
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
Scriptaid (formerly GCK1026; Scriptide) is a novel and potent histone deacetylase (HDAC) inhibitor with potential anticancer activity. It exhibits strong in vitro anti-proliferative activity against a variety of cancer cell types, including ovarian cancer cell lines SK-OV-3 and endometrial cancer cell lines Ishikawa. Additionally, it may be able to treat lymphomas linked to the Epstein-Barr virus (EBV) by sensitizing the activity of antivirals.
| Targets |
HDAC
The target of Scriptaid (GCK-1026; Scriptide) is histone deacetylases (HDACs), with broad inhibitory activity against class I HDACs (HDAC1, HDAC2, HDAC3) and moderate activity against some class II HDACs. In recombinant HDAC enzyme assays: - IC50 against HDAC1: ~0.5 μM [1] - IC50 against HDAC2: ~0.8 μM [1] - IC50 against HDAC3: ~1.2 μM [1] - IC50 against total HDAC activity in HeLa cell nuclear extracts: ~1.0 μM [5] It shows no significant inhibitory activity against class III HDACs (sirtuins) at concentrations up to 10 μM [5] |
|---|---|
| ln Vitro |
In vitro activity: Scriptaid (6 μM) causes a >100-fold increase in histone acetylation in PANC-1 cell. Scriptaid (8 μM) has minimal effects (80% survival rate) on MDAMB-468 but is not fatal to PANC-1 cells. Independent of a positive transcription inducer, scriptaid increases the transcription of pCMVb, p6SBE-luc, and p6MBE-luc. Using viral (SV40 and CMV) or human (ubiquitin c, UB6) promoters, Scriptaid can induce high expression of p6MBE-luc, pCMVb, and pUB6/V5-LacZ. This is independent of the reporter gene product (luciferase versus b-gal), the type of promoter (viral versus cellular), or the integration status of the reporter construct.[1] Scriptaid causes oocytes from somatic cell nuclear transfer (SCNT) to develop at high rates to the blastocyst stage. At all concentrations (50, 100, 250, 500, and 2000 nM, respectively), full-term development (3.4, 4.2, 7.6, 6.8, and 4.1%) is permitted. With a dose-dependent effect that peaks at 250 nM, scriptaid enhances the long-term development of cloned B6D2F1 embryos. The key inbred mouse strains, including DBA/2, C57BL/6, C3H/He, and 129/Sv, can all be cloned thanks to Scriptaid. Cloned embryos treated with Scriptaid have higher levels of newly synthesized mRNA. An ICSI-fertilized embryo's ability to develop is unaffected by a 48-hour exposure to 250 nM Scriptaid.[2] At an IC50 of 39 nM, scriptaid suppresses the growth of T. gondii tachyzoites. The HS68 monolayers are totally shielded from T. gondii tachyzoite by scriptaid (0.225 μM).[3] Scriptaid, after 48 hours of treatment, inhibits the growth of ER negative cell lines, MDA-MB-231, MDA-MB-435, and Hs578t, with an IC50 of 0.5-1.0 μg/mL. One microgram per milliliter After 48 hours of scriptaid treatment, ER mRNA transcript can increase by up to 20,000 times and both acetylated H3 and H4 histone tail proteins accumulate.[4] Scriptaid exhibits minimal sensitivity on normal human endometrial epithelial cells, but it inhibits the growth and viability of the Ishikawa endometrial cancer cell line and the SK-OV-3 ovarian cancer cell line with IC50 values of 9 μM and 55 μM, respectively. Cultured for two days in the presence of Scriptaid, endometrial and ovarian cancer cells exhibit an accumulation in the G0/G1 phase (5 μM of Scriptaid) and G2/M phase (10 μM of Scriptaid) of the cell cycle, accompanied by a decrease in the proportion of those in the S phase. Scriptaid at a concentration of 10 μM causes 56.1% of Ishikawa cells to undergo apoptosis, resulting in a loss of mitochondrial membrane potential and a 50% and 20% reduction in cyclin A and bcl-2 levels, respectively.[5]
1. Antiproliferative activity in cancer cells: - In human breast cancer cell lines (MCF-7, MDA-MB-231), Scriptaid (GCK-1026; Scriptide) inhibited cell proliferation in a dose-dependent manner. The IC50 values were ~2.5 μM (MCF-7) and ~3.0 μM (MDA-MB-231) after 72-hour treatment, as measured by MTT assay. Combination with tamoxifen (1 μM) enhanced antiproliferative effects, reducing IC50 to ~1.0 μM (MCF-7) [4] - In human colon cancer cell line (HCT116) and lung cancer cell line (A549), 72-hour treatment with Scriptaid (GCK-1026; Scriptide) resulted in IC50 values of ~1.8 μM and ~2.2 μM, respectively. At concentrations ≥5 μM, cell proliferation was inhibited by >80% compared to untreated controls [1] - In human hepatocellular carcinoma cell line (HepG2), Scriptaid (GCK-1026; Scriptide) (0.5-10 μM) suppressed cell growth with an IC50 of ~2.0 μM. Treatment for 48 hours also reduced colony formation efficiency: 5 μM Scriptaid decreased colony number by ~60% compared to controls [5] 2. HDAC inhibition and histone acetylation: - In HeLa cells, treatment with Scriptaid (GCK-1026; Scriptide) (0.1-5 μM) for 24 hours increased acetylation levels of histone H3 (Lys9/14) and histone H4 (Lys5/8/12/16) in a dose-dependent manner, as detected by western blot. Significant acetylation was observed at ≥0.5 μM, with maximum effects at 5 μM (3.5-fold increase for H3, 4.0-fold increase for H4 vs. controls) [5] - In MCF-7 breast cancer cells, 2 μM Scriptaid (GCK-1026; Scriptide) treatment for 24 hours increased acetyl-histone H3 levels by ~2.8-fold, which was associated with upregulation of p21WAF1/CIP1 (a cell cycle inhibitor) mRNA expression (2.5-fold increase, measured by RT-PCR) [4] 3. Apoptosis induction: - In HCT116 colon cancer cells, Scriptaid (GCK-1026; Scriptide) (5 μM) treatment for 48 hours induced apoptosis in ~35% of cells, as determined by Annexin V-FITC/PI staining and flow cytometry (vs. ~5% in controls). Western blot analysis showed increased cleavage of caspase-3 and PARP (apoptosis markers) at 24-48 hours post-treatment [1] - In HepG2 cells, 5 μM Scriptaid (GCK-1026; Scriptide) for 48 hours increased apoptotic cell rate to ~30%, accompanied by downregulation of anti-apoptotic protein Bcl-2 (0.4-fold vs. controls) and upregulation of pro-apoptotic protein Bax (1.8-fold vs. controls) [5] 4. Antiparasitic activity: - In vitro against the protozoan parasite Eimeria tenella (sporozoites), Scriptaid (GCK-1026; Scriptide) inhibited sporozoite invasion of chicken embryo fibroblasts (CEFs) and parasite replication. At 10 μM, invasion rate was reduced by ~70%, and intracellular parasite load (measured by RT-PCR for parasite 18S rRNA) was decreased by ~85% compared to controls. The IC50 for inhibiting parasite replication was ~3.5 μM [3] |
| ln Vivo |
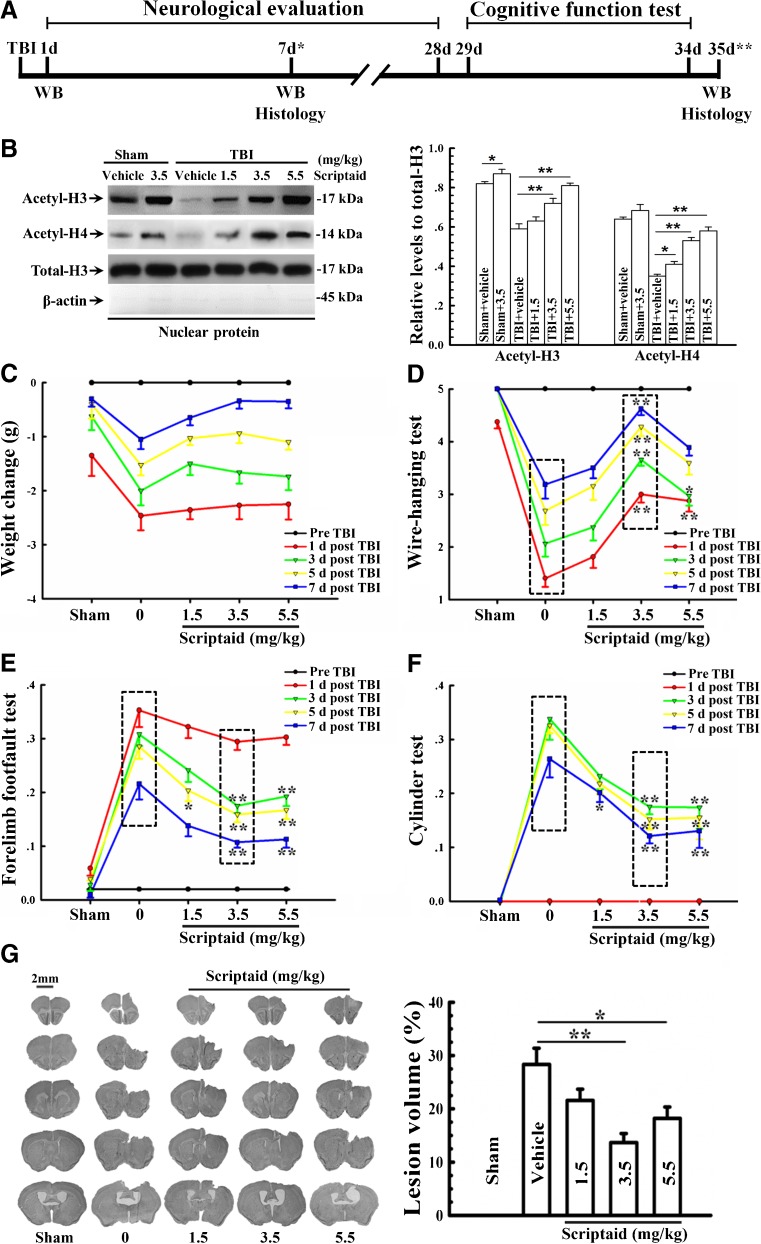
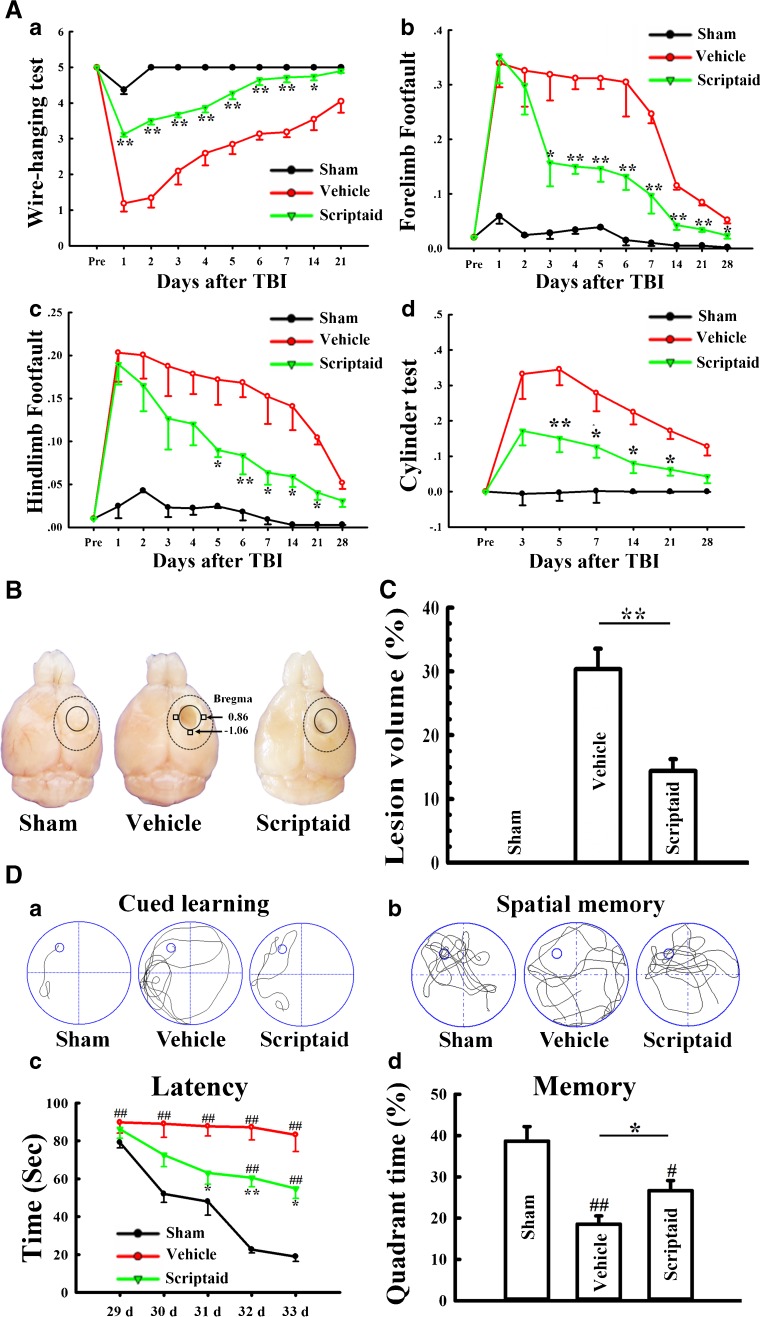
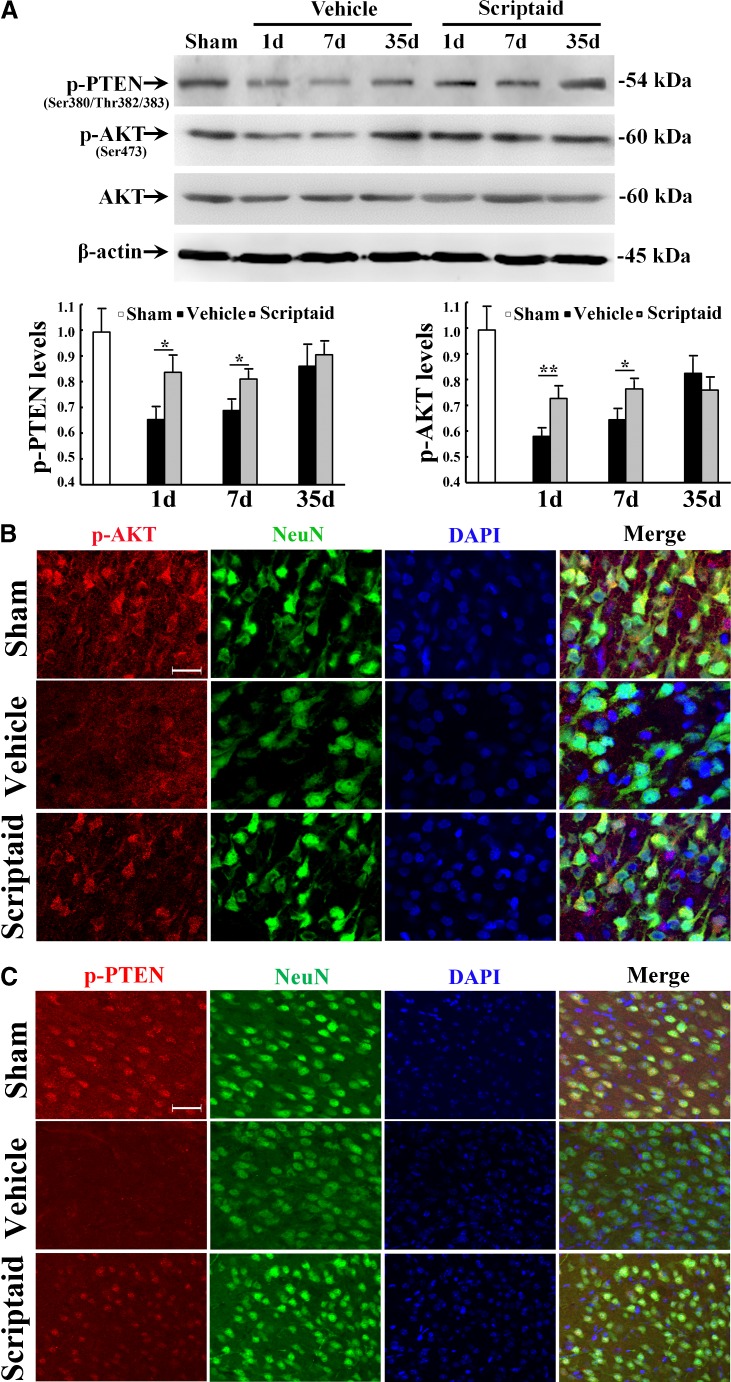
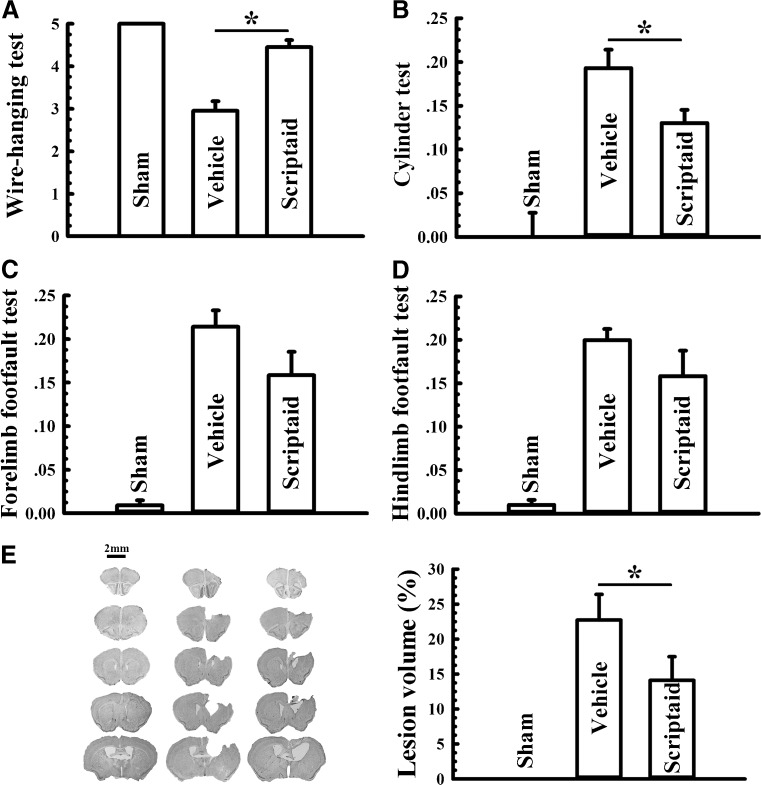
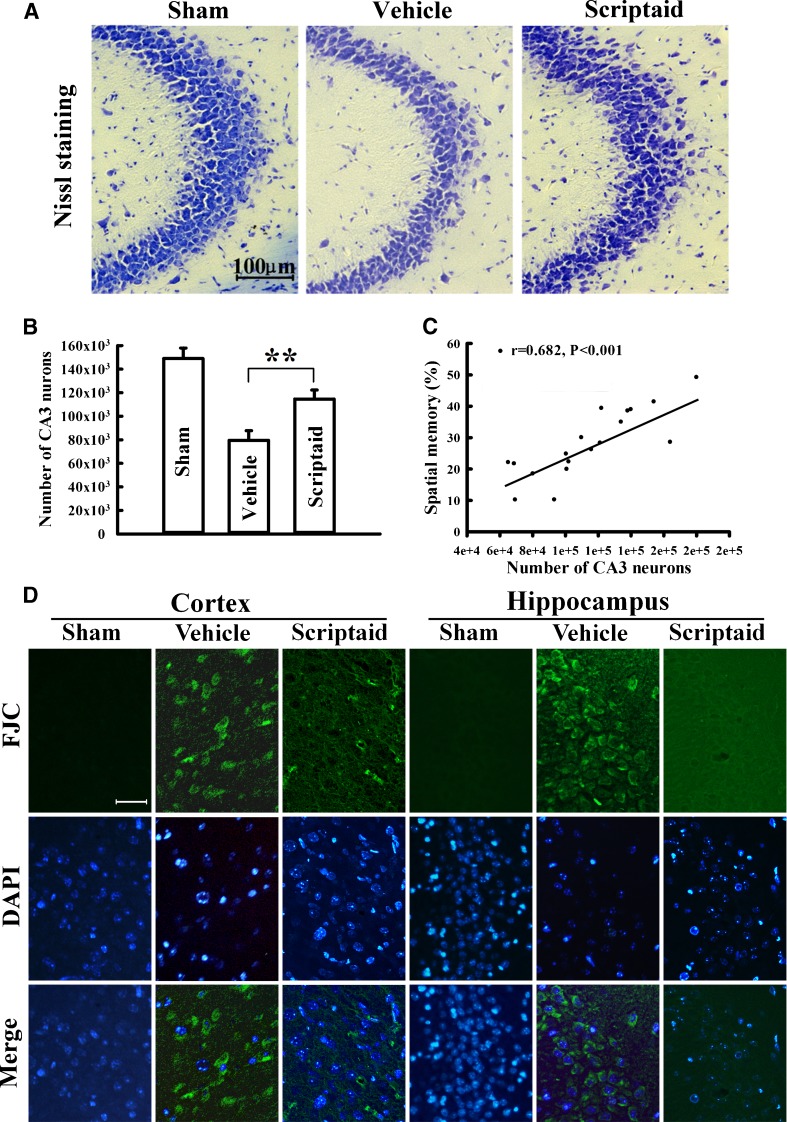
Scriptaid delivers 30 minutes post-injury and causes a dose-dependent reduction in lesion size (up to a maximum reduction of 45%), along with a concurrent attenuation of motor and cognitive deficits in a model of mode rate TBI. Treating an injury 12 hours after it occurs still results in comparable protection. As comparable gains are seen 35 days after the injury, the preservation of motor and cognitive abilities is long-lasting. In both the pericontusional cortex and the CA3 region of the hippocampus, scriptaid increases the number and length of surviving neurons (42%) as well as their relative numbers. The reduction in phospho-AKT (p-AKT) and phosphorylated phosphatase and tensin homolog deleted on chromosome 10 (p-PTEN) in cortical and CA3 hippocampal neurons caused by TBI is prevented by scriptaid treatment. [6] In a human breast cancer xenograft MDA-MB-231 model, scriptaid treatment (3.5 mg/kg) significantly inhibits tumor growth, resulting in a 75% reduction in tumor volume.[4]
1. Antitumor efficacy in mouse xenograft models: - In HCT116 colon cancer xenografts (nude mice), Scriptaid (GCK-1026; Scriptide) was administered intraperitoneally (i.p.) at 25 mg/kg and 50 mg/kg once daily for 21 days. The 25 mg/kg dose inhibited tumor growth by ~40%, while 50 mg/kg inhibited growth by ~65% compared to vehicle controls. Tumor weights at the end of treatment were ~0.8 g (25 mg/kg), ~0.5 g (50 mg/kg), and ~1.3 g (vehicle) [1] - In MCF-7 breast cancer xenografts (nude mice), combination treatment of Scriptaid (GCK-1026; Scriptide) (30 mg/kg i.p., daily) and tamoxifen (5 mg/kg oral, daily) for 28 days resulted in ~75% tumor growth inhibition, which was significantly higher than Scriptaid alone (~45%) or tamoxifen alone (~30%). No significant increase in toxicity was observed in the combination group [4] 2. Antiparasitic efficacy in chicken models: - In chickens infected with Eimeria tenella (oocyst challenge), Scriptaid (GCK-1026; Scriptide) was administered via oral gavage at 10 mg/kg and 20 mg/kg once daily for 5 days (starting 1 day post-infection). The 20 mg/kg dose reduced oocyst output in feces by ~60% and decreased intestinal lesion scores (from 3.5 in infected controls to 1.0 in treated groups). The 10 mg/kg dose showed moderate effects: ~35% reduction in oocysts and lesion score of 2.0 [3] 3. Histone acetylation in vivo: - In tumor tissues from HCT116 xenografts treated with 50 mg/kg Scriptaid (GCK-1026; Scriptide) (i.p., 7 days), western blot analysis showed a 2.8-fold increase in acetyl-histone H3 levels and a 3.2-fold increase in acetyl-histone H4 levels compared to vehicle-treated tumor tissues [1] |
| Enzyme Assay |
Scriptaid (2 μg/mL) is added to PANC-1 cells and left them in culture medium for eighteen hours. Using trypsin-EDTA, treated and untreated cells are separated, rinsed in PBS, and then resuspended in a protein sample buffer. Reagents for the BCA protein assay measure the concentration of proteins. A 12% denaturing polyacrylamide gel is loaded with 50 μg of proteins from every sample. Then, using a MilliblotGraphite Electroblotter I, proteins are transferred to a nylon membrane. Rabbit antihuman acetyl-lysine antibody is incubated on the nylon membrane, and then it is developed using SuperSignal substrates, goat antirabbit antibody coupled with horseradish peroxidase, and film is used to detect the results.
1. Recombinant HDAC enzyme inhibition assay: - Recombinant human HDAC1, HDAC2, or HDAC3 enzyme was mixed with a fluorogenic substrate (Boc-Lys(Ac)-AMC) in reaction buffer (25 mM Tris-HCl pH 8.0, 137 mM NaCl, 2.7 mM KCl, 1 mM MgCl2, 1 mM DTT). Scriptaid (GCK-1026; Scriptide) was added at concentrations ranging from 0.01 μM to 10 μM, and the mixture was incubated at 37°C for 60 minutes. After incubation, trypsin-containing developer solution was added to cleave deacetylated substrate, releasing fluorescent AMC. Fluorescence intensity was measured using a microplate reader (excitation: 360 nm, emission: 460 nm). The percentage of enzyme activity relative to vehicle controls was calculated, and IC50 values were determined by nonlinear regression [1] 2. Cellular HDAC activity assay (HeLa nuclear extracts): - Nuclear extracts were prepared from HeLa cells and mixed with [3H]-acetyl-labeled histone H4 substrate in assay buffer (50 mM Tris-HCl pH 8.0, 100 mM NaCl, 1 mM DTT, 0.1 mM EDTA). Scriptaid (GCK-1026; Scriptide) (0.05-10 μM) was added, and the mixture was incubated at 37°C for 90 minutes. The reaction was stopped by adding trichloroacetic acid (TCA) to precipitate proteins. Released [3H]-acetate in the supernatant was measured by liquid scintillation counting. HDAC activity was calculated as cpm of [3H]-acetate released, and IC50 was determined by dose-response curves [5] |
| Cell Assay |
The MTT assay is used to determine the IC50 concentrations of Scriptaid in MDA-MB-231, MDA-MB-435, and Hs578t cells. In order to conduct cell growth assays, MDA-MB-231, MDA-MB-435, and Hs578t cells are plated in 12-well plates at a density of 5000 cells per well. The cells are then treated with 1.0 µg/mL Scriptaid for a maximum of three days. A Coulter counter is used to count cells every day. Comparing treated and untreated cells yields the percent growth inhibition.
1. Cell proliferation assay (MTT method): - Cancer cells (MCF-7, MDA-MB-231, HCT116, A549, HepG2) were seeded in 96-well plates at 3×10³-5×10³ cells/well and incubated overnight. Scriptaid (GCK-1026; Scriptide) (0.1-20 μM) was added alone or in combination with tamoxifen (1 μM), and cells were cultured for 72 hours at 37°C (5% CO₂). MTT reagent (5 mg/mL) was added to each well (10 μL/well), and incubation continued for 4 hours. Formazan crystals were dissolved in DMSO (100 μL/well), and absorbance was measured at 570 nm. Cell viability (%) = (Absorbance of treated group / Absorbance of control group) × 100. IC50 values were calculated using GraphPad Prism software [1,4,5] 2. Western blot for histone acetylation and apoptosis markers: - Cells were treated with Scriptaid (GCK-1026; Scriptide) (0.1-10 μM) for 24-48 hours, then harvested and lysed in RIPA buffer (with protease inhibitors). Protein concentrations were determined by BCA assay. Equal amounts of protein (20-30 μg) were separated by 12% SDS-PAGE and transferred to PVDF membranes. Membranes were blocked with 5% non-fat milk in TBST for 1 hour, then incubated overnight at 4°C with primary antibodies (acetyl-histone H3, acetyl-histone H4, cleaved caspase-3, PARP, Bcl-2, Bax, β-actin). After washing with TBST, membranes were incubated with HRP-conjugated secondary antibodies for 1 hour at room temperature. Protein bands were visualized using ECL detection reagent, and band intensities were quantified with ImageJ software [1,4,5] 3. Apoptosis assay (Annexin V-FITC/PI staining): - HCT116 or HepG2 cells were treated with Scriptaid (GCK-1026; Scriptide) (1-10 μM) for 48 hours. Cells were harvested, washed with cold PBS, and resuspended in binding buffer (10 mM HEPES pH 7.4, 140 mM NaCl, 2.5 mM CaCl2). Annexin V-FITC (5 μL) and PI (10 μL) were added, and the mixture was incubated in the dark for 15 minutes at room temperature. Apoptotic cells (Annexin V+/PI- for early apoptosis, Annexin V+/PI+ for late apoptosis) were analyzed using a flow cytometer, and data were processed with FlowJo software [1,5] 4. Parasite invasion and replication assay: - Chicken embryo fibroblasts (CEFs) were seeded in 24-well plates and grown to confluence. Scriptaid (GCK-1026; Scriptide) (0.1-20 μM) was added to CEFs 1 hour before adding Eimeria tenella sporozoites (1×10⁵ sporozoites/well). After 24 hours, non-invaded sporozoites were removed by washing. For invasion assay: CEFs were fixed with methanol, stained with Giemsa, and invaded sporozoites were counted under a microscope. For replication assay: Cells were cultured for 72 hours, then total RNA was extracted. Parasite 18S rRNA expression (marker for replication) was measured by RT-PCR, with chicken GAPDH as internal control [3] |
| Animal Protocol |
For the length of the studies, athymic female nude mice, aged four to six weeks, are kept under laminar flow hoods in an environmentally controlled, pathogen-free facility. MDA-MB-231 human breast cancer cells are injected into each flank of mice at a ratio of 2×106. The tumors are left untreated until they reach a diameter of about 0.1 cm3. After that, mice receive intraperitoneal injections of Scriptaid (3.5 µg/g mouse), TSA (0.5 µg/g mouse), or DMSO vehicle for five days in a row, with two days off per week, for a total of four weeks. Every week, the measurements of each individual tumor from each flank are recorded.
1. Human cancer xenograft models (nude mice): - Female nude mice (6-7 weeks old) were used. For HCT116 colon cancer xenografts: 5×10⁶ HCT116 cells (suspended in 0.1 mL PBS + 50% Matrigel) were injected subcutaneously into the right flank. Tumors were allowed to grow to ~100 mm³ before treatment. Mice were randomized into 3 groups (n=6/group): vehicle control (10% DMSO + 40% PEG300 + 50% PBS), Scriptaid 25 mg/kg, Scriptaid 50 mg/kg. Scriptaid (GCK-1026; Scriptide) was administered via intraperitoneal injection once daily for 21 days. Tumor volume was measured twice weekly (Volume = length × width² / 2), and body weight was recorded [1] - For MCF-7 breast cancer xenografts: 5×10⁶ MCF-7 cells (in PBS/Matrigel) were injected subcutaneously. When tumors reached ~100 mm³, mice were divided into 4 groups (n=6/group): vehicle, Scriptaid 30 mg/kg (i.p., daily), tamoxifen 5 mg/kg (oral gavage, daily), combination (Scriptaid + tamoxifen). Treatment lasted 28 days. Tumor volume and body weight were monitored twice weekly. At the end of treatment, tumors were harvested for western blot analysis [4] 2. Eimeria tenella infection model (chickens): - One-week-old specific pathogen-free (SPF) chickens were randomized into 3 groups (n=10/group): non-infected control, infected control, infected + Scriptaid (10 mg/kg or 20 mg/kg). Chickens in infected groups were orally challenged with 1×10⁴ E. tenella oocysts. Scriptaid (GCK-1026; Scriptide) was dissolved in 0.5% carboxymethyl cellulose (CMC) and administered via oral gavage once daily for 5 days (starting 1 day post-infection). Feces were collected from days 5-8 post-infection to count oocysts. On day 8, chickens were euthanized, and intestinal tissues were scored for lesions (0-4 scale: 0 = no lesions, 4 = severe lesions) [3] |
| ADME/Pharmacokinetics |
Pharmacokinetics of mouse plasma: Nude mice were injected intraperitoneally with 50 mg/kg Scriptaid (GCK-1026; Scriptide) once, and blood samples were collected at 0.25, 0.5, 1, 2, 4, 6, 8 and 24 hours. Plasma was separated by centrifugation, and drug concentration was determined by high performance liquid chromatography-ultraviolet detection (HPLC-UV). The maximum plasma concentration (Cmax) was approximately 8.5 μM (0.5 hours), the terminal half-life (t₁/₂) was approximately 3.2 hours, and the AUC₀₋∞ was approximately 28 μM·h [1]
- Tissue distribution in mice: Mice were euthanized 1 hour (Cmax time) after intraperitoneal injection of 50 mg/kg Scriptaid (GCK-1026; Scriptide). Tumor, liver, kidney and lung tissues were collected, homogenized and analyzed for drug concentration. The highest drug concentration was found in the liver (approximately 15 μM), followed by tumors (approximately 7.2 μM), kidneys (approximately 6.8 μM), and lungs (approximately 4.5 μM). No drug was detected in brain tissue (<0.1 μM) [1] - Oral bioavailability in chickens: Chickens were given a single oral dose of 20 mg/kg of Scriptaid (GCK-1026; Scriptide). Plasma Cmax was approximately 3.0 μM (1 hour), t₁/₂ was approximately 2.5 hours, and oral bioavailability (compared to an intravenous dose of 5 mg/kg) was approximately 25% [3] |
| Toxicity/Toxicokinetics |
1. Acute toxicity in mice: - Mice were given a single intraperitoneal injection of Scriptaid (GCK-1026; Scriptide) (50-200 mg/kg). No deaths were observed in the ≤100 mg/kg dose group. In the 150 mg/kg dose group, 1 out of 6 mice died within 48 hours; in the 200 mg/kg dose group, 3 out of 6 mice died. Mice treated with ≥100 mg/kg showed transient lethargy and weight loss (5-10% of initial body weight) on days 1-2, which recovered by day 5 [1] 2. Chronic toxicity in xenograft studies: - In a 21-day HCT116 xenograft study (25-50 mg/kg intraperitoneally daily), no significant changes in blood cell counts (white blood cells, red blood cells, platelets) or serum biochemical parameters (ALT, AST, creatinine, BUN) were observed compared with the vector control group. No abnormal lesions were found in liver and kidney histopathological examination [1] - In the 28-day MCF-7 xenotransplantation study (30 mg/kg intraperitoneally daily), body weight change was ≤3% (compared to the control group), and no signs of organ toxicity were detected [4] 3. Toxicity in chickens: - Chickens treated with 10-20 mg/kg of Scriptaid (GCK-1026; Scriptide) (oral, 5 days) did not experience death or clinical symptoms (e.g., diarrhea, lethargy). Body weight gain was similar to that of the uninfected control group (while the infected control group had 15% less body weight gain). Serum AST and ALT levels were within the normal range [3] 4. Plasma protein binding: - Scriptaid (GCK-1026; Scriptide) (1 μM and 10 μM) was added to human plasma and incubated at 37°C for 30 minutes. Free drug and protein-bound drug were separated by ultrafiltration. Drug concentration in ultrafiltrate and plasma was determined by HPLC-UV. The plasma protein binding rate at both concentrations was approximately 92%[1]
|
| References | |
| Additional Infomation |
6-(1,3-dioxo-2-benzo[de]isoquinolinyl)-N-hydroxyhexanoamide is a member of the isoquinoline class of compounds. Scriptaid is a histone deacetylase (HDAC) inhibitor that has been studied for its antitumor properties. 1. Mechanism of Action: Scriptaid (GCK-1026; Scriptide) inhibits HDAC, leading to the accumulation of acetylated histones. This epigenetic modification relaxes chromatin structure, promoting the transcription of tumor suppressor genes (e.g., p21WAF1/CIP1, Bax) and antiparasitic genes, thereby inhibiting cancer cell proliferation and inducing apoptosis, and inhibiting parasite invasion/replication [1,3,4,5]. 2. Clinical Significance in Cancer: Scriptaid (GCK-1026; Scriptide) has shown potential as an anticancer drug, particularly in breast cancer (with synergistic effects with tamoxifen) and colon cancer. Its ability to inhibit HDAC without serious organ toxicity supports its further development in combination therapy for solid tumors [1,4]
3. Parasitological applications: Scriptaid (GCK-1026; Scriptide) is a promising candidate for the treatment of coccidiosis (caused by Eimeria spp.), one of the major diseases in poultry. Its good oral bioavailability and low toxicity in chickens make it suitable for agricultural use [3] 4. Comparison with other HDAC inhibitors: Scriptaid (GCK-1026; Scriptide) is less toxic than pan-HDAC inhibitors such as trachostatin A (TSA) in animal models. For example, intraperitoneal injection of 50 mg/kg Scriptaid was well tolerated in mice, while 10 mg/kg TSA resulted in a weight loss of more than 15% [1] |
| Molecular Formula |
C18H18N2O4
|
|
|---|---|---|
| Molecular Weight |
326.35
|
|
| Exact Mass |
326.126
|
|
| Elemental Analysis |
C, 66.25; H, 5.56; N, 8.58; O, 19.61
|
|
| CAS # |
287383-59-9
|
|
| Related CAS # |
|
|
| PubChem CID |
5186
|
|
| Appearance |
White to off-white solid powder
|
|
| Density |
1.3±0.1 g/cm3
|
|
| Melting Point |
160-161℃
|
|
| Index of Refraction |
1.646
|
|
| LogP |
0.43
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
4
|
|
| Rotatable Bond Count |
6
|
|
| Heavy Atom Count |
24
|
|
| Complexity |
477
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
O=C1C2=C([H])C([H])=C([H])C3C([H])=C([H])C([H])=C(C(N1C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C(N([H])O[H])=O)=O)C2=3
|
|
| InChi Key |
JTDYUFSDZATMKU-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C18H18N2O4/c21-15(19-24)10-2-1-3-11-20-17(22)13-8-4-6-12-7-5-9-14(16(12)13)18(20)23/h4-9,24H,1-3,10-11H2,(H,19,21)
|
|
| Chemical Name |
6-(1,3-dioxobenzo[de]isoquinolin-2-yl)-N-hydroxyhexanamide
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.66 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.66 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (7.66 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0642 mL | 15.3210 mL | 30.6419 mL | |
| 5 mM | 0.6128 mL | 3.0642 mL | 6.1284 mL | |
| 10 mM | 0.3064 mL | 1.5321 mL | 3.0642 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
 |
|---|
 |
 |
 |
|---|
 |