
|
| Appearance |
White to off-white solid powder
|
|
| LogP |
5.216
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
3
|
|
| Rotatable Bond Count |
3
|
|
| Heavy Atom Count |
22
|
|
| Complexity |
316
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
Br.S(C1C(C)=CC(C)=CC=1)C1C(N2CCNCC2)=CC=CC=1
|
|
| InChi Key |
VNGRUFUIHGGOOM-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C18H22N2S.BrH/c1-14-7-8-17(15(2)13-14)21-18-6-4-3-5-16(18)20-11-9-19-10-12-20;/h3-8,13,19H,9-12H2,1-2H3;1H
|
|
| Chemical Name |
1-[2-(2,4-dimethylphenyl)sulfanylphenyl]piperazine;hydrobromide
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.59 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.59 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (6.59 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 15% Captisol, pH 9: 10 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6360 mL | 13.1801 mL | 26.3602 mL | |
| 5 mM | 0.5272 mL | 2.6360 mL | 5.2720 mL | |
| 10 mM | 0.2636 mL | 1.3180 mL | 2.6360 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Vortioxetine for the Treatment of Hoarding Disorder
CTID: NCT04035850
Phase: Phase 3 Status: Withdrawn
Date: 2024-02-02
 |
|---|
|
|