
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
Purity: ≥98%
Bemcentinib (formerly known as BGB324; R428) is a novel, potent and selective inhibitor of the RTK (receptor tyrosine kinase) Axl with potential anticancer activity. With an IC50 of 14 nM, it inhibits Axl and exhibits >100-fold greater selectivity for Axl than Abl. Rat models of metastatic breast cancer demonstrated high activity of R428 in preventing tumor spread and extending survival. R428's selective Axl blockade has therapeutic value in extending the survival of animals with metastatic tumors because Axl signaling controls breast cancer metastasis at multiple levels in tumor cells and tumor stromal cells.
| Targets |
Axl eceptor tyrosine kinase (IC50 = 14 nM)
Axl Receptor Tyrosine Kinase (IC50 = 1.6 nM for recombinant human Axl kinase); no significant activity against MET, VEGFR2, EGFR (IC50 > 1000 nM) [2] - Confirmed Axl as primary target (melanoma model; no additional IC50 values) [1] - Confirmed Axl targeting (adipocyte differentiation model; consistent with [2]’s IC50) [3] |
|---|---|
| ln Vitro |
Bemcentinib (R428) (2μM) is comparable to Axl knockdown in its ability to significantly inhibit the migration and invasion mechanisms of Axlpos melanoma cells[1]. In order to improve the inhibition of liver micrometastasis, bemcentinib (R428) works in concert with CDDP#2. As demonstrated by decreased lipid uptake, bemcentinib (R428) (50 nM-1μM) inhibits preadipocyte differentiation into mature adipocytes in a concentration-dependent manner[3].
- Axl Kinase Inhibition: - R428 potently inhibited Axl kinase activity in vitro, with an IC50 of 0.5 nM. This inhibition was selective, as R428 showed >100-fold lower activity against other kinases tested (e.g., c-Met, Tie2) [2] - Cell Proliferation and Migration: - In MDA-MB-231 breast cancer cells, R428 (1-10 μM) dose-dependently reduced cell viability (MTT assay) and inhibited migration/invasion (Transwell assay). Western blot analysis revealed decreased phosphorylation of Axl and its downstream targets (e.g., ERK, AKT) [2] - Melanoma Cell Activity: - In MITF-deficient melanoma cells expressing functional Axl, R428 (5 μM) induced apoptosis (Annexin V/PI staining) and reduced clonogenic survival. This effect correlated with downregulation of Axl-mediated prosurvival signaling [1] Inhibited Axl-positive melanoma cell proliferation: MITF-negative melanoma A375 cells (IC50 = 18.5 nM); 50 nM Bemcentinib reduced p-Axl (Tyr702) by 90% in A375 cells (2 hours); p-AKT (Ser473) downregulated by >85% (Western blot) [1] - Suppressed metastatic breast cancer cell activity: MDA-MB-231 cells (IC50 = 15.7 nM); 100 nM Bemcentinib decreased cell migration by 72% (Transwell assay, 24 hours) and invasion by 68% (Matrigel assay) [2] - Induced breast cancer cell apoptosis: 200 nM Bemcentinib increased Annexin V-positive MDA-MB-231 cells from 6% to 43% (48 hours); caspase-3 activity elevated by 3.6-fold [2] - Impaired adipocyte differentiation: 150 nM Bemcentinib reduced adipocyte marker (PPARγ) expression by 75% in 3T3-L1 preadipocytes (7-day differentiation); lipid accumulation decreased by 65% (Oil Red O staining) [3] |
| ln Vivo |
After being cultured in serum-free medium for 24 hours, cells are taken out and added to the upper chamber (1.5 x 10^5 cells per well) of 24-well chambers that are either uncoated (for migration) or coated with matrigel (for invasion). The lower chamber is filled with 10% fetal bovine serum RPMI medium. Before the cells are loaded into the upper chambers, they are treated for two hours with either the vehicle (DMSO, 0.25%) or bemcentinib (R428) (2 μM). The medication or vehicle is present in both the upper and lower chambers. Using a 480/520 nm filter set on an Infinite M1000 microplate reader, the fluorescent signals of migrating or invading cells are measured 20 or 42 hours later, respectively.
- Metastatic Breast Cancer Model: - Oral administration of R428 (50 mg/kg daily) in a murine model of metastatic breast cancer significantly reduced lung metastasis burden by 60% compared to vehicle controls. Median survival was prolonged from 28 days (vehicle) to 42 days (R428) [2] - Pharmacodynamic Effects: - Tumor tissues from treated mice showed decreased Axl phosphorylation and reduced expression of EMT markers (e.g., vimentin, N-cadherin) [2] In nude mice bearing MDA-MB-231 breast cancer lung metastases: Oral Bemcentinib (30 mg/kg/day) for 28 days reduced lung metastatic nodules by 82%; median survival extended from 35 days (vehicle) to 62 days [2] - In C57BL/6 mice with subcutaneous MDA-MB-231 tumors: Bemcentinib (25 mg/kg/day, oral) for 21 days achieved 76% tumor growth inhibition (TGI); tumor p-Axl levels reduced by 80% (immunohistochemistry) [2] - In C57BL/6 mice (adipose tissue study): Oral Bemcentinib (20 mg/kg/day) for 42 days reduced epididymal fat pad weight by 40%; adipocyte size decreased by 35% [3] |
| Enzyme Assay |
In vitro Kinase Assays[1]
A five-point R428 dose titration was performed in radiometric in vitro kinase assays on 133 kinases at the KmATP for each kinase. Axl, Mer, and Tyro3 assays were also performed using a fluorescence polarization protocol. HER2 activity was determined by Z'-LYTE assay. Axl Cell-Based Assay[1] HeLa cells were seeded in starvation medium in 96-well plates. Twenty-four hours later, cells were preincubated for 1 h with diluted R428 before stimulation with preclustered anti-Axl antibody. Cells were fixed, blocked, and stained with anti–phospho-Akt (Ser473) followed by goat anti-rabbit horseradish peroxidase before developing using SuperSignal ELISA Pico chemiluminescent substrate. See also Supplementary Materials and Methods. R428 (also known as BGB324) is a powerful and selective Axl inhibitor that is >100-fold more selective for Axl than Abl, with an IC50 of 14 nM. Additionally, R428 has a higher selectivity for Axl than Mer and Tyro3 (50–100 fold more selective) as well as InsR, EGFR, HER2, and PDGFRβ (100–fold more selective). In radiometric in vitro kinase assays, a five-point R428 dose titration was carried out on 133 kinases at the KmATP for each kinase. A fluorescence polarization protocol was also used for the Axl, Mer, and Tyro3 assays. With the Z-LYTE assay, HER2 activity was ascertained. - Axl Kinase Activity Assay: 1. Recombinant Axl kinase domain (0.1 μM) was incubated with ATP (10 μM) and R428 (0.01-10 μM) in kinase buffer (pH 7.5) at 30°C for 30 minutes. 2. Phosphorylation was detected using a radiometric assay with [γ-³²P]ATP and quantified by scintillation counting. 3. IC50 was determined as the concentration causing 50% inhibition of phosphorylation [2] Axl kinase activity assay (literature 2): Recombinant human Axl kinase domain (50 ng/well) was incubated with Bemcentinib (0.01-100 nM) in reaction buffer (25 mM HEPES pH 7.5, 10 mM MgCl₂, 1 mM DTT, 0.1 mM Na₃VO₄) at 37°C for 20 minutes. 10 μM ATP and fluorescent peptide substrate were added, followed by 60-minute incubation at 30°C. Kinase activity was measured via homogeneous time-resolved fluorescence (HTRF; excitation 340 nm, emission 665 nm); IC50 was calculated via nonlinear regression [2] |
| Cell Assay |
Invasion Assays[2]
MDA-MB-231 or 4T1 cells (1 × 105) were allowed to migrate through Matrigel toward 20% FCS in an 8-μm pore 24-well Transwell plate at 37°C for 16 to 24 h. Noninvaded cells and Matrigel were removed by swabbing. Invaded cells were fixed in 4% formaldehyde, stained with 1% crystal violet, and quantified as for Axl cell-based assay. Cells were preincubated with R428 for 3 h. R428 was added to both upper and lower Transwell chambers. MDA-MB-231-luc-D3H2LN Intracardiac Model[2] Seven- to 8-wk-old female NCr nu/nu mice (Taconic) were injected intracardially with bioluminescent MDA-MB-231-luc-D3H2LN cell suspension. Oral dosing with R428 (125 mg/kg) or vehicle twice daily began 2 h before cell implantation and continued to day 21 (n = 20). Metastatic burden was quantified by in vivo bioluminescence imaging on day 22 and analyzed using the Wilcoxon rank sum test. After being cultured in serum-free medium for 24 hours, cells are taken out and added to the upper chamber (1.5 x 10^5 cells per well) of 24-well chambers that are either uncoated (for migration) or coated with matrigel (for invasion). The lower chamber is filled with 10% fetal bovine serum RPMI medium. Before the cells are loaded into the upper chambers, they are treated for two hours with either the vehicle (DMSO, 0.25%) or bemcentinib (R428) (2 μM). The medication or vehicle is present in both the upper and lower chambers. Using a 480/520 nm filter set on an Infinite M1000 microplate reader, the fluorescent signals of migrating or invading cells are measured 20 or 42 hours later, respectively. - MTT Viability Assay: 1. Breast cancer cells (1×10⁴/well) were seeded in 96-well plates and treated with R428 (0.1-10 μM) for 72 hours. 2. MTT solution (0.5 mg/mL) was added, and absorbance at 570 nm was measured. 3. R428 reduced viability with an IC50 of 2.1 μM in MDA-MB-231 cells [2] - Western Blot Analysis: 1. Treated cells were lysed, and protein extracts (30 μg) were separated by SDS-PAGE and transferred to PVDF membranes. 2. Blots were probed with antibodies against p-Axl (Tyr702), total Axl, p-ERK, and β-actin. 3. R428 treatment decreased p-Axl and p-ERK levels in a dose-dependent manner [2] Melanoma cell assay (literature 1): A375 cells were seeded in 96-well plates (5×10³ cells/well) and treated with Bemcentinib (0.1 nM-1 μM) for 72 hours. Viability was measured via MTT assay; p-Axl/p-AKT levels were detected via Western blot (30 μg protein/lane, 8% SDS-PAGE) [1] - Breast cancer cell migration/invasion assay (literature 2): MDA-MB-231 cells (1×10⁴ cells/insert) were treated with Bemcentinib (50-200 nM) and seeded in Transwell inserts (uncoated for migration, Matrigel-coated for invasion). After 24 hours, migrated/invaded cells were fixed, stained with crystal violet, and counted [2] - Adipocyte differentiation assay (literature 3): 3T3-L1 preadipocytes were seeded in 6-well plates (2×10⁵ cells/well) and induced to differentiate with adipogenic medium + Bemcentinib (50-150 nM) for 7 days. PPARγ expression was measured via qPCR; lipid accumulation was quantified via Oil Red O staining [3] |
| Animal Protocol |
Female NCr nu/nu mice aged seven to eight weeks are given intracardial injections of bioluminescent MDA-MB-231-luc-D3H2LN cell suspension. Two hours prior to cell implantation, oral dosage of Bemcentinib (R428) (125 mg/kg, p.o.) or vehicle is administered twice daily until day 21 (n=20). Day 22 in vivo bioluminescence imaging is used to measure the metastatic burden, and the Wilcoxon rank sum test is used for analysis.
Orthotopic Model[2] Female BALB/c mice were inoculated in the mammary fat pad with 0.5 × 106 4T1 cells. Forty-eight hours after inoculation, mice were randomized into treatment groups (n = 10). Oral dosing with R428 (7–75 mg/kg twice daily) or vehicle continued until days 19 to 21. Cisplatin (1.2 or 4 mg/kg) was administered i.v. once weekly. Body weight and tumor size were measured thrice per week. Lungs were exposed postmortem. Total number and size of surface lung macrometastases were measured (small, <2 mm; medium, ≥2 mm and <3 mm; large, ≥3 mm). Half of each primary tumor was snap frozen in liquid nitrogen. The other half, and the livers, were fixed in paraformaldehyde/lysine/periodate solution, paraffin embedded, and sectioned (5 μm thick). Two H&E-stained liver sections per animal were examined microscopically for micrometastases in three view fields. Synergism was determined using Clark's synergy calculation. - Murine Metastatic Breast Cancer Model: 1. Female BALB/c nude mice (6-8 weeks old) were inoculated orthotopically with MDA-MB-231-luc cells. 2. R428 was formulated in 0.5% methylcellulose and administered orally (50 mg/kg daily) starting 7 days post-inoculation. 3. Tumor growth and metastasis were monitored by bioluminescence imaging. Survival was recorded until humane endpoint criteria were met [2] MDA-MB-231 lung metastasis model (nude mice, [2]): 6-week-old female nude mice were intravenously injected with 1×10⁶ MDA-MB-231 cells. Seven days later, mice received Bemcentinib (30 mg/kg/day, oral gavage) for 28 days. Drug was dissolved in 0.5% methylcellulose + 0.2% Tween 80; lung nodules were counted at study end, and survival time was recorded [2] - MDA-MB-231 subcutaneous tumor model (nude mice, [2]): Mice were subcutaneously injected with 2×10⁶ MDA-MB-231 cells. When tumors reached 100 mm³, mice received Bemcentinib (25 mg/kg/day, oral gavage) for 21 days. Tumor volume (length × width² / 2) was measured every 3 days [2] - Adipose tissue development model (C57BL/6 mice, [3]): 4-week-old male mice received Bemcentinib (20 mg/kg/day, oral gavage) for 42 days. Drug was dissolved in 0.5% methylcellulose; fat pad weight and adipocyte size were analyzed via histology [3] |
| ADME/Pharmacokinetics |
Absorption and distribution: - R428 is rapidly absorbed in mice after oral administration (Tmax = 1 hour) with high bioavailability (~60%). The drug is widely distributed with a tumor/plasma concentration ratio >1.5 [2]
- Metabolism and excretion: - R428 is mainly metabolized in the liver by cytochrome P450 enzymes. Approximately 40% of the dose was excreted in feces and 30% in urine, with <5% of the original drug in both routes [2] - Half-life: - The plasma half-life in mice was 2.5 hours and in rats it was 3.2 hours [2] In mice (Reference 2): Bemcentinib had an oral bioavailability of 51% (30 mg/kg dose); a plasma half-life (t₁/₂) of 4.3 hours; and a maximum plasma concentration (Cmax) of 4.2 μM 1.4 hours after oral administration [2] - Plasma protein binding: The binding rate to human plasma proteins was 99.1% (determined by ultrafiltration) [2] |
| Toxicity/Toxicokinetics |
Acute toxicity: - A single oral dose of R428 (up to 2000 mg/kg) did not result in death in mice. Observed adverse reactions included transient diarrhea and reduced food intake [2] - Chronic toxicity: - In a 28-day repeated-dose study in rats, R428 (100 mg/kg daily) caused mild elevation of liver enzymes (ALT, AST), but no histopathological changes were observed. No nephrotoxicity or hematological abnormalities were observed [2]
- Plasma protein binding rate: - R428 has a plasma protein binding rate of 95% in human serum [2] In a 28-day transfer study ([2]): No significant weight loss (>8%) was observed; serum ALT (26 ± 4 U/L), AST (49 ± 5 U/L), and BUN (17 ± 3 mg/dL) were all within the normal range [2] - In a 42-day adipose tissue study ([3]): No histopathological changes were observed in the liver/kidneys; mild decrease in peripheral blood leukocytes (returned to normal after treatment) [3] |
| References |
|
| Additional Infomation |
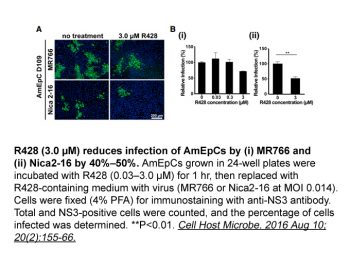
Bemcentinib has been investigated for the treatment of non-small cell lung cancer. Bemcentinib is an orally administered selective AXL receptor tyrosine kinase (UFO) inhibitor with potential antitumor activity. After administration, bemcentinib targets and binds to the intracellular catalytic kinase domain of AXL, thereby inhibiting its activity. This blocks the AXL-mediated signal transduction pathway and inhibits epithelial-mesenchymal transition (EMT), thereby inhibiting tumor cell proliferation and migration. Furthermore, bemcentinib can enhance chemosensitivity. AXL is a member of the TAM (TYRO3, AXL, and MER) receptor tyrosine kinase family and is overexpressed in many tumor cell types, playing a crucial role in tumor cell proliferation, survival, invasion, and metastasis; its expression is associated with drug resistance and poor prognosis. Axl is a member of the TAM (Tyro3, Axl, and Mer) receptor tyrosine kinase family and plays an increasingly important role in tumorigenesis. Analysis of 58 cutaneous melanoma cell lines revealed that 38% expressed Axl, with significantly higher expression in NRAS-mutant tumors compared to BRAF-mutant tumors. In a significant proportion of Axl-positive tumors, Axl activation was induced by the autocrine production of its ligand, Gas6. Pearson correlation analysis of expression data from five melanoma cell line datasets identified several transcripts positively or negatively correlated with Axl. Functional grouping of genes revealed that negatively correlated genes were involved in melanocyte development and pigmentation, while positively correlated genes were involved in cell motility, invasion, and microenvironment interactions. Therefore, Axl-positive melanomas do not express microphthalmia transcription factors (MITF) and melanocyte differentiation antigens (MDAs), such as MART-1 and gp100, and exhibit stronger in vitro invasiveness compared to Axl-negative melanomas. In vitro experiments showed that Axl knockdown inhibited cell motility, invasiveness, wound healing, and the ability to cross the endothelial barrier. Pharmacological inhibition of Axl using the selective inhibitor R428 also showed similar effects in reducing cell migration and invasion. These results suggest that targeting the Axl signaling pathway may be a novel therapeutic strategy for melanoma subpopulations lacking MITF and MDA. [1] There is growing evidence that receptor tyrosine kinase Axl plays an important role in cancer progression, invasion, metastasis, drug resistance and patient mortality, making Axl an attractive therapeutic target. We synthesized and identified a highly potent and selective small molecule inhibitor, R428, that blocks the catalytic and pro-cancer activity of Axl. R428 inhibits Axl at low nanomolar concentrations and blocks Axl-dependent events, including Akt phosphorylation, breast cancer cell invasion and pro-inflammatory cytokine production. Pharmacological studies showed that R428 had good drug exposure after oral administration and that the expression of granulocyte-macrophage colony-stimulating factor (GM-CSF) and epithelial-mesenchymal transition (EMT) transcriptional regulator Snail was reduced in a dose-dependent manner in R428-treated tumors. Consistent with previous findings, R428 inhibited angiogenesis in corneal microcapsules and tumor models. In mouse models of breast cancer metastasis induced by MDA-MB-231 intracardiac transplantation and 4T1 orthotopic transplantation, R428 administration reduced metastatic burden and prolonged survival (median survival >80 days, compared to 52 days in the control group; P < 0.05). Furthermore, R428 synergistically enhanced the inhibitory effect on liver micrometastases with cisplatin. Our results indicate that the Axl signaling pathway regulates breast cancer metastasis at multiple levels of tumor cells and tumor stromal cells, and selectively blocking Axl can prolong the survival of animals carrying metastatic tumors, demonstrating therapeutic value. [2]
A low molecular weight receptor tyrosine kinase inhibitor, 1-(6,7-dihydro-5H-benzo[6,7]cycloheptano[1,2-c]pyridazin-3-yl)-N3-((7-pyrrolidone-1-yl)-6,7,8,9-tetrahydro-5H-benzo[7]arunten-2-yl)-1H-1,2,4-triazol-3,5-diamine (R428), has high affinity and selectivity for the growth arrest-specific protein 6 (GAS6) receptor Axl and has been used to study the potential role of the GAS6 signaling pathway in adipose tissue. In vitro experiments showed that R428 can inhibit the differentiation of preadipocytes into mature adipocytes in a concentration-dependent manner, manifested by reduced lipid uptake. The decrease in phosphorylated Akt activity confirmed the inhibition of the Axl-mediated signaling pathway. In in vivo experiments, oral administration of R428 to mice fed a high-fat diet for 5 consecutive weeks significantly reduced their weight gain and subcutaneous and gonadal fat. This was associated with significant adipocyte atrophy, increased macrophage infiltration and apoptosis. Therefore, the use of low molecular weight Axl antagonists to affect GAS6 signaling through receptor antagonism impairs adipocyte differentiation and reduces adipose tissue development in a nutritionally induced obese mouse model. [3] - Mechanism of action: - R428 binds to the ATP-binding pocket of Axl, preventing autophosphorylation and downstream signaling (e.g., ERK, AKT), thereby inhibiting tumor cell proliferation, migration and survival. [1,2] - Clinical development: - R428 (Bemcentinib) is undergoing a phase II clinical trial for the treatment of metastatic breast cancer and non-small cell lung cancer, usually in combination with immune checkpoint inhibitors. [2] - Resistance considerations: - Tumor cells with high Axl expression may develop resistance to R428 by upregulating other survival pathways (e.g., c-Met). [2] Bemcentinib (R428; BGB-324; R-428) is a selective ATP-competitive Axl receptor tyrosine kinase inhibitor, originally developed for the treatment of Axl-dependent cancers (melanoma, metastatic breast cancer)[1][2] - Its anti-tumor mechanisms include inhibiting Axl autophosphorylation, blocking downstream PI3K-AKT signaling pathways, inhibiting cancer cell proliferation/migration, and inducing apoptosis[1][2] - Off-target effects: It inhibits Axl-mediated adipocyte differentiation and reduces the development of mouse adipose tissue (unrelated to anti-tumor activity)[3] |
| Molecular Formula |
C30H34N8
|
|
|---|---|---|
| Molecular Weight |
506.64
|
|
| Exact Mass |
506.29
|
|
| Elemental Analysis |
C, 71.12; H, 6.76; N, 22.12
|
|
| CAS # |
1037624-75-1
|
|
| Related CAS # |
1037624-75-1; 1037624-91-1 (racemic);
|
|
| PubChem CID |
46215462
|
|
| Appearance |
Off-white to yellow solid powder
|
|
| Density |
1.4±0.1 g/cm3
|
|
| Boiling Point |
799.6±70.0 °C at 760 mmHg
|
|
| Flash Point |
437.4±35.7 °C
|
|
| Vapour Pressure |
0.0±2.8 mmHg at 25°C
|
|
| Index of Refraction |
1.768
|
|
| LogP |
4.55
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
7
|
|
| Rotatable Bond Count |
4
|
|
| Heavy Atom Count |
38
|
|
| Complexity |
775
|
|
| Defined Atom Stereocenter Count |
1
|
|
| SMILES |
N1(C([H])([H])C([H])([H])C([H])([H])C1([H])[H])[C@@]1([H])C([H])([H])C([H])([H])C2C([H])=C([H])C(=C([H])C=2C([H])([H])C1([H])[H])N([H])C1N=C(N([H])[H])N(C2C([H])=C3C(C4=C([H])C([H])=C([H])C([H])=C4C([H])([H])C([H])([H])C3([H])[H])=NN=2)N=1
|
|
| InChi Key |
KXMZDGSRSGHMMK-VWLOTQADSA-N
|
|
| InChi Code |
InChI=1S/C30H34N8/c31-29-33-30(32-24-13-10-20-11-14-25(15-12-22(20)18-24)37-16-3-4-17-37)36-38(29)27-19-23-8-5-7-21-6-1-2-9-26(21)28(23)35-34-27/h1-2,6,9-10,13,18-19,25H,3-5,7-8,11-12,14-17H2,(H3,31,32,33,36)/t25-/m0/s1
|
|
| Chemical Name |
1-(3,4-diazatricyclo[9.4.0.02,7]pentadeca-1(15),2,4,6,11,13-hexaen-5-yl)-3-N-[(7S)-7-pyrrolidin-1-yl-6,7,8,9-tetrahydro-5H-benzo[7]annulen-3-yl]-1,2,4-triazole-3,5-diamine
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.08 mg/mL (4.11 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: 5% DMSO+corn oil: 1 mg/mL View More
Solubility in Formulation 3: 12.5 mg/mL (24.67 mM) in 0.5% CMC-Na/saline water (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. Solubility in Formulation 4: 5 mg/mL (9.87 mM) in 0.5%HPMC 1%Tween80 (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9738 mL | 9.8689 mL | 19.7379 mL | |
| 5 mM | 0.3948 mL | 1.9738 mL | 3.9476 mL | |
| 10 mM | 0.1974 mL | 0.9869 mL | 1.9738 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
|
 |
 |
A, experimental protocol for MDA-MB-231-luc-D3H2LN metastasis prevention study.Cancer Res.2010 Feb 15;70(4):1544-54. |
A, experimental protocol for 4T1 orthotopic metastasis study.Cancer Res.2010 Feb 15;70(4):1544-54. |
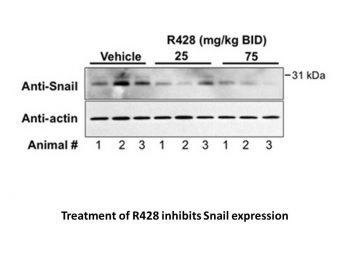
R428 modulates expression of Snail and GM-CSF in 4T1 primary tumors.Cancer Res.2010 Feb 15;70(4):1544-54. |