
| Size | Price | Stock | Qty |
|---|---|---|---|
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 2g |
|
||
| 5g |
|
||
| 10g | |||
| Other Sizes |
Purity: ≥98%
Sunitinib (formerly also known as SU11248; trade name: Sutent) is a potent, orally bioavailable and multi-targeted RTK (receptor tyrosine kinase) inhibitor with potential antitumor activity. In cell-free assays, it inhibits c-Kit in addition to VEGFR2 (Flk-1) and PDGFRβ, with IC50s of 80 nM and 2 nM, respectively. In 2006, the US FDA approved sunitinib for the treatment of gastrointestinal stromal tumors that were resistant to imatinib and renal cell carcinoma. Sunitinib inhibits angiogenesis and cell proliferation by blocking the tyrosine kinase activities of platelet-derived growth factor receptor b (PDGFRb), c-kit, and vascular endothelial growth factor receptor 2 (VEGFR2).
| Targets |
PDGFRβ (IC50 = 2 nM); VEGFR2 (IC50 = 80 nM); FLT3; c-Kit
1. Sunitinib free base (SU-11248) is a multi-targeted tyrosine kinase inhibitor with the following IC50 values: VEGFR1 (Flt-1): 2–10 nM, VEGFR2 (KDR): 1–8 nM, VEGFR3 (Flt-4): 2–8 nM, PDGFRα: 3–36 nM, PDGFRβ: 1–5 nM, c-Kit: 1–10 nM, FLT3: 3–30 nM, CSF-1R: 20–100 nM [1] 2. It inhibits RET proto-oncogene with an IC50 of 15–25 nM, while showing no significant inhibition (IC50 > 1 μM) against EGFR, HER2, and Src kinases [3] 3. For mutant FLT3 (FLT3-ITD), Sunitinib free base exhibits an IC50 of 5–12 nM, comparable to its activity against wild-type FLT3 [5] |
|---|---|
| ln Vitro |
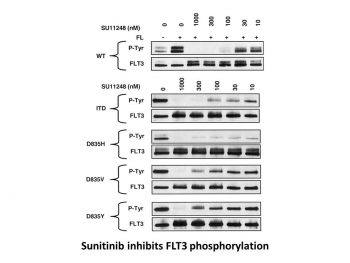
Sunitinib inhibits FLT-3 and Kit with considerable potency.[1] With a Ki of 9 nM and 8 nM, respectively, sunitinib is a strong ATP-competitive inhibitor of VEGFR2 (Flk1) and PDGFRβ. It provides >10-fold greater selectivity for VEGFR2 and PDGFR than FGFR-1, EGFR, Cdk2, Met, IGFR-1, Abl, and src. With IC50 values of 10 nM and 10 nM, respectively, sunitinib inhibits the phosphorylation of VEGFR2 in response to VEGF and PDGFRβ in response to PDGF in serum-starved NIH-3T3 cells expressing VEGFR2 or PDGFRβ. Sunitinib has an IC50 of 40 nM for VEGF-induced proliferation of serum-starved HUVECs and an IC50 of 39 nM and 69 nM for PDGF-induced proliferation of NIH-3T3 cells overexpressing PDGFRβ or PDGFRβ, respectively.[2] With an IC50 of 250 nM, 50 nM, and 30 nM, respectively, sunitinib inhibits the phosphorylation of wild-type FLT3, FLT3-ITD, and FLT3-Asp835. With IC50 values of 8 nM and 14 nM, respectively, sunitinib suppresses the growth of MV4;11 and OC1-AML5 cells and, in a dose-dependent fashion, triggers apoptosis.[3]
1. Human tumor cell lines: Sunitinib free base inhibits proliferation of A549 (lung cancer) with an IC50 of 2.5 μM, HT-29 (colon cancer) with an IC50 of 3.8 μM, and SK-OV-3 (ovarian cancer) with an IC50 of 4.2 μM after 72-hour treatment [2] 2. HUVECs (human umbilical vein endothelial cells): Sunitinib free base (0.1–10 μM) dose-dependently inhibits VEGF-induced cell migration and tube formation. At 1 μM, migration is reduced by ~65% and tube formation by ~70% compared to the VEGF-stimulated control [1] 3. MV4-11 cells (FLT3-ITD-positive acute myeloid leukemia, AML): Sunitinib free base (10–100 nM) induces apoptosis. After 48-hour treatment with 50 nM, the apoptotic rate (Annexin V-positive cells) increases from ~5% (control) to ~45% [5] 4. GIST882 cells (c-Kit-mutant gastrointestinal stromal tumor): Western blot shows Sunitinib free base (1 μM) reduces phosphorylation of c-Kit (Tyr719) by ~80%, and downstream p-AKT (Ser473) and p-ERK1/2 by ~75% and ~70% respectively, compared to the untreated group [4] 5. Caki-1 renal cell carcinoma (RCC) cells: Sunitinib free base (0.5–5 μM) suppresses hypoxia-induced HIF-1α protein expression. At 2 μM, HIF-1α levels are reduced by ~60% after 24-hour hypoxia exposure [6] |
| ln Vivo |
Sunitinib (20–80 mg/kg/day) exhibits broad and potent dose-dependent anti-tumor activity against a variety of tumor xenograft models, including HT-29, A431, Colo205, H-460, SF763T, C6, A375, or MDA-MB-435. This is consistent with the significant and selective inhibition of VEGFR2 or PDGFR phosphorylation and signaling in vivo. Six out of eight mice receiving 80 mg/kg/day of sunitinib for 21 days experience complete tumor regression, and 110 days after the end of the treatment, there is no regrowth of the tumor.Tumors that do not completely regress after the first round of treatment can still be successfully treated with sunitinib in a second round. Tumor MVD significantly decreases with sunitinib treatment, with SF763T glioma tumors reduced by approximately 40%. Tumor size remains unchanged, but luciferase-expressing PC-3M xenografts treated with SU11248 completely inhibits further tumor growth.[2] Treatment with sunitinib (20 mg/kg/day) increases survival in the FLT3-ITD bone marrow engraftment model and significantly suppresses the growth subcutaneous MV4;11 (FLT3-ITD) xenografts.[3]
1. Nude mouse A549 lung cancer xenograft model: Oral administration of Sunitinib free base (20 mg/kg, once daily for 21 days) results in a tumor growth inhibition (TGI) rate of ~65%. Tumor volume in the treated group is ~35% of the vehicle control [2] 2. SCID mouse MV4-11 AML intravenous xenograft model: Sunitinib free base (40 mg/kg, oral gavage, once daily for 14 days) prolongs survival. Median survival time increases from 21 days (control) to 38 days, and 3 out of 8 mice survive beyond 60 days [5] 3. Nude mouse GIST882 gastrointestinal stromal tumor model: Sunitinib free base (30 mg/kg, oral, once daily for 28 days) reduces tumor weight by ~70% and intratumoral microvessel density (CD31-positive vessels) by ~60% compared to the vehicle group [4] 4. Rat orthotopic Caki-1 RCC model: Sunitinib free base (50 mg/kg, oral, once daily for 35 days) inhibits primary tumor growth (TGI ~75%) and reduces lung metastasis (number of metastatic nodules decreases by ~80%) [6] 5. Mouse laser-induced choroidal neovascularization (CNV) model: Sunitinib free base (15 mg/kg, oral, once daily for 10 days) reduces CNV area by ~55% compared to the vehicle control [1] |
| Enzyme Assay |
Sunitinib's IC50 values against PDGFRβ and VEGFR2 (Flk-1) are ascertained by employing glutathione S-transferasefusion proteins that encompass the entire RTK cytoplasmic domain. In order to measure the trans-phosphorylation activity of VEGFR2 (Flk-1) and PDGFRβ, biochemical tyrosine kinase assays are carried out in 96-well microtiter plates that have been precoated (20 μg/well in PBS) and incubated with the peptide substrate poly-Glu,Tyr (4:1) for an entire night at 4 °C. Adding 1-5% (w/v) BSA to PBS blocks excess protein binding sites. The cells of insects infected with baculovirus produce purified GST-fusion proteins. The microtiter wells are then filled with GST-VEGFR2 and GST-PDGFRβ in a 2 × concentration kinase dilution buffer that contains 40 μM NaVO4, 50 mM NaCl, 100 mM HEPES, and 0.02% (w/v) BSA. 50 ng/mL is the final enzyme concentration for GST-VEGFR2 or GST-PDGFRβ. To create a range of inhibitor concentrations suitable for every enzyme, 25 μL of diluted Sunitinib is then added to each reaction well. A solution of MnCl2 is mixed with varying concentrations of ATP to start the kinase reaction. The final concentration of MnCl2 is 10 mM, and the final ATP concentrations span the Km for the enzyme. After allowing the plates to sit at room temperature for five to fifteen minutes, the reaction is halted by adding EDTA. After that, TBST is used to wash the plates three times. After adding rabbit polyclonal antiphosphotyrosine antisera at a 1:10,000 dilution to the wells in TBST containing 0.025% (w/v) nonfat dry milk, 0.5% (w/v) BSA, and 100 μM NaVO4, the wells are incubated at 37 °C for one hour. After three TBST washes, the plates are inoculated with goat anti-rabbit antisera conjugated with horseradish peroxidase (1:10,000 dilution in TBST). The plates are cleaned three times with TBST after an hour of incubation at 37 °C. Once 2,2′-azino-di-[3-ethylbenzthiazoline sulfonate] has been added as substrate, the amount of phosphotyrosine in each well is quantified.
1. Recombinant VEGFR2 (KDR) kinase activity assay: The assay is performed in a buffer containing 50 mM Tris-HCl (pH 7.5), 10 mM MgCl2, 1 mM DTT, 25 μM ATP, and 1 μg/well Poly(Glu,Tyr)4:1 substrate. Different concentrations of Sunitinib free base (0.1 nM–1 μM) are pre-incubated with recombinant VEGFR2 (5 ng/well) for 10 minutes at 30°C. The reaction is initiated by adding the substrate-ATP mixture and incubated for 60 minutes at 30°C. Phosphorylated substrate is detected with a phosphotyrosine-specific antibody and colorimetric readout at 450 nm. IC50 is calculated via nonlinear regression of inhibition curves [1] 2. FLT3-ITD kinase activity assay: Recombinant FLT3-ITD (10 ng/well) is incubated with Sunitinib free base (0.5 nM–50 nM) in a buffer with 20 mM HEPES (pH 7.4), 5 mM MnCl2, 1 mM DTT, 10 μM ATP, and 0.5 μg/well peptide substrate (sequence: EAIYAAPFAKKK). The reaction is carried out at 37°C for 45 minutes, stopped with 3% phosphoric acid, and transferred to a P81 phosphocellulose plate. Radioactivity (from [γ-32P]ATP) is measured with a scintillation counter to determine IC50 [5] 3. PDGFRβ kinase assay: Recombinant PDGFRβ (8 ng/well) is mixed with Sunitinib free base (0.2 nM–20 nM) in a buffer containing 50 mM Tris-HCl (pH 7.6), 10 mM MgSO4, 1 mM EGTA, 20 μM ATP, and 1 μg/well myelin basic protein (MBP) substrate. After 30 minutes at 30°C, the reaction is stopped with SDS sample buffer. Phosphorylated MBP is detected by Western blot with an anti-phospho-MBP antibody, and band intensity is quantified to calculate IC50 [1] |
| Cell Assay |
The cells are starved for an entire night in a medium containing 0.1% FBS before FL (50 ng/mL; FLT3-WT cells only) and sunitinib are added. After 48 hours of culture, proliferation is assessed using trypan blue cell viability assays or the Alamar Blue assay. Apoptosis is quantified using Western blotting 24 hours after Sunitinib addition in order to identify caspase-3 levels or poly (ADP-ribose) polymerase (PARP) cleavage.
1. Tumor cell proliferation assay (MTT method): Human tumor cells (A549, HT-29, SK-OV-3) are seeded in 96-well plates at 3×10³ cells/well and cultured overnight. Sunitinib free base (0.1 μM–10 μM) is added, and cells are incubated for 72 hours at 37°C. MTT reagent (5 mg/mL, 10 μL/well) is added for 4 hours, then formazan crystals are dissolved in DMSO (100 μL/well). Absorbance is measured at 570 nm, cell viability is expressed as a percentage of the control, and IC50 is derived from dose-response curves [2] 2. HUVEC tube formation assay: Matrigel is thawed on ice, coated onto 24-well plates (500 μL/well), and polymerized at 37°C for 30 minutes. HUVECs (2×10⁴ cells/well) are suspended in medium containing Sunitinib free base (0.1–10 μM) and VEGF (50 ng/mL), then seeded onto Matrigel. After 6 hours, tube-like structures are photographed, and total tube length per well is quantified with image analysis software. Inhibition rate is calculated relative to the VEGF control [1] 3. MV4-11 cell apoptosis assay (Annexin V-FITC/PI staining): MV4-11 cells (1×10⁵ cells/mL) are treated with Sunitinib free base (10–100 nM) for 48 hours. Cells are harvested, washed with PBS, and stained with Annexin V-FITC and PI per kit protocol. Apoptotic cells are analyzed by flow cytometry, and the apoptotic rate is calculated [5] 4. GIST882 cell Western blot for c-Kit signaling: GIST882 cells (5×10⁵ cells/well) are seeded in 6-well plates and cultured overnight. Sunitinib free base (1 μM) is added for 2 hours, then cells are lysed in RIPA buffer with protease/phosphatase inhibitors. Protein concentration is measured by BCA assay. Equal amounts of protein (40 μg) are separated by 10% SDS-PAGE, transferred to PVDF membranes, and probed with antibodies against p-c-Kit (Tyr719), c-Kit, p-AKT (Ser473), AKT, p-ERK1/2, and ERK1/2. HRP-conjugated secondary antibodies and ECL reagent are used for detection, and band intensity is quantified with ImageJ [4] |
| Animal Protocol |
Mice: The mice used are female nu/nu (8–12 weeks old, 25 grams). In short, on day 0, mice receive a subcutaneous injection of 3-5×106 tumor cells into the hind flank region. After tumors reach the indicated average size, mice bearing tumors are treated daily orally with carboxymethyl cellulose suspension or as a citrate buffered (pH 3.5) solution containing sunitinib. Tumor growth is assessed using tumor volume measurements taken twice a week. When tumors in animals receiving vehicle treatment reach an average size of 1000 mm3 or are determined to negatively impact the animals' quality of life, studies are usually stopped.
Rats: The Wistar rats are adult males weighing between 325 and 349 g. In two drug studies, the efficacy of the time-lapse imaging method in assessing the anti-angiogenic effects of a particular drug treatment is verified. First, mesenteric windows are taken from adult male Wistar rats, and the tissues are cultured for three days in two different experimental groups: 1) 10% serum (n = 8 tissues from 4 rats), and 2) 10% serum + Sunitinib (5 μM; n=8 tissues from 4 rats). 1. Nude mouse A549 xenograft model: Female athymic nude mice (6–8 weeks old) are subcutaneously injected with 5×10⁶ A549 cells (suspended in 100 μL PBS/Matrigel 1:1) into the right flank. When tumors reach ~100 mm³, mice are randomized into 2 groups (n=6/group): vehicle control (0.5% methylcellulose + 0.1% Tween 80) and Sunitinib free base (20 mg/kg). The drug is administered by oral gavage once daily for 21 days. Tumor volume (V = length×width²/2) is measured every 3 days, and body weight is monitored [2] 2. SCID mouse MV4-11 AML model: Male SCID mice (7–9 weeks old) are intravenously injected with 1×10⁷ MV4-11 cells. Three days later, mice are divided into 2 groups (n=8/group): vehicle (0.5% methylcellulose) and Sunitinib free base (40 mg/kg, oral gavage once daily for 14 days). Survival is recorded daily, and peripheral blood is collected weekly to detect human CD45-positive cells (disease burden) [5] 3. Rat orthotopic Caki-1 RCC model: Male Wistar rats (200–220 g) are anesthetized, and 1×10⁶ Caki-1 cells are injected into the left kidney capsule. Two weeks after implantation, rats are randomized into 2 groups (n=5/group): vehicle (0.2% Tween 80 in saline) and Sunitinib free base (50 mg/kg, oral gavage once daily for 35 days). Rats are euthanized; primary tumors are excised and weighed, and lung tissues are fixed to count metastatic nodules [6] 4. Mouse laser-induced CNV model: Female C57BL/6 mice (8–10 weeks old) are anesthetized, and laser photocoagulation is applied to the choroid to induce CNV. One day later, mice are divided into 2 groups (n=6/group): vehicle (saline) and Sunitinib free base (15 mg/kg, oral gavage once daily for 10 days). Mice are euthanized, choroidal flat mounts are prepared and stained with isolectin B4, and CNV area is measured with confocal microscopy [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Following oral administration, sunitinib typically reaches peak plasma concentration (Cmax) between 6 and 12 hours (Tmax). Food has no effect on the bioavailability of sunitinib. Sunitinib can be taken with or without food. The pharmacokinetic profile of sunitinib was similar in healthy volunteers and in the tested solid tumor populations, including patients with gastrointestinal stromal tumors (GIST) and renal cell carcinoma (RCC). Sunitinib is primarily metabolized by the cytochrome P450 enzyme CYP3A4 to its major active metabolite, which is further metabolized by CYP3A4. Sunitinib is primarily excreted in feces. In the [14C]sunitinib human weight balance study, 61% of the dose was excreted in feces, and 16% was excreted by the kidneys. 2230 L (apparent volume of distribution, Vd/F) 34 - 62 L/h [Total Oral Clearance] Peak plasma concentrations of sunitinib typically occur within 6-12 hours after oral administration. Food has no effect on the bioavailability of sunitinib. Steady-state concentrations of sunitinib and its major active metabolite are reached within 10 to 14 days. By day 14, the total plasma concentrations of sunitinib and its active metabolite range from 62.9 to 101 ng/mL. No significant changes in the pharmacokinetics of sunitinib or its major active metabolite were observed with repeated daily dosing or repeated dosing cycles in the tested dosing regimens. Sunitinib and its major active metabolite are 95% and 90% bound to human plasma proteins in vitro, respectively. The apparent volume of distribution (Vd/F) of sunitinib is 2230 L. Within the dose range of 25-100 mg, the area under the plasma concentration-time curve (AUC) and Cmax increase proportionally with dose. For more complete data on absorption, distribution, and excretion of sunitinib (11 items in total), please visit the HSDB record page. Metabolism/Metabolites Sunitinib is primarily metabolized by the cytochrome P450 enzyme CYP3A4 to its major active metabolite, which is further metabolized by other enzymes. CYP3A4. Sunitinib is primarily metabolized by the cytochrome P-450 (CYP) isoenzyme 3A4 to several metabolites. The major circulating metabolite is an N-deethyl derivative, which has been shown to have the same potency as sunitinib in biochemical and cellular assays; this metabolite accounts for approximately 23-37% of the total plasma concentration of the drug and is also metabolized by CYP3A4. Sunitinib and its major active metabolite were the main drug-related compounds identified in plasma, urine, and feces, accounting for 91.5%, 86.4%, and 73.8% of the radioactivity in the mixed samples, respectively. Biological Half-Life Following a single oral administration in healthy volunteers, the terminal half-lives of sunitinib and its major active metabolite were approximately 40–60 hours and 80–110 hours, respectively. Following a single oral administration in healthy volunteers, the terminal half-lives of sunitinib or its major active metabolite were approximately 40–60 hours or 80–110 hours, respectively. 1. Rats: After oral administration of sunitinib free base (20 mg/kg), the oral bioavailability (F) was 48%, the peak plasma concentration (Cmax) was 1.2 μg/mL, the time to peak concentration (Tmax) was 1.5 hours, and the terminal half-life (t1/2) was 6.8 hours. After intravenous injection (5 mg/kg), t1/2 was 5.2 hours [1] 2. Dogs: After oral administration of sunitinib free base (10 mg/kg), F=36%, Cmax=0.8 μg/mL, Tmax=2 hours, t1/2=9.5 hours. Plasma protein binding rate>95% (measured by ultrafiltration)[1] 3. Mice: After a single oral administration of sunitinib free base (30 mg/kg), the highest drug concentrations were found in the liver (12 μg/g) and kidneys (8 μg/g) 2 hours after administration; the brain tissue concentration was <0.5 μg/g, indicating poor blood-brain barrier penetration [4] 4. Metabolism: In human liver microsomes, sunitinib free base is metabolized to N-deethylsunitinib (the main active metabolite), with a metabolic clearance rate of 1.2 mL/min/mg protein [1] |
| Toxicity/Toxicokinetics |
Hepatotoxicity
Elevated serum transaminase levels were common in large clinical trials of sunitinib, occurring in 39% of the sunitinib group and 23% of the placebo group. Only 2% to 3% of patients in the sunitinib group (and 1% in the control group) experienced transaminase levels exceeding 5 times the upper limit of normal (ULN). These abnormalities are usually asymptomatic. If enzyme levels are significantly elevated (ALT or AST persistently exceeding 5 times the ULN, or bilirubin exceeding 3 times the ULN), dose adjustment or temporary discontinuation of treatment is recommended, followed by restarting treatment at a lower dose. Sunitinib treatment has also been associated with a higher rate of elevated serum bilirubin, usually mild to moderate, and unrelated to ALT or AST elevations. These changes may be related to the interaction with hepatic UDP-glucuronyl transferase, which is also responsible for bilirubin excretion. More importantly, several cases of clinically significant liver injury have been reported attributable to sunitinib treatment. Onset typically occurs after several treatment cycles. The pattern of elevated serum enzymes is typically hepatocellular, with clinical presentations resembling acute hepatic necrosis. In some cases, the injury may be due to hypotension, shock, or ischemia, rather than direct hepatotoxicity (Case 1). Regardless, the injury can be severe, with several reports of acute liver failure and death. Immune allergic reactions (rash, fever, and eosinophilia) are uncommon. Finally, there have been reports of sunitinib causing hyperammonemia and encephalopathy in cancer patients receiving standard or even low-dose oral therapy (Case 2). Onset is 1 to 3 weeks, presenting with confusion and irritability, mild elevations in serum enzymes and bilirubin, and significantly elevated serum ammonia (4-10 times the upper limit of normal). Recovery is rapid upon discontinuation of sunitinib, but relapse may occur upon re-administration. Notably, cross-reactivity with other tyrosine kinase inhibitors appears to be minimal. Probability score: B (Highly probable cause of clinically evident liver injury, including hyperammonemia syndrome). Use during pregnancy and lactation ◉ Overview of use during lactation There is currently no information on the clinical use of sunitinib during lactation. Because sunitinib and its metabolites bind to plasma proteins at a rate exceeding 90%, the levels in breast milk may be very low. However, one of the metabolites has a half-life of up to 110 hours and may accumulate in the infant. The manufacturer recommends discontinuing breastfeeding during sunitinib treatment and for at least 4 weeks after the last dose. ◉ Effects on breastfed infants As of the revision date, no relevant published information was found. ◉ Effects on lactation and breast milk As of the revision date, no relevant published information was found. Protein binding The in vitro binding rates of sunitinib and its major metabolites to human plasma proteins are 95% and 90%, respectively. Interaction …A 57-year-old woman, after failing imatinib treatment, began sunitinib treatment for recurrent metastatic gastrointestinal stromal tumors. After 8 cycles of sunitinib (50 mg/day for 4 weeks, followed by a 2-week break), her condition stabilized and her liver function returned to normal. She continued to take acetaminophen (approximately 4.5 g/week) and her usual asthma medication. In the 8th cycle, she took levothyroxine sodium (50-150 mcg/day) orally for approximately 30 days to control hypothyroidism, after which she began the 9th cycle of sunitinib. On day 4 of the 9th cycle, she was hospitalized due to progressively elevated circulating liver enzyme levels. Despite discontinuing sunitinib and initiating intensive supportive care, she died 4 days after admission. Autopsy revealed severe central lobular necrosis in her liver with moderate to severe steatosis, and minimal tumor invasion of the parenchyma. Viral staining results were negative. Liver failure has been reported in rare cases in patients taking sunitinib. Autopsy results ruled out tumor progression and viral infection as causes of death; the patient likely died from the combined effects of sunitinib, acetaminophen, and levothyroxine. Although sunitinib is only considered a possible hepatotoxic substance (Roussel Uclaf causality assessment method), and may even have a hepatoprotective effect against long-term acetaminophen (a possible hepatotoxic substance) over 48 weeks by inducing localized hypothyroidism within the liver, it is presumed that correcting presupposed hepatic hypothyroidism with oral levothyroxine (a possible hepatotoxic substance) and restarting sunitinib treatment may have induced liver necrosis. …Potential CYP3A4 inhibitors, such as ketoconazole, may increase plasma concentrations of sunitinib. It is recommended to choose an alternative concomitant medication with weak or no enzyme inhibition. In healthy volunteers, a single dose of sunitinib, when combined with the potent CYP3A4 inhibitor ketoconazole, resulted in a 49% and 51% increase in the combined levels of sunitinib and its major active metabolites (Cmax and AUC0-∞), respectively. Concomitant use of sunitinib with potent CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, clarithromycin, atazanavir, indinavir, nefazodone, nelfinavir, ritonavir, saquinavir, telithromycin, voriconazole) may increase sunitinib plasma concentrations. Grapefruit may also increase sunitinib plasma concentrations. CYP3A4 inducers (e.g., rifampin) may decrease sunitinib plasma concentrations. It is recommended to choose an alternative drug with weak or no enzyme-inducing activity. In healthy volunteers, a single dose of sunitinib, when concomitantly used with the potent CYP3A4 inducer rifampin, resulted in a 23% and 46% reduction in the combined values of sunitinib and its major active metabolites (Cmax and AUC0-∞), respectively. Concomitant use of sunitinib with CYP3A4 family inducers (e.g., dexamethasone, phenytoin sodium, carbamazepine, rifampin, rifabutin, rifapentine, phenobarbital) may decrease sunitinib plasma concentrations. St. John's wort may cause unpredictable decreases in plasma sunitinib concentrations and should therefore be avoided during sunitinib treatment. 1. Acute toxicity in mice: A single oral dose of sunitinib free base (up to 200 mg/kg) did not result in death within 7 days, but mice in the 150–200 mg/kg group experienced transient weight loss (5–8% reduction within 48 hours) and decreased kinetic activity, which resolved within 7 days [4] 2. Subchronic toxicity in rats (oral administration over 28 days): - 25 mg/kg group: No significant changes in body weight, organ weight, or hematological parameters [6] - 50 mg/kg group: Mild weight loss (3–5%), slight increase in liver weight (10–12%), and a 15% decrease in platelet count; no histopathological changes were observed in the liver/kidneys [6] - 100 mg/kg group: Significant weight loss (8–10%), elevated serum ALT (2-fold) and AST (1.8-fold), and severe thrombocytopenia (40% decrease); 2 out of 5 rats showed significant weight loss. Only mild liver necrosis was observed [6] 3. Nude mouse xenotransplantation studies (treatment 21-35 days): Sunitinib free base (20-50 mg/kg) did not cause more than 10% weight loss or significant organ toxicity (as assessed by histopathological evaluation of liver, kidney and spleen) [2][4] |
| References | |
| Additional Infomation |
Therapeutic Uses
Angiogenesis Inhibitor; Antitumor Drug Sunitinib Malate is indicated for the treatment of gastrointestinal stromal tumors that have progressed or are intolerant to imatinib mesylate. /US Product Label Includes/ Sunitinib Malate is indicated for the treatment of advanced renal cell carcinoma. /US Product Label Includes/ Sunitinib Malate is indicated for the treatment of locally advanced or metastatic unresectable progressive, well-differentiated pancreatic neuroendocrine tumors. /US Product Label Includes/ Drug Warnings /Black Box Warning/ Hepatotoxicity—Hepatotoxicity has been observed in clinical trials and postmarketing experience. This hepatotoxicity can be serious, and deaths have been reported. Sunitinib is associated with hepatotoxicity and may lead to liver failure or death. Liver failure has been observed in clinical trials (7/2281 [0.3%]) and postmarketing experience. Signs of liver failure include jaundice, elevated transaminases and/or hyperbilirubinemia, accompanied by encephalopathy, coagulation disorders and/or renal failure. Liver function parameters (ALT, AST, bilirubin) should be monitored before starting treatment, at each treatment cycle, and as clinically necessary. Sunitinib should be discontinued for Grade 3 or 4 drug-related adverse liver events; if the adverse event does not resolve, treatment should be discontinued. Sunitinib should not be restarted if the patient subsequently develops severe changes in liver function parameters or other signs and symptoms of liver failure. The safety of this product has not been established in patients with ALT or AST > 2.5 times the upper limit of normal (ULN) or ALT or AST > 5.0 times the upper limit of normal (ULN) due to liver metastasis. Post-marketing experience suggests that this product can cause cardiovascular events, including heart failure, myocardial disease, and cardiomyopathy, some of which are fatal. In randomized trials, among patients receiving sunitinib for metastatic renal cell carcinoma, 21% had a left ventricular ejection fraction (LVEF) below the lower limit of normal, and 4% had a decreased LVEF (below 50% or more than 20% from baseline). In patients treated with sunitinib, 1% reported left ventricular dysfunction and less than 1% reported congestive heart failure. For more complete data on sunitinib warnings (33 in total), please visit the HSDB record page. Pharmacodynamics Sunitinib is an oral small-molecule multi-target receptor tyrosine kinase (RTK) inhibitor approved by the FDA on January 26, 2006. 1. The free base of sunitinib exerts its antitumor effect through two mechanisms: by blocking the VEGFR/PDGFR signaling pathway to inhibit angiogenesis, and by directly inhibiting the proliferation of tumor cells expressing c-Kit/FLT3 [1][3]. 2. Its major active metabolite (N-desethylsunitinib) has similar kinase inhibitory activity to the parent drug (IC50 for VEGFR2: 5–12 nM). It accounts for approximately 30% of the total activity in vivo [1] 3. In preclinical models, sunitinib free base showed synergistic antitumor activity when used in combination with docetaxel (lung cancer) and gemcitabine (pancreatic cancer); the combination therapy reduced tumor volume by an additional 20-30% compared with monotherapy [7] 4. Sunitinib free base was effective against tumor models resistant to other anti-angiogenic drugs (e.g., A549 xenograft tumors resistant to bevacizumab), with a tumor growth inhibition rate (TGI) of approximately 60%, compared to approximately 25% for bevacizumab [2] 5. Sunitinib has higher selectivity for tumor-associated kinases (VEGFR, PDGFR) than for housekeeping kinases (e.g., EGFR, Src), thereby reducing off-target toxicity and giving it good preclinical safety [1] |
| Molecular Formula |
C22H27FN4O2
|
|---|---|
| Molecular Weight |
398.47
|
| Exact Mass |
398.211
|
| Elemental Analysis |
C, 66.31; H, 6.83; F, 4.77; N, 14.06; O, 8.03
|
| CAS # |
557795-19-4
|
| Related CAS # |
Sunitinib Malate;341031-54-7;Sunitinib-d10;1126721-82-1;Sunitinib-d4;1126721-79-6; 342641-94-5; 1275588-72-1 (mesylate) ; 1126641-10-8; 1327155-72-5 (HCl); 1221149-36-5 (acetate); 1332306-95-2 (oxalate)
|
| PubChem CID |
5329102
|
| Appearance |
Yellow solid powder
|
| Density |
1.2±0.1 g/cm3
|
| Boiling Point |
572.1±50.0 °C at 760 mmHg
|
| Melting Point |
189-191ºC
|
| Flash Point |
299.8±30.1 °C
|
| Vapour Pressure |
0.0±1.6 mmHg at 25°C
|
| Index of Refraction |
1.611
|
| LogP |
3.15
|
| Hydrogen Bond Donor Count |
3
|
| Hydrogen Bond Acceptor Count |
4
|
| Rotatable Bond Count |
7
|
| Heavy Atom Count |
29
|
| Complexity |
636
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
FC1C=C2C(NC(=O)/C/2=C\C2NC(C)=C(C(NCCN(CC)CC)=O)C=2C)=CC=1
|
| InChi Key |
WINHZLLDWRZWRT-ATVHPVEESA-N
|
| InChi Code |
InChI=1S/C22H27FN4O2/c1-5-27(6-2)10-9-24-22(29)20-13(3)19(25-14(20)4)12-17-16-11-15(23)7-8-18(16)26-21(17)28/h7-8,11-12,25H,5-6,9-10H2,1-4H3,(H,24,29)(H,26,28)/b17-12-
|
| Chemical Name |
N-[2-(diethylamino)ethyl]-5-[(Z)-(5-fluoro-2-oxo-1H-indol-3-ylidene)methyl]-2,4-dimethyl-1H-pyrrole-3-carboxamide
|
| Synonyms |
SU11248; SU 11248; sunitinibum; Su-011248; Sunitinib Base; SU011248; SU-11248; sunitinib; trade name: Sutent.
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.11 mg/mL (2.79 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 11.1 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.11 mg/mL (2.79 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 11.1 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. View More
Solubility in Formulation 3: 5% DMSO+corn oil: 7mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5096 mL | 12.5480 mL | 25.0960 mL | |
| 5 mM | 0.5019 mL | 2.5096 mL | 5.0192 mL | |
| 10 mM | 0.2510 mL | 1.2548 mL | 2.5096 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
TAPUR: Testing the Use of Food and Drug Administration (FDA) Approved Drugs That Target a Specific Abnormality in a Tumor Gene in People With Advanced Stage Cancer
CTID: NCT02693535
Phase: Phase 2 Status: Recruiting
Date: 2024-11-12
|
 |