
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
SB525334 (SB-525334; SB 525334) is a potent and selective inhibitor of TGF-β (transforming growth factor-β) receptor I, ALK5 (activin receptor-like kinase) with potential anti-fibrotic activity. It inhibits TGF-β with an IC50 of 14.3 nM in a cell-free assay. SB525334 is 4-fold less potent to ALK4 than ALK5 and is inactive against ALK2, 3, and 6. In cell-based assays, SB-525334 at the concentration of 1 μM blocked TGF-β1-induced phosphorylation and nuclear translocation of Smad2/3 in renal proximal tubule cells and inhibited TGF-β1-induced increases in plasminogen activator inhibitor-1 (PAI-1) and procollagen alpha1(I) mRNA expression in A498 renal epithelial carcinoma cells.
| Targets |
ALK (IC50 = 14.3 nM)
SB525334 specifically targets transforming growth factor-beta type I receptor (TGF-β RI/ALK5) (ALK5 IC50 = 14 nM) [1][2] SB525334 shows weak or no inhibition of other ALK receptors (ALK1, ALK2, ALK3, ALK4: IC50 > 1 μM) and unrelated kinases (PKA, PKC: IC50 > 10 μM) [1][2] |
|---|---|
| ln Vitro |
The study found that SB525334 (1 μM) reduces the proliferation of familial idiopathic pulmonary arterial hypertension (iPAH) pulmonary artery smooth muscle cells (PASMCs) with an IC50 of 295 nM. The effect is observed 15 minutes before stimulating with 0.625 ng/ml of TGF-β1, and evaluation is done after 6 days.
In rat renal mesangial cells treated with TGF-β1, SB525334 (1 μM) inhibits Smad2 phosphorylation by 82% after 24 hours. It downregulates mRNA expression of fibrosis-related genes (Col1α1: 65% reduction; Col3α1: 60% reduction; TGF-β1: 58% reduction) and reduces collagen protein synthesis by 55% [1] - In primary vascular smooth muscle cells (VSMCs) isolated from patients with familial pulmonary arterial hypertension (FPAH), SB525334 (5 μM) inhibits abnormal cell proliferation by 68% after 72 hours (MTT assay). It blocks TGF-β-induced Smad2 phosphorylation (75% reduction) and downregulates cyclin D1 expression by 62% at protein level [2] - In renal tumor cells derived from Eker rats (TSC2 mutant), SB525334 (10 μM) inhibits cell proliferation by 55% after 48 hours and induces G1 cell cycle arrest (G1 phase cells increased from 40% to 62%). It suppresses TGF-β-mediated upregulation of c-Myc and Cyclin E1 (50-55% reduction at mRNA level) [3] - In normal human renal proximal tubular cells (HRPTCs) and VSMCs, SB525334 shows low toxicity at concentrations up to 25 μM (cell viability > 85% vs. control) [1][2] |
| ln Vivo |
In a rat model of pulmonary arterial hypertension (PAH), SB525334 (3-30 mg/kg; po; daily from days 17 to 35) dramatically reverses pulmonary arterial pressure[2].
In view of this profile, SB-525334 was used to investigate the role of TGF-beta1 in the acute puromycin aminonucleoside (PAN) rat model of renal disease, a model of nephritis-induced renal fibrosis. Orally administered doses of 1, 3, or 10 mg/kg/day SB-525334 for 11 days produced statistically significant reductions in renal PAI-1 mRNA. Also, the compound produced dose-dependent decreases in renal procollagen alpha1(I) and procollagen alpha1(III) mRNA, which reached statistical significance at the 10-mg/kg/day dose when compared with vehicle-treated PAN controls. Furthermore, PAN-induced proteinuria was significantly inhibited at the 10-mg/kg/day dose level. These results provide further evidence for the involvement of TGF-beta1 in the profibrotic changes that occur in the PAN model and for the first time, demonstrate the ability of a small molecule inhibitor of ALK5 to block several of the markers that are predictive of fibrosis and renal injury in this model.[1] We further demonstrate that SB525334 significantly reverses pulmonary arterial pressure and inhibits right ventricular hypertrophy in a rat model of PAH. Immunohistochemical studies confirmed a significant reduction in pulmonary arteriole muscularization induced by monocrotaline (used experimentally to induce PAH) after treatment of rats with SB525334. Collectively, these data are consistent with a role for the activin receptor-like kinase 5 in the progression of idiopathic PAH and imply that strategies to inhibit activin receptor-like kinase 5 signaling may have therapeutic benefit[2]. Blockade of TGF-beta signaling with the ALK5/type I TGF-beta R kinase inhibitor, SB-525334, was efficacious for uterine leiomyoma; significantly decreasing tumor incidence and multiplicity, and reducing the size of these mesenchymal tumors. However, SB-525334 was also mitogenic and antiapoptotic for epithelial cells in the kidney and exacerbated the growth of epithelial lesions present in the kidneys of these animals. Conclusion: Although pharmacologic inhibition of TGF-beta signaling with SB-525334 may be efficacious for mesenchymal tumors, inhibition of this signaling pathway seems to promote the development of epithelial tumors.[3] In rats with puromycin-induced nephritis (PIN), oral administration of SB525334 (100 mg/kg/day for 21 days) reduces renal fibrosis. Glomerular and tubular collagen deposition is decreased by 60% (Masson’s trichrome staining), and mRNA levels of Col1α1, Col3α1, and TGF-β1 in renal tissues are downregulated by 58%, 55%, and 52% respectively. It also improves renal function (urinary protein excretion reduced by 45%) [1] - In monocrotaline (MCT)-induced pulmonary arterial hypertension (PAH) rats, intraperitoneal administration of SB525334 (30 mg/kg/day for 28 days) attenuates disease progression. Mean pulmonary arterial pressure (mPAP) is reduced from 45 mmHg (vehicle) to 28 mmHg, and pulmonary vascular remodeling is inhibited (medial wall thickness reduced by 55%). Lung tissues show downregulated p-Smad2 (68% reduction) and α-SMA (60% reduction) [2] - In Eker rats with hereditary renal tumors, oral SB525334 (50 mg/kg/day for 8 weeks) inhibits tumor growth. Tumor volume is reduced by 63% compared to vehicle controls, and tumor proliferation index (Ki-67-positive cells) is decreased by 58%. Renal tumor tissues show suppressed Smad2 phosphorylation and reduced c-Myc expression [3] |
| Enzyme Assay |
SB-525334 (6-[2-tert-butyl-5-(6-methyl-pyridin-2-yl)-1H-imidazol-4-yl]-quinoxaline) has been characterized as a potent and selective inhibitor of the transforming growth factor-beta1 (TGF-beta1) receptor, activin receptor-like kinase (ALK5). The compound inhibited ALK5 kinase activity with an IC(50) of 14.3 nM and was approximately 4-fold less potent as an inhibitor of ALK4 (IC(50) = 58.5 nM). SB-525334 was inactive as an inhibitor of ALK2, ALK3, and ALK6 (IC(50) > 10,000 nM) [1].
ALK5 kinase activity assay: Purified recombinant human ALK5 was incubated with Smad3-derived substrate peptide and SB525334 (0.1 nM-100 nM) in assay buffer (50 mM Tris-HCl, pH 7.5, 10 mM MgCl₂, 1 mM DTT, 0.1 mM ATP) at 30°C for 60 minutes. Phosphorylated substrate was detected by radiolabeled ATP counting, and IC50 values were calculated from dose-response curves [1][2] - Kinase selectivity assay: SB525334 (10 μM) was screened against a panel of 30+ kinases (including ALK1-4, PKA, PKC, ERK1/2) using respective substrate peptides and assay buffers. Kinase activity was quantified by colorimetric assay, with no significant off-target inhibition (>50% activity reduction) observed [1] |
| Cell Assay |
Cell Proliferation Assay[2]
Cell Types: PASMC cells Tested Concentrations: 1 μM Incubation Duration: Pre-incubated for 15 minutes (before stimulating with 0.625 ng/ml of TGF-β1), assessed after 6 days Experimental Results: Inhibited TGF-β1-mediated proliferation of familial iPAH PASMCs at an IC50 of 295 nM. Renal mesangial cell fibrosis assay: Rat renal mesangial cells were seeded in 6-well plates at 2×10⁵ cells/well and activated with TGF-β1 (10 ng/mL) for 24 hours. SB525334 (0.1-5 μM) was added, and cells were cultured for 48 hours. Western blot detected p-Smad2 and total Smad2; qPCR analyzed Col1α1/Col3α1/TGF-β1 mRNA levels; ELISA measured collagen protein synthesis [1] - FPAH VSMC proliferation assay: Primary human FPAH VSMCs were seeded in 96-well plates at 3×10³ cells/well and treated with SB525334 (0.5-10 μM) for 72 hours. MTT assay assessed cell viability; Western blot detected cyclin D1 and p-Smad2; flow cytometry analyzed cell cycle distribution [2] - Eker rat renal tumor cell assay: Renal tumor cells from Eker rats were seeded in 6-well plates at 1.5×10⁵ cells/well and treated with SB525334 (1-20 μM) for 48 hours. Cell proliferation was measured by CCK-8 assay; qPCR detected c-Myc/Cyclin E1 mRNA levels; flow cytometry analyzed cell cycle (propidium iodide staining) [3] |
| Animal Protocol |
Animal/Disease Models: Adult male SD (Sprague-Dawley) rats (MCT rat model of pulmonary hypertension)[2]
\nDoses: 3, 30 mg/kg \nRoute of Administration: Oral administration; daily from days 17 to 35 \nExperimental Results: decreased the proportion of fully muscularized vessels to 28% at 3 mg/kg and returned fully muscularized vessel distribution beyond that seen at day 17 and approaching the phenotype observed in saline-exposed controls at 30 mg/kg. \nMCT Rat Model of Pulmonary Hypertension[2] \nAnimals were housed at 24°C in a 12-hour light-dark cycle. Food and water were accessible ad libitum. The studies reported here conformed to the UK Animals (scientific procedures) Act 1986. MCT-induced PAH was performed as previously described.15 Briefly, adult male Sprague-Dawley rats (n = 10 per group) were anesthetized and subcutaneously injected with 40 mg/kg of MCT or sterile saline. Before commencement of dosing at day 17 the extent of hypertensive pathology was determined in animals (n = 5) per group via echocardiography. A further group of animals was also assessed via surgery and catheterization. SB-525334 compound was dosed orally (3 or 30 mg/kg) or vehicle alone was dosed daily until day 35, when the remaining animals were reassessed by echocardiography, surgery, and catheterization.[2] \nIn vivo study. [3] \nThe protocols involving the use of these rats were approved by the M.D. Anderson Cancer Center Institutional Animal Care and Use Committee. Animals were maintained on a 12 h light/dark cycle, with food and water provided ad libitum. To determine the effects of a TGF-β receptor inhibitor on uterine leiomyoma, female Eker rats 12 or 14 months old were given SB-525334 at a dose of 200 mg/L drinking water (estimated dose of 10 mg/kg/d) or received normal drinking water for 2 and 4 months. At 16 months of age, animals were sacrificed by CO2 asphyxiation and tissues were harvested and either snap-frozen in liquid nitrogen and stored at −80°C or fixed in 10% neutral buffered formalin and paraffin embedded. To further analyze the effects of SB-525334 on kidneys, 9-month-old male Eker rats were given plain drinking water or the compound in drinking water at 200 mg/L for 2 months. Rats were then sacrificed and tissues were harvested, fixed, and stored as described above. For histology, tissues were stained with H&E, and kidneys and multiple sections of female reproductive tract (uterus, vagina, and cervix) were examined microscopically by a pathologist blinded as to treatment group (see below). All tumors and proliferative lesions were identified and evaluated as previously described.[3] \nRat puromycin-induced nephritis (PIN) model: Male Sprague-Dawley rats were injected intraperitoneally with puromycin (15 mg/kg) to induce nephritis. One week post-induction, SB525334 was suspended in 0.5% carboxymethylcellulose sodium and administered orally at 100 mg/kg/day for 21 days. Vehicle group received carboxymethylcellulose sodium. Urinary protein excretion was measured weekly; renal tissues were collected for Masson’s trichrome staining and qPCR (fibrosis-related genes) [1] \n- Rat MCT-induced PAH model: Male Wistar rats were injected subcutaneous with monocrotaline (60 mg/kg) to induce PAH. Seven days post-injection, SB525334 was dissolved in saline and administered intraperitoneally at 30 mg/kg/day for 28 days. Vehicle group received saline. Mean pulmonary arterial pressure (mPAP) was measured by catheterization; lung tissues were collected for α-SMA immunostaining and Western blot (p-Smad2) [2] \n- Eker rat renal tumor model: Female Eker rats (6-week-old) with hereditary renal tumors were randomly divided into vehicle and SB525334 groups. SB525334 was suspended in 0.5% carboxymethylcellulose sodium and administered orally at 50 mg/kg/day for 8 weeks. Vehicle group received carboxymethylcellulose sodium. Tumor volume was measured every 2 weeks; renal tissues were collected for Ki-67 immunostaining and Western blot (c-Myc, p-Smad2) [3] |
| Toxicity/Toxicokinetics |
In vitro experiments showed that SB525334 had low cytotoxicity to normal human cells (HRPTCs IC50 > 25 μM; normal VSMCs IC50 > 30 μM) [1][2]
- In vivo studies showed that oral or intraperitoneal injection of SB525334 at the test dose (30-100 mg/kg/day) did not cause significant weight loss (<5% vs. baseline) or significant death in rats [1][2][3] - Compared with the vector control group, there were no significant changes in liver function (ALT, AST) or kidney function (creatinine, BUN) in the SB525334 treatment group [1][3] - The plasma protein binding rate of SB525334 in rats was 91-94% (in vitro plasma binding assay) [1][2] |
| References |
|
| Additional Infomation |
6-[2-tert-butyl-5-(6-methyl-2-pyridinyl)-1H-imidazol-4-yl]quinoxaline is a quinoxaline derivative. SB-525334 (6-[2-tert-butyl-5-(6-methyl-pyridinyl)-1H-imidazol-4-yl]quinoxaline) has been shown to be a potent and selective inhibitor of transforming growth factor-β1 (TGF-β1) receptor activator receptor-like kinase (ALK5). This compound exhibits an IC50 of 14.3 nM for inhibiting ALK5 kinase activity, while its inhibitory potency against ALK4 is approximately one-quarter that against ALK5 (IC50 = 58.5 nM). SB-525334 has no inhibitory activity against ALK2, ALK3, and ALK6 (IC50 > 10,000 nM). In cellular experiments, SB-525334 (1 μM) blocked TGF-β1-induced phosphorylation and nuclear translocation of Smad2/3 in proximal renal tubules and inhibited the increase in plasminogen activator inhibitor-1 (PAI-1) and type I procollagen mRNA expression in TGF-β1-induced A498 renal epithelial carcinoma cells. Therefore, this study used SB-525334 to investigate the role of TGF-β1 in an acute puromycin aminonucleoside (PAN) rat model of renal disease (a nephritis-induced renal fibrosis model). Oral administration of SB-525334 at doses of 1, 3, or 10 mg/kg/day for 11 consecutive days significantly reduced renal PAI-1 mRNA expression. Furthermore, this compound dose-dependently reduced renal type I and type III procollagen mRNA expression, with statistically significant reductions observed in the 10 mg/kg/day dose group compared to the vector-treated PAN control group. Moreover, the 10 mg/kg/day dose group significantly inhibited PAN-induced proteinuria. These results further confirm that TGF-β1 is involved in the profibrotic changes that occur in the PAN model and demonstrate for the first time that small molecule ALK5 inhibitors can block several biomarkers predicting fibrosis and kidney injury in this model. [1] Mutations in the gene of the transforming growth factor (TGF)-β superfamily receptor bone morphogenetic protein receptor II are the cause of hereditary pulmonary hypertension (PAH). Aberrant signaling of TGF-β receptor I/activin receptor-like kinase 5 may be crucial for the occurrence and development of PAH. We investigated the potential of a well-characterized and highly potent activin receptor-like kinase 5 inhibitor, SB525334 [6-(2-tert-butyl-5-{6-methyl-pyridin-2-yl}-1H-imidazol-4-yl)-quinoxaline], to treat pulmonary hypertension (PAH). This study showed that pulmonary artery smooth muscle cells from patients with familial idiopathic PAH exhibited higher sensitivity to TGF-β1 in vitro, and SB525334 reduced this sensitivity. We further demonstrated that SB525334 significantly reversed pulmonary artery pressure and inhibited right ventricular hypertrophy in a PAH rat model. Immunohistochemical studies confirmed that treatment of rats with SB525334 significantly reduced pulmonary arteriolar myoplasia induced by monoclonal ketamine (used in the experiment to induce pulmonary hypertension). Overall, these data are consistent with the role of activin receptor-like kinase 5 in the progression of idiopathic pulmonary hypertension and suggest that inhibition of the activin receptor-like kinase 5 signaling pathway may have therapeutic benefits. [2]
Objective: Transforming growth factor β (TGF-β) usually stimulates the growth of mesenchymal-derived cells but inhibits the growth of epithelial cells, and is therefore considered a potential target for cancer therapy. However, there are concerns that while inhibition of the TGF-β signaling pathway may be effective against lesions that promote TGF-β tumorigenesis and/or progression, systemic drug blockade of this signaling pathway may also promote the growth of epithelial lesions. Experimental design: We investigated the effects of TGF-β inhibitors on mesenchymal tumors (uterine fibroids) and epithelial tumors (renal cell carcinoma) in Eker rats. Eker rats have a high tumor incidence and are genetically susceptible. Results: Blocking the TGF-β signaling pathway with the ALK5/type I TGF-β receptor kinase inhibitor SB-525334 effectively treated uterine fibroids, significantly reducing tumor incidence and number, and shrinking the volume of these stromal tumors. However, SB-525334 had mitogenic and anti-apoptotic effects on renal epithelial cells and exacerbated the growth of pre-existing epithelial lesions in the kidneys of these animals. Conclusion: While pharmacological inhibition of the TGF-β signaling pathway using SB-525334 may be effective for stromal tumors, inhibition of this signaling pathway appears to promote the development and progression of epithelial tumors. [3] SB525334 is a potent, selective small molecule TGF-β type I receptor (ALK5) inhibitor[1][2] - Its mechanism of action involves competitive binding to the ATP-binding pocket of ALK5, inhibiting its kinase activity and blocking the phosphorylation of downstream Smad2/3, thereby inhibiting TGF-β-mediated transcriptional activation of profibrotic, pro-proliferative and pro-tumor genes[1][2][3] - SB525334 has shown antifibrotic and anti-proliferative activity in the kidney, blood vessels and tumor cells in vitro, and has therapeutic effects in vivo. Nephritis, pulmonary hypertension, and renal tumor models [1][2][3] - It has been widely used as a tool compound to study the role of the TGF-β/ALK5 signaling pathway in fibrosis, cardiovascular disease, and tumorigenesis [1][2][3] - The drug's selectivity for ALK5 and its manageable toxicity support its potential application in treating TGF-β-driven diseases such as renal fibrosis, pulmonary hypertension, and certain tumors [1][2][3] |
| Molecular Formula |
C21H21N5
|
|
|---|---|---|
| Molecular Weight |
343.42
|
|
| Exact Mass |
343.179
|
|
| Elemental Analysis |
C, 73.44; H, 6.16; N, 20.39
|
|
| CAS # |
356559-20-1
|
|
| Related CAS # |
|
|
| PubChem CID |
9967941
|
|
| Appearance |
Yellow to orange solid
|
|
| Density |
1.2±0.1 g/cm3
|
|
| Boiling Point |
540.5±45.0 °C at 760 mmHg
|
|
| Melting Point |
159 °C
|
|
| Flash Point |
238.6±21.7 °C
|
|
| Vapour Pressure |
0.0±1.4 mmHg at 25°C
|
|
| Index of Refraction |
1.636
|
|
| LogP |
4.05
|
|
| Hydrogen Bond Donor Count |
1
|
|
| Hydrogen Bond Acceptor Count |
4
|
|
| Rotatable Bond Count |
3
|
|
| Heavy Atom Count |
26
|
|
| Complexity |
476
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
N1([H])C(C2=C([H])C([H])=C([H])C(C([H])([H])[H])=N2)=C(C2C([H])=C([H])C3C(C=2[H])=NC([H])=C([H])N=3)N=C1C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H]
|
|
| InChi Key |
DKPQHFZUICCZHF-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C21H21N5/c1-13-6-5-7-16(24-13)19-18(25-20(26-19)21(2,3)4)14-8-9-15-17(12-14)23-11-10-22-15/h5-12H,1-4H3,(H,25,26)
|
|
| Chemical Name |
6-(2-(tert-butyl)-5-(6-methylpyridin-2-yl)-1H-imidazol-4-yl)quinoxaline
|
|
| Synonyms |
SB 525334; SB-525334; 6-(2-(tert-Butyl)-5-(6-methylpyridin-2-yl)-1H-imidazol-4-yl)quinoxaline; 6-[2-TERT-BUTYL-5-(6-METHYL-PYRIDIN-2-YL)-1H-IMIDAZOL-4-YL]-QUINOXALINE; MFCD11045307; 6-[2-tert-butyl-5-(6-methylpyridin-2-yl)-1H-imidazol-4-yl]quinoxaline; SB525334
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.28 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.28 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: 5% DMSO+corn oil:20 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9119 mL | 14.5594 mL | 29.1189 mL | |
| 5 mM | 0.5824 mL | 2.9119 mL | 5.8238 mL | |
| 10 mM | 0.2912 mL | 1.4559 mL | 2.9119 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
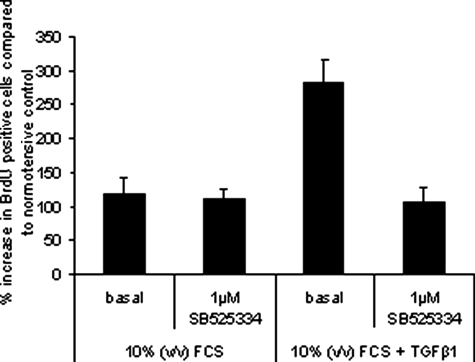 PASMCs derived from iPAH patients were plated at equal cell densities in 96-well plates.
Echocardiographic measurement of pulmonary hypertensive parameters in animals.Am J Pathol.2009 Feb;174(2):380-9. |
RV systolic pressure levels (A) and Fulton index measures (RV/LV + S weight ratio) (B) in rats exposed to MCT or saline-negative control.Am J Pathol.20 |