
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
(R)-baclofen (formerly STX-209; STX209; STX 209), a derivative of gamma-aminobutyric acid, is a selective GABAB receptor agonist that has been primarily used to treat spasticity. Baclofen is a synthetic chlorophenyl-butanoic acid derivative used to treat spasms due to spinal cord damage and multiple sclerosis, muscle-relaxing. It acts at spinal and supraspinal sites, reducing excitatory transmission. GABAB receptors are metabotropic receptors which produce slow inhibitory signals.
| Targets |
GABAB receptors (Ki = 1.2 μM for rat brain membrane preparations) [2]
- GABAA receptors (weak affinity, Ki > 100 μM for rat brain membrane preparations) [2] |
|---|---|
| ln Vitro |
In vitro activity: Baclofen is a skeletal muscle relaxant used in the treatment of spasticity of spinal and cerebral origin. Although it is marketed as a racemic mixture, only the baclofen R-enantiomer is stereospecifically active at so called GAGAB-receptors. Researchers believe that the enantiomers of baclofen have different properties. It is claimed that the R-(−)-enantiomer is about 100 times more active than the S-(+)-enantiomer.
(R)-baclofen showed high affinity for GABAB receptors in rat brain membrane preparations, with a Ki value of 1.2 μM, while its affinity for GABAA receptors was negligible (Ki > 100 μM) [2] - (R)-baclofen did not affect GABA uptake in rat brain membrane preparations at concentrations up to 100 μM, indicating no interaction with GABA transporters [2] |
| ln Vivo |
At an effective dose of 1 mg/kg, (R)-Baclofen (STX209) reduces audiogenic seizures in Fmr1-knockout mice on a seizure-resistant C57BL/6 background [5]. With a minimum effective dosage (MED) of 1.5 mg/kg, acute intraperitoneal administration of (R)-Baclofen (STX209) considerably decreased the percentage of mice having seizures (seizure incidence) [5].
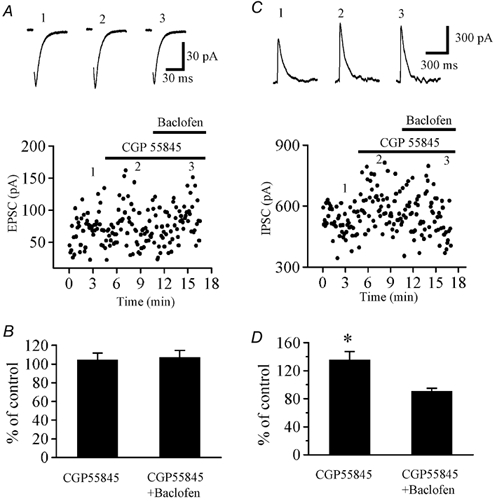
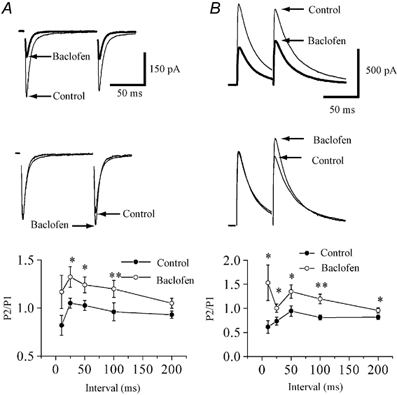
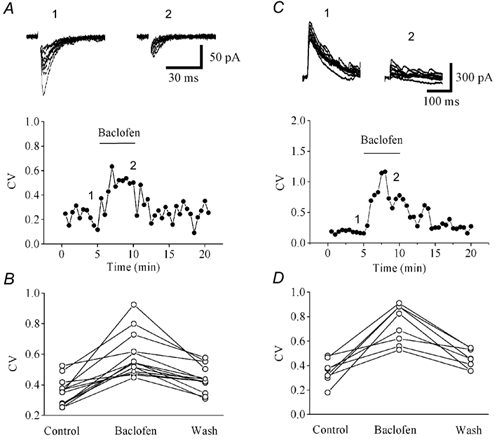
Intrathecal administration of (R)-baclofen (1–10 μg/rat) in rats induced dose-dependent cardiovascular responses, including a decrease in mean arterial blood pressure (maximal reduction of 28.3% ± 3.2% at 10 μg) and a slowing of heart rate (maximal reduction of 19.6% ± 2.7% at 10 μg). These effects were completely antagonized by the GABAB receptor antagonist phaclofen (20 μg/rat, intrathecal) [1] - In spastic multiple sclerosis patients, intravenous administration of (R)-baclofen (50 μg) significantly inhibited transmission in spinal pathways. The amplitude of motor evoked potentials (MEPs) recorded from the abductor pollicis brevis muscle was reduced by 35.7% ± 4.8% compared to baseline, and the central motor conduction time was prolonged by 12.3% ± 2.1% [4] |
| Enzyme Assay |
Rat brain membrane preparations were prepared by homogenizing brain tissues in ice-cold buffer, followed by centrifugation to obtain crude membrane fractions. Membranes were incubated with tritiated GABA (for GABAB receptor binding) or tritiated muscimol (for GABAA receptor binding) in the presence of increasing concentrations of (R)-baclofen (0.1–100 μM) at 4°C for 60 minutes. Unbound ligands were removed by rapid filtration through glass fiber filters, and the radioactivity of bound ligands was measured by liquid scintillation counting to calculate Ki values [2]
- For GABA uptake assays, rat brain membrane preparations were incubated with tritiated GABA and (R)-baclofen (0.1–100 μM) at 37°C for 10 minutes. The reaction was terminated by adding ice-cold buffer, and the mixture was filtered. Radioactivity of the filtered membranes was counted to assess the effect of (R)-baclofen on GABA uptake [2] |
| Animal Protocol |
1, 1.5 mg/kg; i.p.
Mice Male Wistar rats (250–300 g) were anesthetized and implanted with an intrathecal catheter targeting the lumbar spinal cord. After a 3-day recovery period, (R)-baclofen was dissolved in sterile saline and administered intrathecally at doses of 1, 3, 10 μg/rat. Phaclofen was administered intrathecally 10 minutes before (R)-baclofen in antagonism experiments. Mean arterial blood pressure and heart rate were recorded continuously for 60 minutes after drug administration using a femoral artery catheter connected to a pressure transducer [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Unlike baclofen, abaclofen absorption is not limited to the upper small intestine. Abaclofen can also be absorbed in the lower small intestine and colon, which makes the development of sustained-release formulations possible. Over 80% of R-baclofen is excreted unchanged via the kidneys. One study showed that the half-life of blood clearance after intravenous injection of R-baclofen in rats, monkeys, and dogs was 1.6–3.4 hours. The reported total blood clearance was 0.51 ± 0.13 L/h/kg in rats, 0.31 ± 0.11 L/h/kg in monkeys, and 0.24 ± 0.01 L/h/kg in dogs. (2) |
| References |
|
| Additional Infomation |
Abaclofen is an oxygen- and nitrogen-containing organic compound whose function is related to γ-amino acids. Abaclofen, or STX209, is the R-enantiomer of baclofen. It is considered a selective γ-aminobutyric acid (GABA) type B receptor agonist and has been investigated in randomized, double-blind, placebo-controlled trials for the treatment of autism spectrum disorder and Fragile X syndrome. It has also been investigated for the treatment of spasticity caused by multiple sclerosis and spinal cord injury. Abaclofen has also been investigated for the treatment of gastroesophageal reflux disease (GERD). However, the results were disappointing. Drug Indications In clinical trials, this drug has been investigated as a potential therapy for the treatment of spasticity in multiple sclerosis, autism spectrum disorder, and social withdrawal in Fragile X syndrome. Mechanism of Action Abaclofen (or R-baclofen) acts upstream of the mGluR5 receptor to enhance inhibitory neurotransmission. It is an isomer of baclofen with antispasmodic activity.
(R)-baclofen (STX209) is the bioactive enantiomer of baclofen and acts as a selective GABAB receptor agonist [2] - The effects of intrathecal (R)-baclofen on the cardiovascular system in rats are entirely mediated by spinal GABAB receptors, as confirmed by the complete antagonism of faclofen [1] - In patients with spastic multiple sclerosis, (R)-baclofen inhibits spinal motor pathway transmission, which may be the basis for its clinical efficacy in reducing spastic symptoms [4] - (R)-baclofen exhibits significant stereoselectivity in binding to GABAB receptors, while the (S)-enantiomer has negligible affinity for GABAB receptors (Ki > 100 μM) [2] |
| Molecular Formula |
C10H12CLNO2
|
|
|---|---|---|
| Molecular Weight |
213.66
|
|
| Exact Mass |
213.055
|
|
| CAS # |
69308-37-8
|
|
| Related CAS # |
63701-55-3
|
|
| PubChem CID |
44602
|
|
| Appearance |
White to off-white solid powder
|
|
| Density |
1.3±0.1 g/cm3
|
|
| Boiling Point |
364.3±32.0 °C at 760 mmHg
|
|
| Melting Point |
171-174°C
|
|
| Flash Point |
174.1±25.1 °C
|
|
| Vapour Pressure |
0.0±0.9 mmHg at 25°C
|
|
| Index of Refraction |
1.577
|
|
| LogP |
1.56
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
3
|
|
| Rotatable Bond Count |
4
|
|
| Heavy Atom Count |
14
|
|
| Complexity |
191
|
|
| Defined Atom Stereocenter Count |
1
|
|
| SMILES |
C1=CC(=CC=C1[C@@H](CC(=O)O)CN)Cl
|
|
| InChi Key |
KPYSYYIEGFHWSV-QMMMGPOBSA-N
|
|
| InChi Code |
InChI=1S/C10H12ClNO2/c11-9-3-1-7(2-4-9)8(6-12)5-10(13)14/h1-4,8H,5-6,12H2,(H,13,14)/t8-/m0/s1
|
|
| Chemical Name |
(3R)-4-amino-3-(4-chlorophenyl)butanoic acid
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: 4.17 mg/mL (19.52 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication (<60°C).
(Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.6803 mL | 23.4017 mL | 46.8033 mL | |
| 5 mM | 0.9361 mL | 4.6803 mL | 9.3607 mL | |
| 10 mM | 0.4680 mL | 2.3402 mL | 4.6803 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
 |
|---|
 |
 |