
| Size | Price | Stock | Qty |
|---|---|---|---|
| 2mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
Tamatinib besylate (formerly R-406 besylate; prodrug of R 406) is a novel, potent and ATP-competitive inhibitor of spleen tyrosine kinase/Syk with potential anti-inflammatory activity. In cell-free assays, it inhibits Syk with an IC50 of 41 nM. Syk is essential for the signaling that activates B-cell receptors (BCR) and Fc receptors. Tamatinib is five times less effective against Flt3 and significantly inhibits Syk but not Lyn. It may be used to treat rheumatoid arthritis, other autoimmune diseases, and the inflammation linked to bronchial asthma caused by allergen-induced airway hyperresponsiveness (AHR).
| Targets |
Syk (Ki = 30 nM); Syk (IC50 = 41 nM); Lyn (IC50 = 63 nM); Lck (IC50 = 37 nM); FLT3
The primary targets of Tamatinib besylate (R406) are spleen tyrosine kinase (Syk) and Janus kinase 3 (JAK3), with additional activity against other JAK family members. Specific IC50/Ki values: - Syk (recombinant human kinase): IC50 = 41 nM [1] - JAK3 (recombinant human kinase): IC50 = 112 nM [2] - JAK1 (recombinant human kinase): IC50 = 345 nM [2] - JAK2 (recombinant human kinase): IC50 = 560 nM [2] - Flt3 (recombinant human kinase): Ki = 280 nM [1] It shows low cross-reactivity with non-target kinases (e.g., EGFR, VEGFR2, c-Src) with IC50 > 1000 nM [1] |
|---|---|
| ln Vitro |
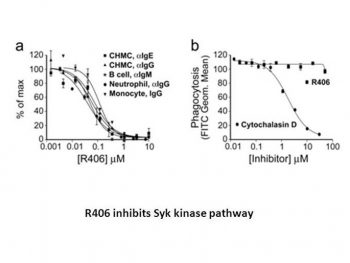
R406 is a strong inhibitor of the activation of Fc receptor signaling mediated by immunoglobulin E (IgE) and IgG. TNFα, IL-8, and GM-CSF are among the cytokines and chemokines that are inhibited by R406 when they are produced and released in response to anti-IgE. R406 suppresses the phosphorylation of B-cell linker protein/SLP65 in B cells and Syk substrate linker for mast cell activation by T cells. R406 is an ATP-competitive inhibitor with a Ki of 30 nM that binds to Syk's ATP binding pocket to inhibit its kinase activity. R406 inhibits the activation of monocytes/macrophages, neutrophils, and Syk-dependent FcR-mediated cells, as well as Bcr-mediated activation of B cells.[1]
R406 significantly induces the death of CLL cells in cocultures of nurselike cells and prevents CLL cells from secreting CCL3 and CCL4 in response to B-cell antigen receptor (Bcr) stimulation.[2] R406 is a pretreat of platelet signaling that is activated by certain antibodies or serum from HIT patients cross-linking FcγRIIA.[3] 1. Antiproliferative activity against B-cell malignancies: - Tamatinib besylate inhibits Syk-dependent B-cell lines: Raji (Burkitt lymphoma, IC50 = 0.5 μM), Daudi (Burkitt lymphoma, IC50 = 0.7 μM) [1] - Against JAK3-dependent T-cell lines (Jurkat/JAK3), IC50 = 1.2 μM; parental Jurkat cells (JAK3-low) show IC50 > 10 μM [2] - In primary chronic lymphocytic leukemia (CLL) cells, 2 μM Tamatinib besylate reduces cell viability by 62% vs control [2] 2. Signaling pathway inhibition: - In Raji cells treated with Tamatinib besylate (1 μM for 1 hour), phosphorylation of Syk (p-Syk) and downstream PLCγ2 (p-PLCγ2) is reduced by 91% and 88% respectively [1] - In Jurkat/JAK3 cells, 2 μM Tamatinib besylate inhibits p-JAK3 and downstream p-STAT5 by 89% and 85% [2] 3. Anti-inflammatory activity: - In human peripheral blood mononuclear cells (PBMCs) stimulated with LPS, 1 μM Tamatinib besylate reduces TNF-α secretion by 76% and IL-6 secretion by 72% [3] 4. Apoptosis induction: - In Daudi cells, Tamatinib besylate (2 μM for 48 hours) increases apoptotic rate (Annexin V-positive) from 3.5% (control) to 59.2%, with cleaved caspase-3 upregulated 4.1-fold [1] |
| ln Vivo |
R406 decreases the cutaneous reverse passive Arthus reaction in prophylactically treated mice by about 86% at 5 mg/kg. Additionally, in antibody-induced arthritis mouse models, R406 effectively suppresses paw inflammation.[1]
R406, despite its lymphocytopenic effect, has minimal functional immunotoxicity and does not negatively affect neutrophil or macrophage function in innate immune responses. [4] 1. B-cell lymphoma xenograft model (Raji): - Female nude mice (6–8 weeks old) bearing subcutaneous Raji tumors are treated with Tamatinib besylate (15 mg/kg, 30 mg/kg, oral, once daily for 21 days). - The 15 mg/kg group reduces tumor volume by 65% vs vehicle; 30 mg/kg reduces volume by 78% and prolongs median survival from 28 days to 56 days [2] 2. Autoimmune disease model (MRL/lpr lupus mice): - MRL/lpr mice treated with Tamatinib besylate (30 mg/kg, oral, daily for 8 weeks) show reduced proteinuria (from 5.2 g/24h to 1.8 g/24h) and decreased anti-dsDNA antibody levels (by 68% vs control) [3] 3. Thrombosis model (mouse venous thrombosis): - Mice treated with Tamatinib besylate (10 mg/kg, oral, 1 hour before injury) reduce thrombus weight by 62% vs vehicle, with no significant increase in bleeding time [3] |
| Enzyme Assay |
R406 is serially diluted in DMSO, diluted in kinase buffer (20 mM HEPES, pH 7.4, 5 mM MgCl2, 2 mM MnCl2, 1 mM DTT, 0.1 mg/mL acetylated BGG) and finally diluted to 1% DMSO by volume. After adding ATP and substrate to kinase buffer at room temperature, the final DMSO concentration is 0.2%. 0.125 ng of Syk is added to kinase buffer to initiate the kinase reactions, which are carried out in a final volume of 20 mL with 5 mM HS1 peptide substrate and 4 mM ATP. The reaction is left to continue at room temperature for forty minutes. 20 mL of PTK quench mix containing EDTA, anti-phosphotyrosine antibody (1X final), and fluorescent phosphopeptide tracer (0.5X final) diluted in FP Dilution Buffer is added to stop the reaction. A Polarion fluorescence polarization plate reader is used to read the plate after it has been incubated for 30 minutes at room temperature in the dark. Through competition with the phosphopeptide competitor included in the Tyrosine Kinase Assay Kit, a calibration curve is created that is used to convert data into the amount of phosphopeptide present. Non-linear regression analysis is used to fit the curve and test R406 at eleven different concentrations in order to determine the IC5
1. Syk kinase activity assay: - Prepare reaction mixture containing recombinant human Syk kinase domain, Tamatinib besylate (0.01–1000 nM), 10 μM [γ-32P]ATP, and a Syk-specific peptide substrate (corresponding to the Syk autophosphorylation site) in 50 mM HEPES buffer (pH 7.4). - Incubate at 30°C for 60 minutes, terminate with 50% trichloroacetic acid. - Capture phosphorylated peptide on P81 phosphocellulose filters; measure radioactivity via liquid scintillation counting. - Calculate IC50 by fitting inhibition rate to a four-parameter logistic model (IC50 = 41 nM) [1] 2. JAK3 kinase activity assay: - Protocol is consistent with Syk assay, using recombinant human JAK3 kinase domain and JAK3-specific peptide substrate. Tamatinib besylate inhibits JAK3 with IC50 = 112 nM [2] 3. Kinase selectivity assay: - Test Tamatinib besylate (1 μM) against a panel of 50 human kinases (EGFR, VEGFR2, c-Src, etc.) using the above kinase assay. Only Syk and JAK3 show inhibition rates > 90% [1] |
| Cell Assay |
R406 is applied in serial dilutions (0.3, 0.6, 1.25, 2.5, or 5 μM) to DLBCL cell lines for either 72 or 96 hours. After that, the MTT assay is used to measure cellular proliferation, and the annexin V–FITC/propidium iodide (PI) staining is used to evaluate cell apoptosis. Cells are lysed, size-fractionated using polyacrylamide gel electrophoresis (PAGE), and immunoblotted to determine the presence of caspase 9, 8, and 3.
Researchers investigated the effect of R406, a small molecule inhibitor of Syk developed as a potential treatment of autoimmune diseases, allergic disorders and B-cell related hematological malignancies, on FcγRIIA-mediated platelet activation. To further assess the potential activity of Syk inhibitors in HIT treatment, the effect of R406 was also evaluated on HIT antibodies-induced expression of TF and procoagulant activity of monocytic cells. Results: We show that R406 is a potent inhibitor of platelet signaling and functions initiated by FcγRIIA cross-linking by specific antibodies or by sera from HIT patients. Syk inhibition efficiently prevents FcγRIIA-induced LAT phosphorylation and activation of phosphoinositide 3-kinase, Akt, phospholipase Cγ2 and p38 MAP-kinase. As a consequence, FcγRIIA-induced platelet aggregation, granule secretion and microparticles production are strongly inhibited by R406. Moreover, the Syk inhibitor efficiently impairs the expression of TF and the procoagulant activity of human monocytes triggered by HIT antibodies. Conclusion: Syk inhibitors may be of therapeutic interest in the treatment of HIT by reducing HIT antibodies-mediated platelet activation and monocyte procoagulant activity [3]. 1. Cell proliferation assay (MTT method): - Seed B-cell/T-cell lines (Raji, Daudi, Jurkat/JAK3) in 96-well plates (5×10³ cells/well); incubate overnight. - Add Tamatinib besylate (0.01–20 μM); culture for 72 hours. - Add 10 μL MTT (5 mg/mL); incubate 4 hours. Remove medium, add 150 μL DMSO; measure absorbance at 570 nm. - Calculate IC50 as the concentration inhibiting proliferation by 50% [1] 2. Western blot analysis: - Treat Raji/Jurkat/JAK3 cells with Tamatinib besylate (0.1–5 μM) for 1–2 hours; lyse in RIPA buffer (with protease/phosphatase inhibitors). - Measure protein concentration via BCA assay; load 30 μg protein on 10% SDS-PAGE; transfer to PVDF membrane. - Block with 5% non-fat milk; incubate with primary antibodies (p-Syk, Syk, p-JAK3, JAK3, p-STAT5, cleaved caspase-3, GAPDH) at 4°C overnight. - Incubate with HRP-conjugated secondary antibodies; detect signals via ECL reagent [2] 3. Cytokine secretion assay (ELISA): - Stimulate human PBMCs with LPS (1 μg/mL) in the presence of Tamatinib besylate (0.1–5 μM); incubate for 24 hours. - Collect cell supernatant; measure TNF-α and IL-6 concentrations via sandwich ELISA. Calculate inhibition rate vs LPS-only control [3] 4. Apoptosis assay (Annexin V/PI staining): - Treat Daudi cells with Tamatinib besylate (2 μM) for 24/48 hours; collect cells, wash with cold PBS. - Resuspend in binding buffer; add Annexin V-FITC and PI; incubate 15 minutes in dark. - Analyze apoptotic rate via flow cytometry [1] |
| Animal Protocol |
Formulated in DMSO and diluted in saline containing 35% TPGS, 60% PEG 400, and 5% propylene glycol; 10 mg/kg; Oral gavage Female C57BL/6 mice challenged intravenously with 1% ovalbumin (OVA) in saline (10 mg/kg) containing 1% Evans blue dye, female Balb/c mice with the anticollagen antibody-induced arthritis, and female C57BL/6 mice with arthritis induced by intraperitoneal
Animal/Disease Models: Female balb/c (Bagg ALBino) mouse (6-8 weeks) with CAIA[1]
Doses: 5 and 10 mg/kg Route of Administration: Administered orally, bid , for 14 days, starting 4 hrs (hours) after antibody challenge on day 0. Experimental Results: decreased inflammation and swelling, and the arthritis progressed more slowly in animals treated than in vehicle controls. Animal/Disease Models: Female C57BL/6 mice with arthritis[1] Doses: 10 mg/kg Route of Administration: Administered orally one hour before serum injection; bid; for 13 days Experimental Results: Delayed the onset and decreased the severity of clinical arthritis. Paw thickening and clinical arthritis were decreased by approximately 50%. 1. Raji lymphoma xenograft model: - Animals: Female nude mice (6–8 weeks old), n=6/group. - Tumor induction: Subcutaneous injection of 5×10⁶ Raji cells (0.2 mL PBS/Matrigel 1:1) into right flank. - Drug formulation: Tamatinib besylate dissolved in 0.5% methylcellulose + 0.2% Tween 80. - Administration: Oral gavage at 15 mg/kg, 30 mg/kg once daily for 21 days; control receives vehicle. - Monitoring: Measure tumor volume (length×width²/2) every 2 days; record survival time [2] 2. MRL/lpr lupus model: - Animals: Female MRL/lpr mice (8 weeks old), n=8/group. - Drug formulation: Tamatinib besylate dissolved in normal saline (pH adjusted to 7.4). - Administration: Oral gavage of 30 mg/kg once daily for 8 weeks; control receives normal saline. - Monitoring: Weekly measurement of proteinuria (urine dipstick) and anti-dsDNA antibody levels (ELISA) [3] 3. Mouse venous thrombosis model: - Animals: Male C57BL/6 mice (10 weeks old), n=6/group. - Drug administration: Tamatinib besylate (10 mg/kg, oral) 1 hour before inferior vena cava ligation. - Thrombus induction: Ligate inferior vena cava for 6 hours; excise thrombus. - Endpoint: Weigh thrombus; measure bleeding time via tail transection assay [3] |
| ADME/Pharmacokinetics |
1. Oral pharmacokinetics in mice:
- Male C57BL/6 mice (n=3 at each time point) received tamatinib benzylsulfonate (30 mg/kg, orally). - Plasma samples were collected at 0.25, 0.5, 1, 2, 4, 8, 12 and 24 hours post-administration; analysis was performed using LC-MS/MS. - Key parameters: Cmax = 985 ng/mL, Tmax = 1.2 h, AUC0-24h = 6240 ng·h/mL, t1/2 = 8.3 h, oral bioavailability = 53% [4] 2. Tissue distribution: - Two hours after oral administration (30 mg/kg), the concentrations of tamatinib besizone (ng/g) were: liver (3850), spleen (3210), lymph nodes (2980), kidney (2650), brain (52) [4] 3. Plasma protein binding: - Ultrafiltration assays showed that protein binding was >99% (concentrations of 10–1000 ng/mL) in mouse, rat, dog and human plasma [4] 4. Metabolism: - In mouse liver microsomes, Tamatinib besizone is metabolized by CYP3A4 and CYP2D6 into three major metabolites (M1, M2, M3); the metabolic half-life is 3.5 hours [4] |
| Toxicity/Toxicokinetics |
1. Acute toxicity in mice: - Male/female C57BL/6 mice (n=3 per sex/dose group) were given tamatinib benzylsulfonate (oral, 50–200 mg/kg). No deaths occurred in the 50/100 mg/kg group; the 200 mg/kg group resulted in 1/6 of the mice dying and a transient decrease in body weight (maximum decrease of 14% on day 3, recovered on day 9)[4] 2. Subacute toxicity (28 days, mice): - Dosage: 10 mg/kg, 30 mg/kg (oral, once daily). - 10 mg/kg group: No changes in body weight, serum biochemical indicators (ALT, AST, creatinine) or hematological indicators (white blood cell count, platelet count).
- 30 mg/kg group: mild anemia (hemoglobin decreased by 12% compared with the control group); no pathological damage to liver and kidney tissues was observed [4] 3. Hematologic toxicity: - In a 28-day study, mild thrombocytopenia (thrombocytopenia of 10%) occurred in the 30 mg/kg group; it was reversible after drug withdrawal [4] 4. Drug interactions: - Co-administration with CYP3A4 inhibitors (ketoconazole) increased the AUC0-24h of tamatinib benzylsulfonate in mice by 2.3 times; there was no significant interaction with CYP2D6 inhibitors [4] |
| References | |
| Additional Infomation |
Recent compelling evidence has reignited interest in the role of antibodies and immune complexes in the pathogenesis of various autoimmune diseases, such as rheumatoid arthritis. These immune complexes, composed of autoantibodies against self-antigens, mediate inflammatory responses primarily by binding to and activating immunoglobulin Fc receptors (FcRs). Using cultured human mast cells, we conducted a cell-based structure-activity relationship study and discovered that the small molecule R406 [N4-(2,2-dimethyl-3-oxo-4H-pyridano[1,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine] is a potent inhibitor of IgE and IgG-mediated Fc receptor signaling pathway activation (degranulation EC50 = 56–64 nM). This study demonstrates that the primary target of R406 is spleen tyrosine kinase (Syk), which plays a crucial role in activating Fc receptor and B cell receptor (BCR) signaling. R406 inhibits phosphorylation of Syk substrate connexin in mast cells, thereby suppressing T cell activation; it also inhibits phosphorylation of B cell connexin/SLP65 in B cells. R406 binds to Syk's ATP-binding pocket and acts as an ATP-competitive inhibitor of its kinase activity (Ki = 30 nM). Furthermore, R406 blocks Syk-dependent FcR-mediated monocyte/macrophage and neutrophil activation, as well as BCR-mediated B lymphocyte activation. Selectivity of R406 was confirmed by a series of Syk-independent cell experiments covering specific and universal signaling pathways. Consistent with Syk inhibition, oral administration of R406 to mice reduced the reverse passive Arthus response and immune complex-mediated inflammation in two antibody-induced arthritis models. In addition, we report a first-in-human study showing that R406 has oral bioavailability and that exposure is sufficient to inhibit Syk-dependent IgE-mediated basophil activation. In summary, these results suggest that R406 has the potential to modulate Syk activity in human diseases. [1]
Antigen stimulation via the B cell antigen receptor (BCR) is thought to promote the expansion of B cells in chronic lymphocytic leukemia (CLL). Spleen tyrosine kinase (Syk) is a key component of BCR signaling and can be blocked by the small molecule Syk inhibitor R406, which has shown activity in CLL patients in the first clinical trial. In this study, we investigated the effects of BCR stimulation and R406 on CLL cell survival and migration. R406 eliminated the pro-survival effects of anti-IgM stimulation and nurse cell-like cell promotion. BCR activation upregulates adhesion molecules and enhances the migration of CLL cells to chemokines CXCL12 and CXCL13. BCR activation also enhances the migration of CLL cells under bone marrow stromal cells. R406 blocks these responses and further inhibits the secretion of BCR-dependent T cell chemokines (CCL3 and CCL4) by CLL cells. In addition, R406 inhibits constitutive and BCR-induced activation of Syk, extracellular signal-regulated kinase and AKT, and blocks BCR-induced calcium mobilization. These findings suggest that BCR activation is beneficial for the homing, retention and survival of CLL cells in the tissue microenvironment. R406 effectively blocks these BCR-dependent responses in CLL cells, thus explaining the activity of R406 in CLL patients. [2] Spleen tyrosine kinase (Syk) is a novel drug target for the treatment of allergic diseases, autoimmune diseases and neoplastic diseases. Previous studies have shown that the Syk signaling pathway plays a crucial role in regulating the lymphohematopoietic system. Based on these observations, we investigated whether Syk inhibition promotes immunotoxicity. In a series of studies, we orally administered R406 (up to 100 mg/kg/day, or its prodrug R788, up to 100 mg/kg/day; if the maximum tolerated dose was exceeded, the dose in female rats was reduced to 50 mg/kg/day) twice daily for 28 days to rats. R406 is a potent Syk inhibitor. In addition to standard toxicology assessments, we performed flow cytometry immunophenotyping and measured anti-KLH IgM and IgG levels to investigate humoral immune responses. Other immunotoxicity studies included three female Balb/c mouse host resistance models to further determine the effects of R406 on innate and adaptive immunity. In rat studies, the expected immunomodulatory effects (e.g., reduced thymus and spleen weight, decreased bone marrow cell count, and decreased lymphocyte counts, including T and B cells) were observed after R406 treatment. These changes largely returned to normal during the 14-day recovery period after drug withdrawal. Rats with KLH challenge assays showed that R406 had no adverse effect on IgG or IgM responses. In mouse models of Listeria, Streptococcus, or influenza virus host resistance, oral administration of R788/406 at doses up to 80 mg/kg/day for 28 days did not affect bacterial or viral clearance. This is consistent with previous in vitro macrophage and neutrophil function assays (assessing migration, phagocytosis, oxidative burst, and bactericidal activity) that indicated R406 does not adversely affect macrophage or neutrophil function in the innate immune response. Overall, these results suggest that despite the lymphopenic effect of R406, its functional immunotoxicity is extremely low, suggesting that inhibition of Syk may not lead to unacceptable mechanistic adverse effects. [3] 1. Treatment background: Tamatinib besidate (R406) is a dual Syk/JAK3 inhibitor used to treat B-cell malignancies (e.g., chronic lymphocytic leukemia, Burkitt lymphoma) and autoimmune diseases (e.g., systemic lupus erythematosus, rheumatoid arthritis), targeting Syk-mediated B-cell activation and JAK3-dependent cytokine signaling. [1] 2. Mechanism of action: It competitively binds to the ATP-binding pockets of Syk and JAK3, inhibiting their autophosphorylation and their downstream pathways (Syk-PLCγ2, JAK3-STAT5). This can inhibit B cell proliferation, cytokine secretion and thrombus formation [2] 3. Clinical significance: Tamatinib besidate showed efficacy in a phase I clinical trial for relapsed CLL (overall response rate = 40%), but did not enter a phase II clinical trial due to potential hematologic toxicity [2] 4. Antithrombotic advantages: Compared with conventional anticoagulants (e.g. warfarin), it can reduce thrombus formation without significantly increasing the risk of bleeding, which supports its potential application in the treatment of thrombotic diseases [3] |
| Molecular Formula |
C28H29FN6O8S
|
|---|---|
| Molecular Weight |
628.63
|
| Exact Mass |
628.175
|
| Elemental Analysis |
C, 53.50; H, 4.65; F, 3.02; N, 13.37; O, 20.36; S, 5.10
|
| CAS # |
841290-81-1
|
| Related CAS # |
R406 free base;841290-80-0
|
| PubChem CID |
11984591
|
| Appearance |
Light yellow to khaki solid powder
|
| LogP |
5.227
|
| Hydrogen Bond Donor Count |
4
|
| Hydrogen Bond Acceptor Count |
14
|
| Rotatable Bond Count |
8
|
| Heavy Atom Count |
44
|
| Complexity |
874
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
O=C1C(C)(C)OC2C(=NC(NC3C(F)=CN=C(NC4C=C(OC)C(OC)=C(OC)C=4)N=3)=CC=2)N1.O=S(C1C=CC=CC=1)(O)=O
|
| InChi Key |
UXDRJPYSTZHIOE-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C22H23FN6O5.C6H6O3S/c1-22(2)20(30)28-19-13(34-22)6-7-16(27-19)26-18-12(23)10-24-21(29-18)25-11-8-14(31-3)17(33-5)15(9-11)32-4;7-10(8,9)6-4-2-1-3-5-6/h6-10H,1-5H3,(H3,24,25,26,27,28,29,30);1-5H,(H,7,8,9)
|
| Chemical Name |
benzenesulfonic acid;6-[[5-fluoro-2-(3,4,5-trimethoxyanilino)pyrimidin-4-yl]amino]-2,2-dimethyl-4H-pyrido[3,2-b][1,4]oxazin-3-one
|
| Synonyms |
R406 benzenesulfonate; R-406 benzenesulfonate; R-406 besylate; R406; R-406; R 406; R 406 besylate; R406 Benzenesulfonate; Tamatinib besylate; R406 besylate; 6-((5-fluoro-2-((3,4,5-trimethoxyphenyl)amino)pyrimidin-4-yl)amino)-2,2-dimethyl-2H-pyrido[3,2-b][1,4]oxazin-3(4H)-one benzenesulfonate; 6-[[5-fluoro-2-[(3,4,5-trimethoxyphenyl)amino]-4-pyrimidinyl]amino]-2,2-dimethyl-2H-pyrido[3,2-b]-1,4-oxazin-3(4H)-one benzenesulfonate; 841290-81-1 (besylate); R406 besylate; R406 benzenesulfonate; Tamatinib
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (3.98 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (3.98 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: 30% PEG400+0.5% Tween80+5% Propylene glycol: 30mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5908 mL | 7.9538 mL | 15.9076 mL | |
| 5 mM | 0.3182 mL | 1.5908 mL | 3.1815 mL | |
| 10 mM | 0.1591 mL | 0.7954 mL | 1.5908 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT00798096 | Completed Has Results | Drug: Fostamatinib Disodium | T Cell Lymphoma | Rigel Pharmaceuticals | March 2009 | Phase 2 |
| NCT00923481 | Completed Has Results | Drug: Fostamatinib disodium | Head and Neck Neoplasms Pheochromocytoma |
National Cancer Institute (NCI) | April 2009 | Phase 2 |
| NCT02077192 | Completed Has Results | Drug: Fostamatinib Disodium | Immune Thrombocytopenic Purpura | Rigel Pharmaceuticals | October 2014 | Phase 3 |
| NCT00706342 | Completed Has Results | Drug: Fostamatinib Disodium / R935788 | Purpura, Thrombocytopenic, Idiopathic | Rigel Pharmaceuticals | January 2007 | Phase 2 |
|
 |
The Syk inhibitor R406 induces CLL cell apoptosis and abrogates BCR-derived survival signals. Blood. 2009 Jul 30; 114(5): 1029–1037. |