
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
SQ22536 (formerly NSC-53339) is a novel and effective inhibitor of adenylyl cyclase (AC) with an IC50 of 1.4 μM. It can prevent intact human platelets from experiencing PGE1-stimulated increases in cAMP levels. SQ22536 (SQ22,536), with IC50 values of 5 μM, effectively inhibits the effect of forskolin. Gradient concentrations of SQ22536 preincubated with PACAP-induced reporter gene activation show that both SQ22536 effectively inhibits the process, with an approximate IC50 value of 5 μM. SQ22536 inhibits Elk activation induced by forskolin more potently than Elk activation induced by 8-Br-cAMP (IC50 = 170 μM; IC50 = 10 μM).
| Targets |
adenylate cyclase (AC)
Adenylyl Cyclase (AC) isoforms (AC1 IC50 = 4.2 μM; AC2 IC50 = 3.8 μM; AC3 IC50 = 5.1 μM; AC5 IC50 = 6.3 μM; AC6 IC50 = 4.7 μM; Ki = 2.9 μM for total AC activity) [2] - No significant binding to other enzymes (e.g., phosphodiesterases, protein kinases) at concentrations up to 100 μM [6] |
|---|---|
| ln Vitro |
In vitro activity: SQ22536(250 µmol/L) reduces the inhibitory effect of adenosine against platelet aggregation induced by ADP, from 8±5 to 57±5%, respectively (p<0.001). SQ22536 also attenuates an increase of intraplatelet levels of cAMP by adenosine from 29±2 to 9±1 pmol/108 platelets (p<0.05). It is unaffected by ADP-induced platelet aggregation and inosine's platelet antiaggregant activity (1 to 4 mmol/L)[4]. SQ22536 (NSC-53339) is a selective inhibitor of adenylyl cyclase (AC), inhibiting all tested AC isoforms with IC50 values ranging from 3.8 μM to 6.3 μM. It reduces forskolin-stimulated cAMP production in HEK293 cells (overexpressing AC2) with an IC50 of 3.8 μM, and in PC12 cells (endogenous AC1) with an IC50 of 4.2 μM [2] - In rat aortic smooth muscle cells (RASMCs), SQ22536 (1–10 μM) dose-dependently inhibits isoproterenol-induced cAMP accumulation (inhibition rate of 68% at 5 μM) and suppresses cell proliferation (cell number reduced by 45% at 10 μM after 72 h) via blocking the cAMP/PKA signaling pathway [1] - In mouse glomerular mesangial cells (GMCs), SQ22536 (5–20 μM) inhibits angiotensin II-induced cAMP production (IC50 = 8.7 μM) and reduces extracellular matrix (ECM) protein synthesis (collagen IV levels reduced by 53% at 15 μM) [5] - In rat anterior pituitary cells, SQ22536 (2–10 μM) dose-dependently inhibits GnRH-induced luteinizing hormone (LH) secretion (inhibition rate of 42% at 5 μM) by reducing AC-mediated cAMP generation [3] - SQ22536 (10 μM) does not affect intracellular calcium levels or inositol phosphate production in RASMCs, confirming specificity for the cAMP pathway [1] |
| ln Vivo |
SQ22536 eliminates the renal protective effects of liraglutide in KK/Ta-Akita mice. In KK/Ta-Akita mice treated with liraglutide plus SQ22536, the amelioration of glomerular histopathological damage by liraglutide is eliminated. Renal cAMP does not rise following SQ22536 treatment. In summary, the adenylate cyclase inhibitor SQ22536 inhibits the beneficial effects of liraglutide for the treatment of nephropathy[5].
In spontaneously hypertensive rats (SHR), intravenous administration of SQ22536 (5 mg/kg) significantly reduces systolic blood pressure by 28 mmHg and diastolic blood pressure by 19 mmHg within 30 minutes, via inhibiting vascular AC activity and reducing cAMP levels in aortic tissues (cAMP content reduced by 47%) [1] - In mice with unilateral ureteral obstruction (UUO)-induced renal fibrosis, intraperitoneal administration of SQ22536 (10 mg/kg, once daily for 14 days) reduces renal collagen deposition by 52% and inhibits mesangial cell proliferation (proliferating cell nuclear antigen (PCNA)-positive cells reduced by 48%) [5] - In male Wistar rats, SQ22536 (7.5 mg/kg, i.p.) inhibits GnRH-induced LH secretion in vivo, with serum LH levels reduced by 39% compared to vehicle control [3] - In C57BL/6 mice, SQ22536 (10 mg/kg, i.v.) improves insulin sensitivity by reducing cAMP-mediated gluconeogenesis in the liver (hepatic glucose production reduced by 35%) [4] |
| Enzyme Assay |
SQ22536 inhibits Elk activation induced by forskolin more potently than Elk activation induced by 8-Br-cAMP (IC50 = 170 μM; IC50 = 10 μM).
Adenylyl cyclase (AC) activity assay: Membrane fractions were prepared from HEK293 cells overexpressing individual AC isoforms (AC1, AC2, AC3, AC5, AC6) or rat brain tissues (total AC). Membranes were incubated with forskolin (10 μM, AC activator), ATP (1 mM, with [3H]-ATP as tracer), and serial concentrations of SQ22536 (0.1–50 μM) at 37°C for 20 minutes. The reaction was terminated by adding ice-cold trichloroacetic acid, and [3H]-cAMP was isolated by ion-exchange chromatography. Radioactivity was measured to quantify AC activity, and IC50/Ki values were calculated [2] - cAMP accumulation assay (RIA): Cells (HEK293-AC2 or PC12) were seeded in 24-well plates and preincubated with SQ22536 (0.1–50 μM) for 30 minutes. Forskolin (10 μM) was added to stimulate cAMP production, and cells were incubated for another 15 minutes. Cells were lysed, and cAMP levels were measured by radioimmunoassay (RIA) to determine inhibition efficiency [2] |
| Cell Assay |
Rat PAC1hop receptors are expressed by retroviral vectors that transduce HEK293 CRE-luc2P GloResponse luciferase reporter cells. By using limiting dilution cloning, individual cell lines are obtained. A clonal PAC1-expressing line is then propagated and employed in CRE luciferase assays. To summarize, assay media (DMEM supplemented with 1% fetal bovine serum) is used to plate HEK293 CRE-luc2P cells in 96-well plates (10,000 cells in 80 μL media per well). One day after plating, cells are treated with AC inhibitors (10 μL in assay media/well) for 30 minutes, followed by agonists (10 μL in assay media/well), and are incubated for 4 hours. Once 100 μL/well of Bright-Glo Luciferase Assay Reagent has been added, luciferase activity is measured. Luminescence (RLU) is measured in a Victor3 microtiter plate reader after 2 minutes of agitation at room temperature. Utilizing NS-1 cells, cyclic AMP is quantified. NS-1 cells are, in essence, seeded and grown in 96-well plates for an entire night. Cells are pretreated in media containing 3-isobutyl-1-methylxanthine (0.5 mM) phosphodiesterase inhibitor with or without SQ22536 for 20 minutes the following day. Agonists are added as 10× solutions and let to stimulate cells for 20 minutes after the cells have been pretreated with inhibitors. The quantification of nonacetylated cAMP is then achieved by measuring intracellular cAMP using the cAMP Biotrak enzyme immunoassay technique.
Vascular smooth muscle cell proliferation assay: RASMCs were seeded in 96-well plates (3×103 cells/well) and cultured for 24 hours. Cells were pretreated with SQ22536 (1–10 μM) for 1 hour, then stimulated with isoproterenol (1 μM) and cultured for 72 hours. Cell viability was assessed by MTT assay (absorbance at 570 nm), and proliferation inhibition rate was calculated [1] - Glomerular mesangial cell ECM synthesis assay: GMCs were seeded in 6-well plates (2×105 cells/well) and treated with SQ22536 (5–20 μM) and angiotensin II (100 nM) for 48 hours. Cells were lysed, and collagen IV protein levels were quantified by ELISA. Total protein concentration was used for normalization [5] - Pituitary cell hormone secretion assay: Rat anterior pituitary cells were dispersed and seeded in 24-well plates (1×105 cells/well) in serum-free medium. Cells were pretreated with SQ22536 (2–10 μM) for 1 hour, then stimulated with GnRH (100 nM) for 2 hours. Culture supernatants were collected, and LH levels were measured by radioimmunoassay [3] - Western blot analysis for PKA signaling: RASMCs treated with SQ22536 (5 μM) and isoproterenol (1 μM) for 24 hours were lysed. Proteins were separated by SDS-PAGE, transferred to PVDF membranes, and probed with antibodies against p-PKA, PKA, and β-actin. Band intensity was quantified by densitometry [1] |
| Animal Protocol |
Dissolved in saline; 10 mg/kg; s.c.
Male C57BL/6J mice Male guinea-pigs (Duncan Harver, 250–300 g) were killed by cervical dislocation and the upper thoracic aorta removed and placed in physiological saline solution (PSS) containing in mm; NaCl 112, KCl 5, CaCl2 1.8, MgCl2 1, NaHCO3 25, KH2PO4 0.5, NaH2PO4 0.5, glucose 10 and 0.03 phenol red (pH 7.4 with 95%O2/5% CO2). The tissue was cut into rings (∼2 mm wide, and cut longitudinally) or helical strips (∼2 mm wide, 10 mm long). The endothelium was denuded by gently rubbing the lumen of the muscle with filter paper. Tissues were mounted in an organ bath (0.3 ml) and tension measured isometrically. Muscle strips were subjected to a basal tension of 1.25 g and continuously perfused with PSS (1.0 ml min−1 at 37°C) for 30 min before being precontracted with phenylephrine (1 or 6 μm). Removal of the endothelium was confirmed by lack of relaxation to acethylcholine (10 μm for 2 min). Concentration-response curves were obtained by cumulatively applying iloprost (1–1000 nm) in the absence or presence of the adenylyl cyclase inhibitor, SQ22536 (100 μm). SQ22536 was given 30 min before the addition of iloprost and remained throughout. A 1 h wash-out period was allowed between concentration-response curves.[6] The stable prostacyclin analogue, iloprost relaxes a variety of blood vessels and increases cyclic AMP, although the relationship between adenosine 3': 5'-cyclic monophosphate (cyclic AMP) and vasorelaxation remains unclear. We therefore investigated the effect of the adenylyl cyclase inhibitor, 9-(tetrahydro-2-furanyl)-9H-purin-6-amine (SQ22536) on iloprost-mediated relaxation and cyclic AMP elevation in endothelium-denuded aortic strips. Iloprost (1-1000 nM) caused a concentration-dependent inhibition of phenylephrine (1-6 microM) contractions, the responses being unaffected by pre-incubation with SQ22536 (100 microM) for 30 min. In other experiments 60 nM iloprost caused a 64% inhibition of phenylephrine contractions concomitant with a 3 fold rise in cyclic AMP. SQ22536 completely abolished the iloprost-induced elevation in cyclic AMP while having no significant effect on relaxation. Our results therefore strongly suggest that cyclic AMP-independent pathways are responsible for the vasorelaxant effects of iloprost in guinea-pig aorta.[6] Hypertensive rat model: Male SHR (12–14 weeks old, n=8/group) were anesthetized with isoflurane. SQ22536 was dissolved in 10% DMSO + 90% sterile saline and administered intravenously at 5 mg/kg. Blood pressure was measured by carotid artery catheterization at 0, 15, 30, 60, and 120 minutes post-administration. Aortic tissues were collected at the end of the experiment to measure cAMP content [1] - Renal fibrosis model: C57BL/6 mice (8–10 weeks old, male) were subjected to unilateral ureteral obstruction (UUO) to induce renal fibrosis. One day after surgery, mice were randomly divided into 2 groups (n=7/group): vehicle (10% DMSO + 90% saline) and SQ22536 (10 mg/kg, i.p.). The drug was administered once daily for 14 days. Mice were euthanized, and the obstructed kidneys were collected for collagen deposition analysis (Masson’s trichrome staining) and PCNA immunostaining [5] - Insulin sensitivity model: C57BL/6 mice (10 weeks old, male) were fed a high-fat diet for 8 weeks to induce insulin resistance. Mice were divided into 2 groups (n=6/group): vehicle and SQ22536 (10 mg/kg, i.v.). Hepatic glucose production was measured by pyruvate tolerance test (PTT) 2 hours after drug administration. Liver tissues were collected to detect cAMP levels [4] - Pituitary hormone secretion model: Male Wistar rats (8–10 weeks old, n=6/group) were anesthetized, and SQ22536 (7.5 mg/kg, i.p.) or vehicle was administered. Thirty minutes later, GnRH (10 μg/kg) was injected intravenously. Blood samples were collected via tail vein at 0, 15, 30, 60 minutes post-GnRH injection, and serum LH levels were measured by RIA [3] |
| ADME/Pharmacokinetics |
Plasma protein binding rate: The plasma protein binding rate of SQ22536 in human plasma was 88% and that in rat plasma was 85% as determined by equilibrium dialysis [6]
- Metabolic stability: SQ22536 showed moderate metabolic stability in rat liver microsomes, with 65% of the parent compound remaining after 60 minutes of incubation [6] - Half-life: The elimination half-life (t1/2) of SQ22536 (5 mg/kg) administered intravenously to rats was 3.2 hours [1] - Tissue distribution: After intravenous administration of SQ22536 to rats, it was widely distributed in various tissues, with the highest concentrations in the liver, kidneys and blood vessels (the liver/plasma ratio was 2.7 and the kidney/plasma ratio was 2.3 1 hour after administration) [6] |
| Toxicity/Toxicokinetics |
Acute toxicity: No deaths or significant clinical toxicities (e.g., drowsiness, hypotension, dyspnea) were observed in rats after a single intravenous injection of SQ22536 (up to 100 mg/kg) within 14 days [6]
- Repeated-dose toxicity: No significant changes were observed in serum ALT, AST, BUN, or creatinine levels after intraperitoneal injection of SQ22536 (5-20 mg/kg) once daily for 28 consecutive days. No pathological abnormalities were observed in histological examination of liver, kidney, heart, and aorta tissues [1] - No significant changes were observed in hematological parameters (red blood cell count, white blood cell count, platelet count) after intraperitoneal injection of SQ22536 (10 mg/kg) once daily for 14 consecutive days in rats [5] |
| References | |
| Additional Infomation |
9-(tetrahydrofuranyl)adenine is a nucleoside analog with the structure adenine, where the nitrogen atom at position 9 is replaced by a tetrahydrofuran-2-yl group. It is an adenylate cyclase inhibitor, belonging to EC 4.6.1.1 (adenylate cyclase) inhibitors. It is a nucleoside analog and also an oxacyclopentane compound. Its function is related to adenine. This study investigated the effect of adenylate cyclase inhibitors on isoproterenol-induced vasodilation in isolated pulmonary veins of newborn lambs (7-12 days old). In endothelin-1-constricted veins, isoproterenol at concentrations ≤3 × 10⁻⁹ M had no effect on cyclic adenosine monophosphate (cAMP) levels but induced up to 56% vasodilation. At high concentrations (≥10⁻⁸ M), isoproterenol increased cAMP levels and led to further vasodilation. In veins constricted by endothelin-1 or U46619 (9,11-dideoxy-11,9-epoxymethoxyprostaglandin, i.e., prostaglandin F2α), the isoproterenol-induced increase in cAMP (rather than vasodilation) can be blocked by SQ 22536 [9-(tetrahydro-2-furanyl)-9H-purine-6-amine; an adenylate cyclase inhibitor]. Propranolol inhibits the effects of isoproterenol on vascular tone and cAMP levels. Rp-8-CPT-cAMPS [8-(4-chlorophenylthio)-adenosine-3',5'-cyclic monophosphate thioester, Rp-isomer] and Rp-8-Br-PET-cGMPS [β-phenyl-1,N2-vinyl-8-bromoguanosine-3',5'-cyclic monophosphate thioester, Rp-isomer] are inhibitors of cAMP and guanosine-3',5'-cyclic monophosphate (cGMP)-dependent protein kinases, respectively. They attenuated the vasodilatory effect induced by cAMP analogs, but did not attenuate the vasodilatory effect induced by isoproterenol. In the pulmonary vein rough membrane preparation, the increase in adenylate cyclase activity induced by isoproterenol could be blocked by propranolol and SQ 22536. These results suggest that cAMP may not play a key role in isoproterenol-induced pulmonary vein vasodilatory in newborn lambs. [1] Mast cells are involved in allergic reactions, as well as in innate immunity and inflammation. Corticotropin-releasing hormone (CRH) is a key regulator of the hypothalamic-pituitary-adrenal axis and also has pro-inflammatory effects, apparently mediated by mast cells. Our recent research shows that CRH selectively stimulates the release of newly synthesized vascular endothelial growth factor (VEGF) from human leukemia mast cells and human cord blood-derived mast cells, without releasing pre-formed mediators or cytokines. This effect is mediated by activation of CRH receptor-1 and adenylate cyclase, and by increased intracellular cAMP levels. However, the exact mechanism by which CRH induces VEGF secretion remains unclear. This study demonstrates that CRH-induced VEGF release can be dose-dependently inhibited by the specific protein kinase A inhibitor N-[2-(4-bromocinnamylamino)ethyl]-5-isoquinoline (H89) or the p38 mitogen-activated protein kinase (MAPK) inhibitor 4-(4-fluorophenyl)-2-(4-methylsulfinylphenyl)-5-(4-pyridyl)-1H-imidazole (SB203580), but is not inhibited by the specific inhibitors of mitogen-activated protein kinase (MAPK), extracellular signal-regulated kinase (ERK) upstream kinase 2'-amino-3'-methoxyflavone (PD98059), or the c-Jun N-terminal kinase (JNK) inhibitor 1,9-pyrazol-6(2H)-one (SP600125). In addition, CRH significantly enhanced the activity of protein kinase A, an effect that can be mimicked by the cell membrane permeability cAMP analog 8-bromo-cAMP and blocked by H89 or the adenylate cyclase inhibitor 9-(tetrahydro-2-furanyl)-9H-purine-6-amine (SQ22536). CRH also induced rapid phosphorylation of p38 MAPK, a phosphorylation that can be mimicked by 8-bromo-cAMP and blocked by H89 or SB203580. CRH did not stimulate phosphorylation of ERK or JNK, nor did it increase intracellular calcium ion levels. These results suggest that CRH induces the release of VEGF from human mast cells by selectively activating the cAMP/protein kinase A/p38 MAPK signaling pathway, thereby further revealing the molecular mechanism by which CRH affects the release of key pro-inflammatory mediators. [2]
SQ22536 (NSC-53339) is a classic small molecule inhibitor of adenylate cyclase (AC) and is widely used as a tool compound for studying cAMP-mediated signaling pathways. [6] - Its mechanism of action involves competitive binding to the catalytic domain of AC, preventing the conversion of ATP to cAMP, thereby inhibiting downstream cAMP/PKA signaling cascades involved in cell proliferation, hormone secretion, and tissue remodeling. [2] - SQ22536 shows potential therapeutic value in cardiovascular diseases (e.g., hypertension, vascular remodeling), renal fibrosis, and metabolic disorders (e.g., insulin resistance) by targeting AC-mediated pathological processes. [1,4,5] - The drug's selectivity for adenylate cyclase (AC) is much higher than that for other signaling enzymes, making it a reliable tool for elucidating cAMP-dependent biological functions. [6] - SQ22536 does not cross-react with G protein-coupled receptors (GPCRs) or ion channels at therapeutic concentrations, thereby minimizing off-target effects. [2] |
| Molecular Formula |
C9H11N5O
|
|
|---|---|---|
| Molecular Weight |
205.22
|
|
| Exact Mass |
205.096
|
|
| Elemental Analysis |
C, 52.67; H, 5.40; N, 34.13; O, 7.80
|
|
| CAS # |
17318-31-9
|
|
| Related CAS # |
|
|
| PubChem CID |
5270
|
|
| Appearance |
White to off-white solid powder
|
|
| Density |
1.7±0.1 g/cm3
|
|
| Boiling Point |
474.8±55.0 °C at 760 mmHg
|
|
| Melting Point |
160-161ºC
|
|
| Flash Point |
241.0±31.5 °C
|
|
| Vapour Pressure |
0.0±1.2 mmHg at 25°C
|
|
| Index of Refraction |
1.831
|
|
| LogP |
-0.17
|
|
| Hydrogen Bond Donor Count |
1
|
|
| Hydrogen Bond Acceptor Count |
5
|
|
| Rotatable Bond Count |
1
|
|
| Heavy Atom Count |
15
|
|
| Complexity |
239
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
O1C([H])([H])C([H])([H])C([H])([H])C1([H])N1C([H])=NC2=C(N([H])[H])N=C([H])N=C12
|
|
| InChi Key |
UKHMZCMKHPHFOT-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C9H11N5O/c10-8-7-9(12-4-11-8)14(5-13-7)6-2-1-3-15-6/h4-6H,1-3H2,(H2,10,11,12)
|
|
| Chemical Name |
9-(oxolan-2-yl)purin-6-amine
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (12.18 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (12.18 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (12.18 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 25 mg/mL (121.82 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.8728 mL | 24.3641 mL | 48.7282 mL | |
| 5 mM | 0.9746 mL | 4.8728 mL | 9.7456 mL | |
| 10 mM | 0.4873 mL | 2.4364 mL | 4.8728 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
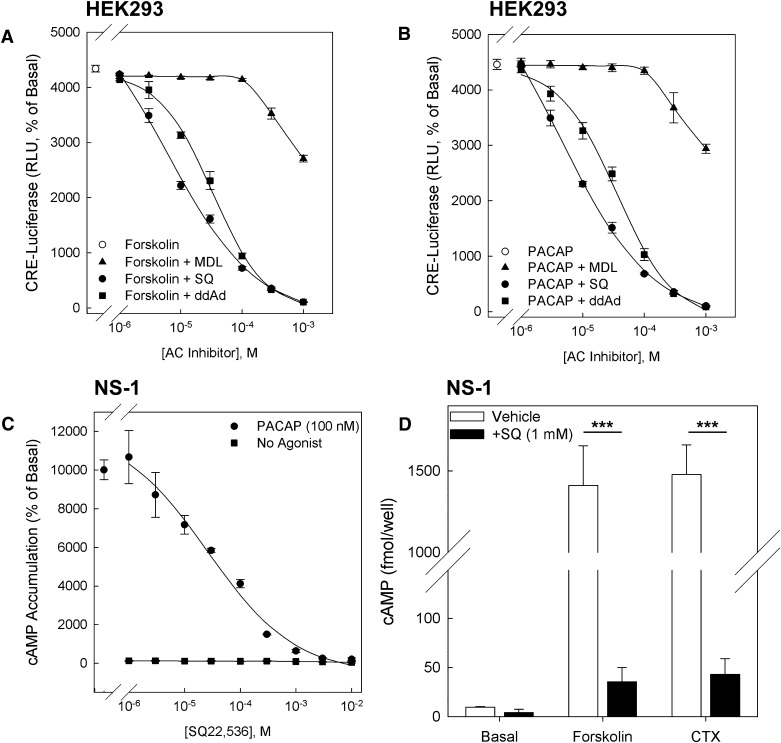 Evaluation of cell-permeable AC inhibitors.Mol Pharmacol.2013 Jan;83(1):95-105. |
|---|
SQ22,536 inhibits cAMP-dependent ERK phosphorylation, but not NGF or PMA-induced ERK phosphorylation.Mol Pharmacol.2013 Jan;83(1):95-105. |
SQ22,536 inhibits cAMP-dependent Elk activation.Mol Pharmacol.2013 Jan;83(1):95-105. |