
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
Ralimetinib dimysylate (also known as LY-2228820; LY2228820) is a novel, potent and selective ATP-competitive inhibitor of p38 MAPK with potential anti-inflammatory activity. In a cell-free assay, it inhibits p38 MAPK with an IC50 of 7 nM and does not affect p38 MAPK activation. In cell-based assays, LY2228820 potently and selectively inhibited phosphorylation of MK2 (Thr334) in anisomycin-stimulated HeLa cells (at 9.8 nmol/L by Western blot analysis) and anisomycin-induced mouse RAW264.7 macrophages (IC(50) = 35.3 nmol/L) with no changes in phosphorylation of p38α MAPK, JNK, ERK1/2, c-Jun, ATF2, or c-Myc ≤ 10 μmol/L.
| Targets |
p38β MAPK (IC50 = 3.2 nM); p38α MAPK (IC50 = 5.8 nM)
p38α (IC₅₀ = 0.0003 μM; Ki = 0.0002 μM); the compound showed >1000-fold selectivity over p38β/γ/δ (IC₅₀ >0.3 μM) and >500-fold selectivity over other MAPKs (ERK1/2: IC₅₀ >1 μM; JNK1/2: IC₅₀ >1 μM) and 50+ non-MAPK kinases (e.g., AKT, EGFR, RAF1) when tested at 10 μM [1] |
|---|---|
| ln Vitro |
Ralimetinib dimesylate inhibits p38α and the quantity of phosphoMAPKAPK-2 (pMK2) in RAW 264.7 cells with IC50 values of 7 nM and 34.3 nM, respectively. Additionally, Ralimetinib dimesylate inhibits lipopolysaccharide (LPS)-induced TNFα synthesis in murine peritoneal macrophages with an IC50 of 5.2 nM[1]. Ralimetinib dimesylate (LY2228820) (200 nM-800 nM) significantly inhibits p38MAPK signaling in multiple myeloma (MM) cells, including INA6, RPMI-8226, U266 and RPMI-Dox40, as shown by its inhibition on phosphorylation of HSP27, a downstream target of p38MAPK, without changing the expression level of HSP27. Ralimetinib dimesylate (200 nM–400 nM) increases bortezomib-induced cytotoxicity and apoptosis, but Ralimetinib dimesylate alone does not inhibit the growth of MM.1S cells. Additionally, ralimetinib dimesylate (200 nM-800 nM) inhibits the release of IL-6 and MIP-1 from long-term BM stromal cells (LT-BMSCs), BM mononuclear cells (BMMNCs), and peripheral blood (PB) CD138+, CD138-, or PB CD14+ cells. Additionally, osteoclastogenesis from CD14+ cells is prevented by ralimetinib dimesylate (400 nM–800 nM)[2].
Enzyme inhibition: Ralimetinib dimysylate (LY2228820 dimysylate) potently inhibited recombinant human p38α kinase activity with an IC₅₀ of 0.3 nM and a Ki of 0.2 nM. It inhibited p38β/γ/δ by ≤2% at 0.1 μM and had no effect on ERK1/2 or JNK1/2 (≤1% inhibition at 1 μM), confirming high p38α selectivity [1, 3] - Antiproliferative activity: In multiple myeloma (MM) cell lines (RPMI 8226, U266), Ralimetinib dimysylate suppressed cell viability with IC₅₀ values of 0.02 μM (RPMI 8226) and 0.03 μM (U266) (72-hour MTT assay). When combined with bortezomib (0.001 μM), it reduced IC₅₀ to 0.008 μM (RPMI 8226) and 0.012 μM (U266), showing synergistic antiproliferative effects [2] - Anti-inflammatory activity: In LPS-stimulated RAW264.7 macrophages, Ralimetinib dimysylate (0.01–0.1 μM) reduced TNF-α secretion by 80–90% (ELISA), IL-6 secretion by 75–85% (ELISA), and iNOS protein expression by ~80% (Western blot) [1] - Signal pathway suppression: In TNF-α-stimulated RPMI 8226 cells, Ralimetinib dimysylate (0.01–0.05 μM) dose-dependently reduced p38α phosphorylation (p-p38α) by ≥95% and downstream MK2 phosphorylation (p-MK2) by ≥90% (Western blot) within 30 minutes. Total p38α and MK2 levels remained unchanged [3] - Apoptosis induction: In RPMI 8226 cells, Ralimetinib dimysylate (0.03 μM, 48 hours) increased apoptotic cell percentage from 2.1% (vehicle) to 40.5% (Annexin V/PI staining). Combination with bortezomib (0.001 μM) elevated apoptosis to 65.2% [2] |
| ln Vivo |
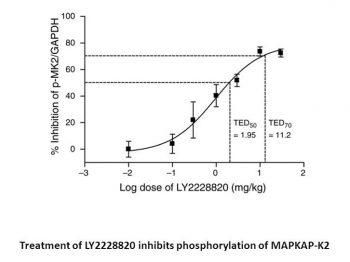
Ralimetinib dimesylate inhibits the production of TNFα in LPS-induced mice with a threshold minimum 50% effective dose (TMED50) of less than 1 mg/kg. Ralimetinib dimesylate exhibits strong effects on paw swelling, bone deterioration, and cartilage destruction in a rat model of collagen-induced arthritis (CIA), with a threshold minimum 50% effective dose (TMED50) of 1.5 mg/kg[1]. In mice implanted with B16-F10 melanoma, ralimetinib dimesylate inhibits tumor phospho-MK2 in a dose-dependent manner (TED50=1.95 mg/kg, TED70=11.17 mg/kg). Mouse in vivo TED50=1.01 mg/kg (compound exposure approximately 100 nM) and human ex vivo IC50=0.12 μM with either mouse or human PBMC[3] are the values for ralimetinib dimesylate's ability to inhibit MK2 phosphorylation.
Anti-inflammatory efficacy: C57BL/6 mice (male, 8-week-old) with LPS-induced acute inflammation were treated with Ralimetinib dimysylate (1 mg/kg, 3 mg/kg, oral gavage, once daily) for 3 days. The 3 mg/kg dose reduced serum TNF-α levels by ~85%, IL-6 levels by ~80%, and lung myeloperoxidase (MPO) activity (neutrophil infiltration marker) by ~75% vs vehicle [1] - Multiple myeloma xenograft efficacy: Nude mice (female, 6–8 weeks) bearing RPMI 8226 xenografts (100–120 mm³) were randomized into 4 groups (n=8/group): (1) vehicle (0.5% methylcellulose/0.1% Tween 80, oral, daily); (2) Ralimetinib dimysylate 5 mg/kg (oral, daily); (3) bortezomib 0.5 mg/kg (intravenous, twice weekly); (4) combination (Ralimetinib dimysylate 5 mg/kg + bortezomib 0.5 mg/kg). After 28 days, the combination group reduced tumor volume by 90% (vs 45% for Ralimetinib dimysylate monotherapy, 50% for bortezomib monotherapy) and extended median survival by 21 days [2] - Tumor signal suppression: In RPMI 8226 xenografts, Ralimetinib dimysylate (5 mg/kg, oral, daily) for 14 days reduced p-p38α and p-MK2 levels by ≥85% (IHC), and Ki-67 (proliferation marker) by ≥80% [3] |
| Enzyme Assay |
Inhibition of p38α is determined using recombinant human p38α in a standard filter binding protocol using ATP[γ-33P] and EGFR 21-mer peptide as substrate. Functional inhibition of TNFα in murine peritoneal macrophages is determined using LPS stimulation in the presence of LY2228820. To assess p38α activity in cells more directly, RAW 264.7 cells are treated with LY2228820 and then stimulated with anisomycin. The level of p38α activity is detected using a phosphoMAPKAPK-2 (pMK2) (Thr 334) antibody which reacts with a residue specifically phosphorylated by p38α.
p38α kinase activity assay (radiometric): Recombinant human p38α (activated by MKK6) was incubated in reaction buffer (25 mM Tris-HCl pH 7.5, 10 mM MgCl₂, 1 mM DTT, 0.01% BSA) with 0.2 mg/mL MBP (substrate), 10 μM ATP (including [γ-³²P]ATP), and serial dilutions of Ralimetinib dimysylate (0.00005–0.1 μM). Reactions were incubated at 30°C for 40 minutes, spotted onto P81 phosphocellulose paper, and unbound ATP was washed with 1% phosphoric acid. Radioactivity (³²P incorporation into MBP) was measured via scintillation counter, and IC₅₀ values were calculated [1] - p38α binding assay (SPR): Recombinant p38α was immobilized on a CM5 sensor chip. Serial dilutions of Ralimetinib dimysylate (0.0001–0.01 μM) were injected over the chip at 25°C in running buffer (10 mM HEPES pH 7.4, 150 mM NaCl, 0.005% Tween 20). Sensorgrams were recorded, and Ki was derived using a 1:1 binding model [3] |
| Cell Assay |
Recombinant human p38α is used in a standard filter binding protocol with ATP[γ-33P] and the EGFR 21-mer peptide as substrate to measure the inhibition of p38. Using LPS stimulation and Ralimetinib, the functional inhibition of TNFα in murine peritoneal macrophages is assessed. RAW 264.7 cells are given ralimetinib treatment before being stimulated with anisomycin in order to more accurately measure p38α activity in the cells. A phosphoMAPKAPK-2 (pMK2) (Thr 334) antibody that reacts with a residue that has been specifically phosphorylated by p38 is used to measure the level of p38α activity.
Cell viability assay (MTT): RPMI 8226/U266 cells (5×10³/well, 96-well plate) were incubated overnight, then treated with Ralimetinib dimysylate (0.001–0.1 μM) alone or with bortezomib (0.001 μM) for 72 hours at 37°C (5% CO₂). MTT reagent (5 mg/mL) was added (10 μL/well) for 4 hours; formazan crystals were dissolved with DMSO, and absorbance was measured at 570 nm. IC₅₀ values were calculated via nonlinear regression [2] - Western blot for p-p38α/MK2: RPMI 8226 cells (1×10⁶/well, 6-well plate) were serum-starved for 24 hours, pre-treated with Ralimetinib dimysylate (0.01–0.05 μM) for 1 hour, then stimulated with TNF-α (10 ng/mL) for 15 minutes. Cells were lysed in RIPA buffer (with protease/phosphatase inhibitors); lysates (20 μg protein) were run on SDS-PAGE, blotted with antibodies against p-p38α (Thr180/Tyr182), total p38α, p-MK2 (Thr334), and β-actin. Band intensity was quantified via densitometry [3] - Cytokine ELISA in macrophages: RAW264.7 cells (1×10⁵/well, 24-well plate) were pre-treated with Ralimetinib dimysylate (0.01–0.1 μM) for 1 hour, then stimulated with LPS (1 μg/mL) for 24 hours. Culture supernatants were collected, and TNF-α/IL-6 levels were measured via sandwich ELISA [1] - Apoptosis assay (Annexin V/PI): RPMI 8226 cells (2×10⁵/well, 6-well plate) were treated with Ralimetinib dimysylate (0.03 μM) alone or with bortezomib (0.001 μM) for 48 hours. Cells were harvested, washed with PBS, stained with Annexin V-FITC and PI, and analyzed by flow cytometry. Apoptotic cells (Annexin V⁺/PI⁻ + Annexin V⁺/PI⁺) were counted [2] |
| Animal Protocol |
Murine B16-F10 melanoma cells are cultured in Dulbecco's Modified Eagle Medium with l-glutamine, high glucose, and 10% FBS (GIBCO 11965-092). B16-F10 cells (2×106) are implanted into the rear flank of C57/bl6 mice, and when the tumors grow to a size of about 200 mm3, Ralimetinib dimesylate in 1% carboxymethylcellulose/0.25% Tween 80 is given orally. Tumors are removed, homogenized, and lysed two hours after the dose for Western blot analysis. Chemiluminescent detection is used to measure MK2 phosphorylation (p-Thr334), which is normalized to total glyceraldehyde-3-phosphate dehydrogenase. In xenograft models, the 50% or 70% threshold effective dose (TED50 and TED70, respectively) is calculated to approximate the effective dose ranges where significant target inhibition is seen. The TED50 or TED70 is the dose at which a statistically significant effect is obtained and there is at least 50% or 70% inhibition, in comparison to the vehicle control, respectively.
LPS inflammation model: Male C57BL/6 mice (n=6/group) were randomized into 3 groups: (1) vehicle (0.5% methylcellulose/0.1% Tween 80, oral gavage, daily); (2) Ralimetinib dimysylate 1 mg/kg (oral, daily); (3) Ralimetinib dimysylate 3 mg/kg (oral, daily). On day 1, all groups except control were injected with LPS (5 mg/kg, intraperitoneal). Treatments continued for 3 days; on day 4, mice were euthanized for serum cytokine (TNF-α, IL-6) and lung MPO activity analysis [1] - MM xenograft study: Female nude mice were subcutaneously injected with 5×10⁶ RPMI 8226 cells (suspended in 100 μL PBS/Matrigel, 1:1) into the right flank. When tumors reached 100–120 mm³, mice were randomized into 4 groups (n=8/group): (1) vehicle (oral, daily); (2) Ralimetinib dimysylate 5 mg/kg (oral, daily); (3) bortezomib 0.5 mg/kg (intravenous, days 1, 4, 7, 10, 13, 16, 19, 22); (4) combination (Ralimetinib dimysylate + bortezomib). Tumor volume was measured twice weekly (volume = length × width² × 0.5). Mice were monitored for survival, and tumors were harvested for IHC [2] - Pharmacokinetic (PK) study: Male CD-1 mice (n=3/time point) received Ralimetinib dimysylate via oral gavage (10 mg/kg, vehicle) or intravenous injection (2 mg/kg, 5% DMSO/95% saline). Blood samples (50 μL) were collected at 0.25, 0.5, 1, 2, 4, 6, 8, 12 hours post-dose. Plasma concentrations were measured via LC-MS/MS; PK parameters were calculated via non-compartmental analysis [3] |
| ADME/Pharmacokinetics |
Oral bioavailability: In CD-1 mice, the oral bioavailability of lelitinib dimethylsulfonate was approximately 52% (oral AUC₀₋∞ = 22.8 μg·h/mL; intravenous AUC₀₋∞ = 43.8 μg·h/mL) [3]
- Plasma pharmacokinetics: After oral administration (10 mg/kg), Cmax was 4.2 μg/mL (Tmax = 1.0 h) and terminal T₁/₂ = 3.5 h. Following intravenous injection (2 mg/kg), Cmax = 11.5 μg/mL, T₁/₂ = 3.0 h [3] - Tissue distribution: In RPMI 8226 xenograft mice, the tumor/plasma ratio of lenitinib dimethylsulfonate (orally 10 mg/kg) was 4.1 (2 h after administration), with higher liver distribution (liver/plasma ratio = 3.2) and lower brain permeability (brain/plasma ratio = 0.15) [3] - Metabolism: In human liver microsomes, lenitinib dimethylsulfonate is primarily metabolized by CYP3A4 (≥70% of total metabolism) and CYP2D6 (approximately 20%). Co-incubation with a CYP3A4 inhibitor (ketoconazole) reduces metabolism by approximately 80% [3] |
| Toxicity/Toxicokinetics |
Plasma protein binding: The plasma protein binding of lelitinib dimethylsulfonate in human plasma is approximately 98% (as determined by balanced dialysis) [3]
- Acute toxicity: In CD-1 mice, a single oral dose up to 300 mg/kg did not cause death or clinical symptoms (e.g., somnolence, weight loss). Serum ALT, AST, BUN and creatinine were within the normal range 24 hours after administration [3] - Chronic toxicity: A 28-day repeated-dose study in rats (5-25 mg/kg, orally, once daily) showed no significant organ toxicity (liver, kidney, spleen) at doses ≤20 mg/kg. At a dose of 25 mg/kg, mild hepatic steatosis was observed in 2 out of 6 rats [3] - Normal cytotoxicity: In primary human bone marrow stromal cells (HS-5), cell viability was >90% after 72 hours of treatment with lelimemetinib dimethylsulfonate (0.01–0.1 μM), indicating low toxicity to normal hematopoietic cells [2] |
| References |
|
| Additional Infomation |
Ralimetinib mesylate is the dimethylsulfonate form of LY2228820, a trisubstituted imidazole derivative available orally. It is a p38 mitogen-activated protein kinase (MAPK) inhibitor with potential anti-inflammatory and antitumor activities. After administration, lelimetinib inhibits p38 activity, particularly the α and β isoforms, thereby inhibiting MAPKAPK2 phosphorylation and blocking p38 MAPK-mediated signaling. This may inhibit the production of various cytokines involved in inflammation, cell proliferation, and angiogenesis, such as tumor necrosis factor-α (TNFα), interleukins (IL)-1, -6, and -8, vascular endothelial growth factor, and macrophage inflammatory protein-1α. Ultimately, this can induce apoptosis and reduce tumor cell proliferation. Furthermore, the inhibitory effect of LY2228820 on the p38 MAPK pathway may enhance the antitumor activity of certain chemotherapeutic agents. p38 MAPK is a serine/threonine protein kinase that is often highly expressed in cancer cells and plays a key role in tumor cell proliferation, angiogenesis, and metastasis. Mechanism of action: Lelitinib dimethylsulfonate is a reversible, ATP-competitive p38α inhibitor. It binds to the ATP-binding pocket of p38α and forms hydrogen bonds with Glu71 (hinge region) and Asp168 (catalytic ring) residues, thereby blocking ATP coordination and kinase activation [1, 3]. Clinical development: This compound has entered Phase II clinical trials for rheumatoid arthritis (RA) and multiple myeloma (MM). This compound showed promising efficacy when used in combination with bortezomib for the treatment of multiple myeloma, but Phase III clinical trial data have not yet been published [2, 3]. Research applications: This compound is being used as a tool compound to study the p38α-mediated pathway in inflammation and hematologic malignancies, particularly to evaluate the efficacy of combined p38α inhibitors with proteasome inhibitors [2, 3].
|
| Molecular Formula |
C24H29FN6.2CH4O3S
|
|---|---|
| Molecular Weight |
612.74
|
| Exact Mass |
612.219
|
| Elemental Analysis |
C, 50.97; H, 6.09; F, 3.10; N, 13.72; O, 15.67; S, 10.46
|
| CAS # |
862507-23-1
|
| Related CAS # |
Ralimetinib;862505-00-8
|
| PubChem CID |
11570805
|
| Appearance |
White to yellow solid powder
|
| Boiling Point |
634.4ºC at 760 mmHg
|
| Flash Point |
337.5ºC
|
| LogP |
6.653
|
| Hydrogen Bond Donor Count |
4
|
| Hydrogen Bond Acceptor Count |
11
|
| Rotatable Bond Count |
5
|
| Heavy Atom Count |
41
|
| Complexity |
701
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
O=S(C)(O)=O.FC1C=CC(C2=C(C3C=CC4N=C(N(C=4N=3)CC(C)(C)C)N)NC(C(C)(C)C)=N2)=CC=1
|
| InChi Key |
NARMJPIBAXVUIE-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C24H29FN6.2CH4O3S/c1-23(2,3)13-31-20-17(28-22(31)26)12-11-16(27-20)19-18(14-7-9-15(25)10-8-14)29-21(30-19)24(4,5)6;2*1-5(2,3)4/h7-12H,13H2,1-6H3,(H2,26,28)(H,29,30);2*1H3,(H,2,3,4)
|
| Chemical Name |
5-[2-tert-butyl-4-(4-fluorophenyl)-1H-imidazol-5-yl]-3-(2,2-dimethylpropyl)imidazo[4,5-b]pyridin-2-amine;methanesulfonic acid
|
| Synonyms |
LY-2228820; LY 2228820; Ralimetinib; LY2228820; LY2228820 dimesylate; Ralimetinib dimesylate
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.08 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.08 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (4.08 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: Saline: 30mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6320 mL | 8.1601 mL | 16.3201 mL | |
| 5 mM | 0.3264 mL | 1.6320 mL | 3.2640 mL | |
| 10 mM | 0.1632 mL | 0.8160 mL | 1.6320 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
|
 |