
| Size | Price | Stock | Qty |
|---|---|---|---|
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| 5g |
|
||
| 10g |
|
||
| 25g | |||
| Other Sizes |
Purity: ≥98%
Tropisetron HCl (Navoban; ICS-205930; ICS 205-930), the hydrochloride salt of Tropisetron which is an anti-emetic drug, is a novel, potent and selective 5-HT3 receptor antagonist and α7-nicotinic receptor agonist that has been approved for the treatment of chemotherapy-induced nausea and vomiting. Tropisetron binds with great affinity to both 5HT3R and th α7 nAChR. With respect to the other nicotinic subtypes tested, tropisetron has very little affinity.
| Targets |
5-HT3 Receptor ( IC50 = 70.1 nM )
|
||
|---|---|---|---|
| ln Vitro |
|
||
| ln Vivo |
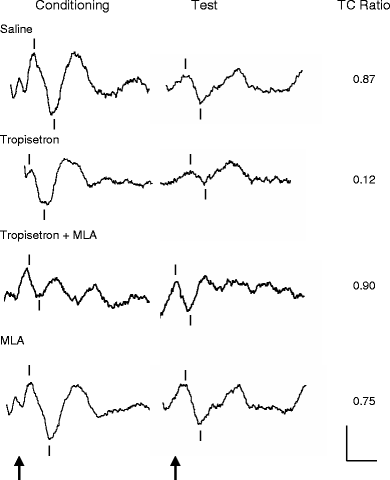
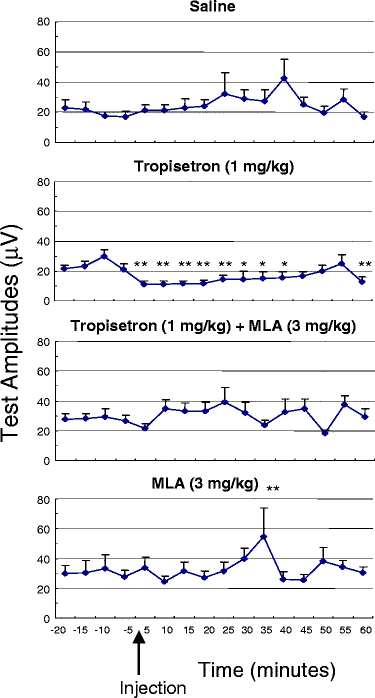
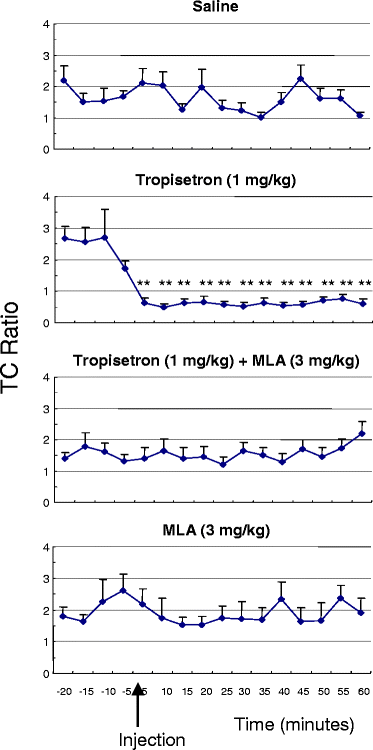
Tropisetron (1 mg/kg i.p.) dramatically improves the impaired inhibitory processing of the P20-N40 auditory evoked potential in DBA/2 mice.[5]
Tropisetron is a potent and selective serotonin 3 (5-hydroxytryptamine3; 5-HT3) receptor antagonist with antiemetic properties, probably mediated via antagonism of receptors both at peripheral sites and in the central nervous system. When compared with antiemetic regimens containing high-dose metoclopramide in a small number of studies, tropisetron was generally as effective at preventing acute and delayed vomiting induced by high-dose cisplatin (> or = 50 mg/m2). In these studies tropisetron completely prevented vomiting occurring in the first 24 hours after chemotherapy in 35 to 76% of patients. Tropisetron was superior to alizapride in preventing emesis induced by high-dose alkylating agents. The effectiveness of tropisetron in patients who had previously had partial control of emesis was improved by the addition of dexamethasone. Tropisetron appears to be well tolerated with the most frequently reported adverse effect being headache. Extrapyramidal effects, which can occur in 5 to 10% of patients receiving high-dose metoclopramide and which may limit its use, have been reported in only isolated cases with tropisetron. Thus, tropisetron is an effective, apparently well tolerated agent which can be administered once daily for the prevention of chemotherapy-induced nausea and vomiting. However, further clinical experience is needed to clarify the optimum role of tropisetron as an antiemetic agent, particularly with regard to other drugs in its class. Nonetheless, preliminary results indicate that tropisetron will be a useful alternative for use in controlling emesis induced by cytotoxic therapy. [2] Administration of tropisetron (1 mg/kg i.p.) significantly improved the deficient inhibitory processing of the P20-N40 auditory evoked potential in DBA/2 mice. Coadministration of methyllycaconitine (MLA; 3 mg/kg i.p.), a partially selective antagonist at alpha 7 nicotinic receptors, significantly blocked the normalizing effect of tropisetron. Furthermore, MLA alone did not alter the deficient inhibitory processing of the P20-N40 auditory evoked potential in DBA/2 mice. Conclusions: The data suggest that tropisetron improves the deficient inhibitory processing of the P20-N40 auditory evoked potential in DBA/2 mice by effects on alpha 7 and perhaps alpha 4 beta 2 nicotinic receptors. Tropisetron may be useful for the treatment of deficient inhibitory processing in schizophrenia [5]. |
||
| Enzyme Assay |
The 5-HT3 receptor antagonist tropisetron (ICS 205-930) was found to be a potent and selective partial agonist at alpha7 nicotinic receptors. Two other 5-HT3 receptor antagonists, ondansetron and LY-278,584, were found to lack high affinity at the alpha7 nicotinic receptor. Quinuclidine analogues (1 and 2) of tropisetron were also found to be potent and selective partial agonists at alpha7 nicotinic receptors. [1]
Tropisetron, an antagonist of serotonin type 3 receptor, has been investigated in chronic inflammatory joint process. Since T cells play a key role in the onset of several inflammatory diseases, we have evaluated the immunosuppressive activity of tropisetron in human T cells, discovering that this compound is a potent inhibitor of early and late events in TCR-mediated T cell activation. Moreover, we found that tropisetron specifically inhibited both IL-2 gene transcription and IL-2 synthesis in stimulated T cells. To further characterize the inhibitory mechanisms of tropisetron at the transcriptional level, we examined the DNA binding and transcriptional activities of NF-(kappa)B, NFAT and AP-1 transcription factors in Jurkat T cells. We found that tropisetron inhibited both the binding to DNA and the transcriptional activity of NFAT and AP-1. We also observed that tropisetron is a potent inhibitor of PMA plus ionomycin-induced NF-(kappa)B activation but in contrast TNF(alpha)-mediated NF-(kappa)B activation was not affected by this antagonist. Finally, overexpression of a constitutively active form of calcineurin indicated that this phosphatase may represent one of the main targets for the inhibitory activity of tropisetron. These findings provide new mechanistic insights into the anti-inflammatory activities of tropisetron, which are probably independent of serotonin receptor signalling and highlight their potential to design novel therapeutic strategies to manage inflammatory diseases [3]. |
||
| Cell Assay |
Cell survival was increased to an average of 105% in retinal ganglion cells (RGCs) pretreated with 100 nM tropisetron before glutamate, in comparison to controls. Tropisetron is thought to be an efficient neuroprotective agent against glutamate-induced excitotoxicity, as it is mediated by α7 nAChR activation, according to inhibition studies conducted with the alpha7 nAChR antagonist MLA (10 nM2). p38 MAPK levels linked to excitotoxicity were dramatically reduced by tropisetron from an average of 15 ng/ml to 6 ng/ml, while pAkt levels were not affected in any appreciable way. Tropisetron over-expresses the CB(1) receptors at both transcriptional and protein levels, inhibits the phosphatase activity of calcineurin significantly (but not granisetron), and lowers the amount of cAMP in cerebellar granule neurons
|
||
| Animal Protocol |
|
||
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Topisetron is rapidly absorbed from the gastrointestinal tract (mean half-life approximately 20 minutes) and is nearly completely absorbed (over 95%). Due to first-pass metabolism in the liver, the absolute bioavailability of a 5 mg oral dose is 60%. Peak plasma concentrations are reached within 3 hours. Approximately 8% of topisetron is excreted unchanged in the urine, 70% as metabolites, and 15% in the feces. Flow rate: 400-600 liters. Flow rate: 1800 ml/min. Metabolism/Metabolites Topisetron is metabolized via hydroxylation at the 5, 6, or 7 position of its indole ring, followed by conjugation with glucuronic acid or sulfate, and excreted in the urine or bile (urine to fecal ratio 5:1). These metabolites have significantly reduced potency against 5-HT3 receptors and do not contribute to the pharmacological action of the drug. The known human metabolites of topisetron include 8-azabicyclo[3.2.1]oct-3-yl 1H-indole-3-carboxylate, (8-methyl-8-azabicyclo[3.2.1]oct-3-yl)5-hydroxy-1H-indole-3-carboxylate, and (8-methyl-8-azabicyclo[3.2.1]oct-3-yl)6-hydroxy-1H-indole-3-carboxylate. Biological half-life 5.7 hours. |
||
| Toxicity/Toxicokinetics |
Protein Binding
71% binds to plasma proteins in a non-specific manner. Rats oral LD50: 265 mg/kg, Monthly Pharmacy, 40(2445), 1998 Rats intravenous LD50: 31400 μg/kg, Monthly Pharmacy, 40(2445), 1998 Mice oral LD50: 487 mg/kg, Monthly Pharmacy, 40(2445), 1998 Mice intravenous LD50: 37900 μg/kg, Monthly Pharmacy, 40(2445), 1998 |
||
| References | |||
| Additional Infomation |
Topisetron is an indole carboxylic acid ester formed by the condensation of the carboxyl group of indole-3-carboxylic acid with the hydroxyl group of tropine. It possesses a variety of pharmacological effects, including serotonergic antagonism, antiemetic, nicotinic acetylcholine receptor agonist, trypanosome killer, immunomodulator, neuroprotective agent, apoptosis inhibitor, and anti-inflammatory agent. It is an indole carboxylic acid ester, an azabicycloalkane, and a tertiary amine compound. Its structure is related to indole-3-carboxylic acid and tropine. It is the conjugate base of topisetron (1+). Topisetron is an indole derivative with antiemetic activity. As a selective serotonin receptor antagonist, topisetron inhibits chemotherapy- and radiotherapy-induced nausea and vomiting by competitively blocking the binding of serotonin to 5-HT3 receptors. Topisetron is well-tolerated, with headache being the most common adverse reaction. Extrapyramidal side effects are rare after using topisetron. Topisetron is an indole derivative with antiemetic activity. As a selective serotonin receptor antagonist, topisetron inhibits chemotherapy- and radiotherapy-induced nausea and vomiting by competitively blocking the binding of serotonin to 5-HT3 receptors. (NCI04)
An indole derivative and 5-HT3 receptor antagonist for the prevention of nausea and vomiting. Pharmaceutical Indications For the prevention of nausea and vomiting induced by cytotoxic therapy and postoperatively. Mechanism of Action Topisetron competitively binds to 5-HT3 receptors, blocking the action of serotonin on 5-HT3 receptors at the peripheral vagal nerve endings in the gastrointestinal tract and in the chemoreceptor trigger zone (CTZ) of the postmedulatum in the central nervous system. This results in the suppression of chemotherapy- and radiotherapy-induced nausea and vomiting. In summary, this study demonstrates that topisetron improves the deficiency in inhibitory processing of the P20-N40 auditory evoked potential in DBA/2 mice through α7 (and possibly α4β2) nicotine receptors. Therefore, topisetron (Koike et al., 2005), like clozapine, is a drug that can enhance inhibitory processing of auditory evoked potentials in humans (Nagamoto et al., 1996, 1999) and animals (Simosky et al., 2003), and both drugs appear to have a common end effect on nicotine receptors, particularly the α7 subtype. If the correlation between sensory inhibition failure and neurophysiological measurements of attentional dysfunction in patients with schizophrenia (Cullum et al., 1993) reflects a common neurobiological mechanism, then 5-HT3 antagonists, such as topisetron and ondansetron, may provide a treatment option for attentional problems and even cognitive difficulties frequently experienced by patients with schizophrenia. [5] |
| Molecular Formula |
C17H21CLN2O2
|
|---|---|
| Molecular Weight |
320.81
|
| Exact Mass |
320.129
|
| Elemental Analysis |
C, 64.57; H, 6.92; Cl, 10.59; N, 8.37; O, 9.56
|
| CAS # |
105826-92-4
|
| Related CAS # |
Tropisetron; 89565-68-4
|
| PubChem CID |
656665
|
| Appearance |
White to off-white solid powder
|
| Boiling Point |
448.5ºC at 760 mmHg
|
| Melting Point |
283-285ºC
|
| Flash Point |
225ºC
|
| Vapour Pressure |
3.09E-08mmHg at 25°C
|
| LogP |
2.83
|
| Hydrogen Bond Donor Count |
1
|
| Hydrogen Bond Acceptor Count |
3
|
| Rotatable Bond Count |
3
|
| Heavy Atom Count |
21
|
| Complexity |
400
|
| Defined Atom Stereocenter Count |
2
|
| SMILES |
Cl[H].O(C(C1=C([H])N([H])C2=C([H])C([H])=C([H])C([H])=C12)=O)C1([H])C([H])([H])[C@]2([H])C([H])([H])C([H])([H])[C@]([H])(C1([H])[H])N2C([H])([H])[H]
|
| InChi Key |
XIEGSJAEZIGKSA-KOQCZNHOSA-N
|
| InChi Code |
InChI=1S/C17H20N2O2.ClH/c1-19-11-6-7-12(19)9-13(8-11)21-17(20)15-10-18-16-5-3-2-4-14(15)16;/h2-5,10-13,18H,6-9H2,1H3;1H/t11-,12+,13?;
|
| Chemical Name |
[(1R,5S)-8-methyl-8-azabicyclo[3.2.1]octan-3-yl] 1H-indole-3-carboxylate;hydrochloride
|
| Synonyms |
ICS-205-930; Tropisetron; ICS-205930; 105826-92-4; Tropisetron hydrochloride; TROPISETRON HCl; Tropisetron monohydrochloride; Tropisetron (Hydrochloride); (1R,3r,5S)-8-methyl-8-azabicyclo[3.2.1]octan-3-yl 1H-indole-3-carboxylate hydrochloride; SDZ-ICS-930; [(1R,5S)-8-methyl-8-azabicyclo[3.2.1]octan-3-yl] 1H-indole-3-carboxylate;hydrochloride; ICS 205930; ICS205930; ICS 205-930; Tropisetron HCl; Brand name: Navoban
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: 120 mg/mL (374.05 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication.
(Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1171 mL | 15.5855 mL | 31.1711 mL | |
| 5 mM | 0.6234 mL | 3.1171 mL | 6.2342 mL | |
| 10 mM | 0.3117 mL | 1.5586 mL | 3.1171 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT05564286 | Recruiting | Drug: Fosaprepitant Drug: tropisetron |
Cervical Cancer Antiemetic |
Shantou University Medical College |
July 1, 2021 | Phase 3 |
| NCT05533281 | Recruiting | Drug: tropisetron Drug: metoclopramide |
Nausea and Vomiting | The Second Affiliated Hospital of Chongqing Medical University |
September 15, 2022 | Early Phase 1 |
| NCT05242874 | Active Recruiting |
Drug: Fosaprepitant , Tropisetron and Olanzapine Drug: Fosaprepitant , Tropisetron, Dexamethasone and Olanzapine |
Chemotherapy-induced Nausea and Vomiting |
Henan Cancer Hospital | January 1, 2022 | Phase 3 |
| NCT04817189 | Recruiting | Drug: NEPA (300mg netupitant /0.5mg palonosetron) Drug: Dexamethasone, 8 mg (oral) or equivalent IV dose |
Chemotherapy-induced Nausea and Vomiting |
Helsinn Healthcare SA | February 1, 2021 | Phase 4 |
| NCT00435370 | Completed | Drug: Tropisetron Drug: Placebo |
Smoking Cessation Schizophrenia |
Baylor College of Medicine | November 2006 | Phase 3 |
 |
|---|
 |
 |