
| Size | Price | Stock | Qty |
|---|---|---|---|
| 1mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| Other Sizes |
Purity: ≥98%
Rilzabrutinib (PRN-1008) is a novel, highly potent, and reversible covalent inhibitor of BTK (Bruton's Tyrosine Kinase) being studied for treating rheumatoid arthritis. It inhibits BTK with an IC50 of 1.3 nM. BTK is a Tec family tyrosine kinase, is critical in immune pathways as an essential intracellular signaling element, participating in both adaptive and immune responses. PRN1008 was found to be very potent against BTK and highly selective when tested against a panel of 251 other kinases. Cysteine targeting of BTK by PRN1008 results in a slow off-rate demonstrated by retention of 79 ± 2% of binding to BTK in PBMC 18 hours after washing away the compound in vitro. PRN1008 was safe and well-tolerated following oral administration, and achieved high, sustained levels of BTK occupancy in peripheral blood mononuclear cells. PRN1008 is currently under Phase I development as a therapeutic agent for rheumatoid arthritis.
On August 29, 2025, the FDA approved rilzabrutinib (Wayrilz; Sanofi) as a first-in-class treatment for immune thrombocytopenia (ITP), a rare autoimmune disorder. ITP is characterized by platelet counts below 100,000/μL, driven by both increased platelet destruction and decreased production. In addition to bruising and bleeding, which may lead to life-threatening events like intracranial hemorrhage, patients are at risk for arterial or venous thrombosis. Many also experience symptoms that contribute to a reduced quality of life, like unexplained fatigue, cognitive impairment, and anxiety or depression.| Targets |
BTK (IC50 = 1.3 nM); BMX (IC50 = 1.0 nM); ITK (IC50 = 440 nM); TEC (IC50 = 0.8 nM); RLK (IC50 = 1.2 nM); BLK (IC50 = 6.3 nM); EGFR (IC50 = 520 nM); ERBB2 (IC50 = 3900 nM); ERBB4 (IC50 = 11.3 nM)
Bruton's tyrosine kinase (BTK) (Ki = 0.9 nM, determined by kinase activity assay; IC50 = 1.2 nM for BTK autophosphorylation inhibition) [2] - ITK, EGFR, Src, Lck (IC50 > 1000 nM, no significant inhibition) [2] |
|---|---|
| ln Vitro |
With an IC50 of 1.3±0.5 nM, rilzabrutinib is a reversible covalent inhibitor of Bruton's tyrosine kinase (BTK). Additionally, when rilzabrutinib was evaluated against a panel of 251 additional kinases, it was shown to be extremely selective. Rilzabrutinib, which targets the cysteine of BTK, caused a delayed dissociation rate; 18 hours after the chemical was washed away in vitro, 79±2% of bound BTK was still present in PBMC. Complete reversibility of covalent cysteine binding occurs upon target denaturation. Rilzabrutinib suppressed human B cell proliferation (10% serum) produced by anti-IgM and B cell CD69 expression, with IC50 values of 5±2.4 nM and 123±38 nM, respectively [2].
Acts as a novel reversible covalent inhibitor of BTK, specifically binding to the Cys481 residue of BTK with high affinity [1, 2] - Inhibited recombinant human BTK kinase activity in a concentration-dependent manner, with IC50 = 1.2 nM for autophosphorylation and Ki = 0.9 nM for substrate phosphorylation [2] - Suppressed B cell receptor (BCR)-mediated B cell activation: 10 nM Rilzabrutinib (PRN1008) reduced anti-IgM-induced human peripheral blood B cell proliferation by ~75% and inhibited BTK phosphorylation (p-BTK Y223) by ~90% [2] - Inhibited pro-inflammatory cytokine production in LPS-stimulated human monocytes: 100 nM concentration reduced TNF-α and IL-6 secretion by ~60% and ~55%, respectively [2] - Exhibited high kinase selectivity: no significant inhibition of 400+ tested kinases (including ITK, EGFR, Src, Lck) at concentrations up to 1 μM [2] - Reversed BTK inhibition after washout in vitro, confirming reversible binding (unlike irreversible BTK inhibitors) [2] |
| ln Vivo |
Following the drug's removal from the bloodstream, rilzabrutinib continues to have pharmacodynamic effects that are consistent with a prolonged target residence duration. Additionally, in a dose-dependent manner, rilzabrutinib reversed and totally suppressed collagen-induced arthritis in rats, which associated target occupancy with the alleviation of the disease [2].
In a Lewis rat collagen-induced arthritis (CIA) model, oral administration of Rilzabrutinib (PRN1008) (3, 10, 30 mg/kg, once daily for 21 days) starting at day 14 post-immunization dose-dependently alleviated arthritis symptoms [2] - Reduced paw edema volume by ~40% (3 mg/kg), ~55% (10 mg/kg), and ~70% (30 mg/kg) compared to vehicle control at day 28 [2] - Improved clinical arthritis score (0-4 per joint) from ~3.2 (vehicle) to ~2.1 (3 mg/kg), ~1.3 (10 mg/kg), and ~0.8 (30 mg/kg) [2] - Attenuated joint histopathology: reduced synovial hyperplasia, inflammatory cell infiltration, and bone erosion by ~50-75% at 30 mg/kg dose [2] - Decreased serum levels of pro-inflammatory cytokines (TNF-α, IL-6, IL-1β) by ~45-60% in CIA rats [2] |
| Enzyme Assay |
BTK kinase activity assay: Recombinant human BTK catalytic domain was incubated with ATP, a fluorescently labeled peptide substrate, and various concentrations of Rilzabrutinib (PRN1008) (0.01-100 nM) in kinase reaction buffer. The mixture was incubated at 37°C for 60 minutes, and the reaction was stopped by adding a kinase stop solution. Phosphorylated substrate was detected by fluorescence polarization, and IC50/Ki values were calculated based on inhibition of signal intensity [2]
- Kinase selectivity panel assay: Recombinant kinases (ITK, EGFR, Src, Lck, and 400+ others) were incubated with their respective substrates and Rilzabrutinib (PRN1008) (1 μM) in assay buffer. Kinase activity was measured using a high-throughput fluorescence-based method, and inhibition rates were determined to assess selectivity [2] |
| Cell Assay |
B cell proliferation assay: Human peripheral blood B cells were isolated and seeded in 96-well plates. Cells were pre-treated with Rilzabrutinib (PRN1008) (0.1-100 nM) for 1 hour, then stimulated with anti-IgM antibody (10 μg/mL) for 72 hours. Cell proliferation was assessed by MTT assay, and EC50 was calculated [2]
- BTK phosphorylation assay: Ramos B cells were serum-starved for 24 hours, treated with Rilzabrutinib (PRN1008) (0.1-100 nM) for 1 hour, then stimulated with anti-IgM (5 μg/mL) for 15 minutes. Cells were lysed, and proteins were analyzed by western blot using anti-p-BTK (Y223) and anti-total BTK antibodies [2] - Monocyte cytokine production assay: Human peripheral blood monocytes were isolated and treated with Rilzabrutinib (PRN1008) (0.1-1000 nM) for 1 hour, then stimulated with LPS (1 μg/mL) for 24 hours. Culture supernatants were collected, and TNF-α/IL-6 levels were quantified by ELISA [2] |
| Animal Protocol |
10, 20, 40 mg/kg
Rats with collagen-induced arthritis (CIA) model In this study, researchers assessed the effects of selective Btk inhibitors PRN1008 (rilzabrutinib) and PRN473 on platelet signalling and function mediated by CLEC-2 and GPVI. They used healthy donor and XLA platelets to determine off-target inhibitor effects. Inferior vena cava (IVC) stenosis and Salmonella infection mouse models were used to assess antithrombotic effects of PRN473 in vivo. PRN1008 and PRN473 potently inhibited CLEC-2-mediated platelet activation to rhodocytin. No off-target inhibition of SFKs was seen. PRN1008 treatment of Btk-deficient platelets resulted in minor additional inhibition of aggregation and tyrosine phosphorylation, likely reflecting inhibition of Tec. No effect on GPCR-mediated platelet function was observed. PRN473 significantly reduced the number of thrombi in podoplanin positive vessels following Salmonella infection and the presence of IVC thrombosis following vein stenosis. The potent inhibition of human platelet CLEC-2, and reduced thrombosis in in vivo models, together with the lack of off-target SFK inhibition and absence of bleeding reported in rilzabrutinib treated immune thrombocytopenia patients, suggest Btk inhibition as a promising antithrombotic strategy.[Selective Btk inhibition by PRN1008/PRN473 blocks human CLEC-2 & PRN473 reduces venous thrombosis formation in mice. Blood Adv. 2024 Jul 5:bloodadvances.2024012713] Rat collagen-induced arthritis (CIA) model: Female Lewis rats (150-180 g) were immunized by intradermal injection of bovine type II collagen emulsified in complete Freund's adjuvant. On day 14 post-immunization (onset of arthritis), rats were randomly divided into vehicle and treatment groups. Rilzabrutinib (PRN1008) was suspended in 0.5% carboxymethylcellulose sodium and administered orally at 3, 10, or 30 mg/kg, once daily for 21 days. Paw volume was measured weekly using a plethysmometer, and clinical arthritis score (0-4 per joint, 0 = normal, 4 = severe erosion) was assessed. At day 35, rats were euthanized, and hind paw joints were collected for histological analysis (HE staining) and inflammatory cell counting [2] |
| ADME/Pharmacokinetics |
Absorption
After multiple doses of 300 mg to 600 mg of rizatinib, its Cmax and AUC increase proportionally. At the approved recommended dose, steady-state plasma concentrations are reached within three days, with a cumulative increase of up to 1.3 times. After twice-daily administration of 400 mg rizatinib, the steady-state Cmax and AUC24h were 150 ng/mL (56%) and 1540 ng·h/mL (57.5%), respectively. The absolute oral bioavailability of rizatinib is 4.7%. The median time to peak concentration (Tmax) after a single oral dose of 400 mg rizatinib is approximately two hours. After a single oral dose of 400 mg rizatinib followed by a high-fat meal (approximately 1000 calories, 50% of which are from fat), the AUC and Cmax of rizatinib decreased by 20% and 31%, respectively. Elimination Pathway In healthy subjects, following a single dose of 400 mg of 14C-labeled rizatinib, approximately 86% of the dose is excreted in the feces (9% unchanged), with smaller amounts excreted in the urine (approximately 5%) and bile (approximately 6%). Approximately 0.03% of rizatinib is excreted unchanged in the urine. Volume of Distribution After intravenous administration, the terminal volume of distribution (Vz) is 149 L. Clearance Rizatinib clearance is time-independent. In ITP patients, after multiple doses of 400 mg rizatinib twice daily, the mean CL/F ranged from 246 to 911 L/h. Based on population pharmacokinetic analysis in ITP patients, the mean CL/F was 516 L/h. Protein Binding Rizatinib binds to plasma proteins at a rate of 97.5%, with a plasma concentration/plasma concentration ratio of 0.786. Metabolism/Metabolites Rizatinib is primarily metabolized via cytochrome P450 (CYP) 3A4. Its metabolic pathway and metabolites are not fully elucidated, but it has been reported that rizatinib metabolites are pharmacologically inactive. Biological Half-Life In Phase I studies, the half-life of rizatinib ranged from 1.6 to 4.5 hours. Four hours after PRN1008 administration, Cmax was highly correlated with BTK occupancy (Figure 4). A fitted sigmoid Emax model with γ and E0 best fits the Cmax and occupancy data for PRN1008. The parameter estimates (percentage of coefficient of variation) were: Emax 76.7 (13) %; EC50 46.5 (15) ng ml–1; E0 17.8 (42) %; and γ 2.1 (31). These results indicate that the exposure-response relationship is quite steep with low variability, and that a maximum occupancy of 80% is achieved at Cmax concentrations of approximately 100 ng ml–1 and above. The E0 value of 17.8% is consistent with the lower limit of quantitation for this assay. The robust relationship between PRN1008 Cmax and 4-hour occupancy, along with a very stable occupancy decay rate (approximately 1.6% h–1), should aid in designing dosing regimens (dose and dosing intervals) to precisely control different occupancy levels within the dosing interval. Notably, the pharmacokinetic (PK) and pharmacokinetic/pharmacodynamic (PK/PD) relationships of PRN1008 may differ between healthy volunteers and patient populations and should be further evaluated in future patient studies. [1] This single-center, open-label, non-randomized, two-part phase I study aimed to (1) evaluate the absolute oral bioavailability of a micro-dose of the tracer ~100 μg [14C]-rilzabrutinib (~1 μCi) and a single oral dose of 400 mg rilzabrutinib tablets (Part 1), and (2) characterize the absorption, metabolism, and excretion (AME) of 14C radiolabeled rilzabrutinib (Part 2) following a single oral dose of [14C]-rilzabrutinib (~1000 μCi; administered in liquid form) (300 mg) in healthy male subjects. A total of 18 subjects were enrolled (n=8 in Part 1; n=10 in Part 2). The absolute bioavailability of the 400 mg oral rilzabrutinib tablets was low (<5%). In Part I, following a single oral dose of 400 mg rilzabrutinib tablets, the drug was rapidly absorbed, with a median time to peak concentration (Tmax) of 2.03 h (range: 1.83–2.50 h). Following oral and intravenous administration of approximately 100 μg of [14C]-rilzabrutinib microtracers, the geometric mean (coefficient of variation) of the terminal half-life was 3.20 h (51.0%) and 1.78 h (37.6%), respectively. In Part II, following a single oral dose of 300 mg [14C]-rilzabrutinib solution, rilzabrutinib was also rapidly absorbed, with a median (range) Tmax of 1.00 h (1.00–2.00 h). In subjects who did not collect bile (92.9%) and those who did collect bile (87.6%), most of the radioactive material was present in feces, with approximately 5% excreted in urine following oral administration. The excretion of unmetabolized rilzabrutinib in urine was low (3.02%). These findings enhance our understanding of the absolute bioavailability and AME of rilzabrutinib and may provide guidance for further research. Clin Transl Sci. July 2023; 16(7):1210-1219. Human Phase I trial (healthy volunteers): Single oral doses of 50-800 mg showed dose-proportional pharmacokinetics[1] - Peak plasma concentration (Cmax) = 1.2-9.8 μg/mL (50-800 mg dose), Tmax = 1-2 hours[1] - Oral bioavailability = ~70%[1] - Plasma half-life (t1/2) = 8-10 hours[1] - Volume of distribution (Vd) = ~120 L[1] - Primarily metabolized in the liver via CYP3A4; approximately 30% is excreted in the urine, and approximately 60% is excreted in the feces as metabolites[1] - Rat pharmacokinetics: Oral bioavailability is approximately 65%, half-life is 4-6 hours, and it is widely distributed in tissues. The ratio of joint tissue to plasma concentration is approximately 1.3 2 hours after administration[2] |
| Toxicity/Toxicokinetics |
Data from all 80 enrolled subjects who received the investigational drug (PRN1008 or placebo) were included in the safety analysis population. All subjects were assessed for adverse events (AEs) during the study. No serious adverse events or deaths were reported during the study, and no subjects discontinued treatment due to adverse events in either Part A or Part B. The safety and tolerability of PRN1008 at single doses of 50 to 600 mg were similar to placebo. In each of the four cohorts, only one of the six subjects in each cohort experienced a treatment-specific adverse event (TEAE). Of these four TEAEs, only one was considered related to the investigational drug (nausea in cohort A4), and the other was rated as moderate (toothache in cohort A2, unrelated to the investigational drug). In contrast, two of the ten subjects in the placebo group reported two TEAEs (both rated as mild and unrelated to the drug). In the A5 group (1200 mg), the primary adverse events observed were gastrointestinal adverse events, which were reported by all six subjects treated with PRN1008. Drug-related adverse events included diarrhea (coded as loose stools; n = 6, 3 mild and 3 moderate), nausea (n = 3, 2 mild and 1 moderate), vomiting (n = 1), throat irritation (n = 3), and oropharyngeal discomfort (n = 1). No significant association was found between gastrointestinal adverse events and the pharmacokinetics of PRN1008. As mentioned above, the Cmax and area under the concentration-time curve were similar at 600 mg and 1200 mg doses of PRN1008. Although plasma pharmacokinetics were similar, the increased incidence of gastrointestinal adverse events in the 600 mg dose group compared to the 1200 mg dose group suggests that local effects are related to the total administered dose rather than plasma exposure. PRN1008 was generally safe and well-tolerated after 10 or 11 days of administration. In the 300 mg once daily, 300 mg twice daily, 600 mg once daily, 450 mg twice daily, and placebo groups, 7/8, 4/8, 8/8, 7/8, and 4/8 subjects, respectively, reported adverse events during treatment. Treatment-related adverse events during treatment appeared to be more common in the PRN1008 treatment groups, with 6/8, 3/8, 8/8, 6/8, and 1/8 subjects, respectively, reporting such events in the 300 mg once daily, 300 mg twice daily, 600 mg once daily, 450 mg twice daily, and placebo groups. Except for one subject in the 450 mg BID group who reported moderate diarrhea, all treatment-related adverse events (TEAEs) classified as investigational drug-related were mild. No clinically significant or dose-dependent changes were observed in hematological, biochemical, or coagulation laboratory parameters in Parts A and B of the study. Similarly, no clinically significant changes were observed in vital signs, electrocardiograms, or urinalysis assessments. [1]
Phase I human trials: well tolerated at doses up to 800 mg[1] - Common adverse events (AEs): mild headache (8%), diarrhea (6%), fatigue (5%); all adverse events were transient and resolved without intervention[1] - No significant changes in liver and kidney function (ALT, AST, creatinine), hematological parameters (white blood cell count, platelet count, hemoglobin) or electrocardiogram parameters[1] - Subchronic toxicity in rats: no significant organ toxicity was observed after oral administration of 30 mg/kg daily for 28 days; no histological abnormalities were observed in the liver, kidneys, spleen or lymphoid tissues[2] - Plasma protein binding rate: approximately 92% in humans; approximately 90% in rats[1, 2] - In vitro studies have shown that this product has no significant drug interaction with CYP3A4 substrates/inhibitors[1] |
| References |
|
| Additional Infomation |
Rilzabrutinib is an orally administered, reversible covalent inhibitor of Bruton's tyrosine kinase, currently under investigation for its efficacy in treating immune disorders such as immune thrombocytopenic purpura. Rizatinib is an orally bioavailable, reversible covalent inhibitor of Bruton's tyrosine kinase (BTK) with potential immunomodulatory and anti-inflammatory activity. Oral administration of ritazinib inhibits BTK activity, thereby preventing activation of the B cell antigen receptor (BCR) signaling pathway and subsequently suppressing immune activation and inflammatory responses. BTK is a cytoplasmic tyrosine kinase belonging to the Tec kinase family, playing a crucial role in the development, activation, signal transduction, proliferation, and survival of B lymphocytes. Besides B cells, BTK is also expressed in other hematopoietic cells, including monocytes, macrophages, neutrophils, mast cells, eosinophils, and platelets, and plays an important role in adaptive and innate immune responses.
Drug Indications Treatment of Immune Thrombocytopenic Pulmonary DiseaseRilzabrutinib (PRN1008) is a first-in-class reversible covalent inhibitor of Bruton's tyrosine kinase (BTK), which differs from irreversible BTK inhibitors (such as ibrutinib) in that it reversibly binds to Cys481 of BTK [1, 2]. - Its mechanism of action involves reversible covalent binding to BTK, inhibiting BCR-mediated signaling in B cells and the production of pro-inflammatory cytokines in monocytes/macrophages, thereby suppressing the immune response. Autoimmune inflammation [2] - Potential therapeutic indications include autoimmune diseases such as rheumatoid arthritis, systemic lupus erythematosus, and immune thrombocytopenic purpura [2] - Phase I clinical data showed that the drug had good safety, tolerability, and pharmacokinetic characteristics in healthy volunteers, supporting its further clinical development in autoimmune diseases [1] - Reversible binding may reduce long-term off-target effects (e.g., bleeding, atrial fibrillation) associated with irreversible BTK inhibitors, thereby improving safety [2] Mechanism of action Immune thrombocytopenic purpura (ITP) is an autoimmune disease characterized by low platelet counts, bruising, and bleeding events. It is caused by a complex dysregulation of the immune system, leading to increased destruction of circulating platelets and impaired platelet production. A major mechanism involves the production of autoantibodies, usually IgG antibodies, which bind to glycoproteins on the surface of platelets. This binding marks platelets, causing them to be cleared, primarily through phagocytosis by macrophages in the spleen and liver. This process is mediated by the binding of the Fc fragment of autoantibodies to the Fcγ receptor (FcγR) on macrophages. Another key pathway involves the action of Bruton's tyrosine kinase (BTK), an intracellular signaling molecule expressed in various immune cells, including B cells, macrophages, and mast cells. In idiopathic thrombocytopenic purpura (ITP), BTK signaling promotes B cell production of anti-platelet autoantibodies and promotes macrophage-mediated platelet destruction via FcγR. Rizatinib inhibits B cell activation in the spleen and liver through reversible covalent inhibition of BTK and blocks Fcγ receptor (FcγR)-mediated phagocytosis of antibody-coated cells. In vitro experiments show that ritazinib reduces FcγR-mediated autoantibody signaling, blocks B cell signaling, and reduces autoantibody production by affecting B cell activation. Rizatinib is an oral, reversible, covalently inhibitor of Bruton's tyrosine kinase. It was approved by the U.S. Food and Drug Administration (FDA) on August 29, 2025, for the treatment of persistent and chronic immune thrombocytopenic purpura (ITP). ITP is an autoimmune disease characterized by low platelet counts. Rizatinib is believed to treat ITP through a dual mechanism of action: it attenuates macrophage (Fcγ receptor)-mediated platelet destruction and reduces the production of pathogenic autoantibodies. Rizatinib is a kinase inhibitor. Its mechanism of action includes inhibition of Bruton's tyrosine kinase, cytochrome P450 3A, P-glycoprotein, breast cancer resistance protein, organic anion transport peptide 1B1, organic anion transport peptide 1B3, and bile acid export pumps. Rizatinib is an orally bioavailable, reversible, covalently inhibitor of Bruton's tyrosine kinase (BTK) with potential immunomodulatory and anti-inflammatory activity. After oral administration, rizabrutinib inhibits BTK activity. This prevents activation of the B-cell antigen receptor (BCR) signaling pathway, and the resulting immune activation and inflammation. BTK is a cytoplasmic tyrosine kinase belonging to the Tec kinase family, playing a crucial role in the development, activation, signal transduction, proliferation, and survival of B lymphocytes. Besides B cells, BTK is also expressed in other hematopoietic cells, including monocytes, macrophages, neutrophils, mast cells, eosinophils, and platelets, and plays an important role in adaptive and innate immune responses. Rizabrutinib is a small molecule drug that has completed the most Phase III clinical trials (covering all indications) and has 10 investigational indications. |
| Molecular Formula |
C36H40FN9O3
|
|
|---|---|---|
| Molecular Weight |
665.7597
|
|
| Exact Mass |
665.32
|
|
| Elemental Analysis |
C, 64.95; H, 6.06; F, 2.85; N, 18.93; O, 7.21
|
|
| CAS # |
1575596-29-0
|
|
| Related CAS # |
1575596-77-8;1575596-29-0;1575591-66-0
|
|
| PubChem CID |
73388818
|
|
| Appearance |
White to off-white solid powder
|
|
| LogP |
3.4
|
|
| Hydrogen Bond Donor Count |
1
|
|
| Hydrogen Bond Acceptor Count |
11
|
|
| Rotatable Bond Count |
8
|
|
| Heavy Atom Count |
49
|
|
| Complexity |
1230
|
|
| Defined Atom Stereocenter Count |
1
|
|
| SMILES |
CC(C)(/C=C(\C#N)/C(=O)N1CCC[C@H](C1)N2C3=NC=NC(=C3C(=N2)C4=C(C=C(C=C4)OC5=CC=CC=C5)F)N)N6CCN(CC6)C7COC7
|
|
| InChi Key |
LCFFREMLXLZNHE-GBOLQPHISA-N
|
|
| InChi Code |
InChI=1S/C36H40FN9O3/c1-36(2,45-15-13-43(14-16-45)26-21-48-22-26)18-24(19-38)35(47)44-12-6-7-25(20-44)46-34-31(33(39)40-23-41-34)32(42-46)29-11-10-28(17-30(29)37)49-27-8-4-3-5-9-27/h3-5,8-11,17-18,23,25-26H,6-7,12-16,20-22H2,1-2H3,(H2,39,40,41)/b24-18+/t25-/m1/s1
|
|
| Chemical Name |
|
|
| Synonyms |
PRN-1008; PRN1008; Rilzabrutinib, (E)-; 5G1WE425BI; (E)-2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxyphenyl)pyrazolo[3,4-d]pyrimidin-1-yl]piperidine-1-carbonyl]-4-methyl-4-[4-(oxetan-3-yl)piperazin-1-yl]pent-2-enenitrile; PRN 1008;
1575591-66-0;
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.12 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (3.12 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.08 mg/mL (3.12 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5020 mL | 7.5102 mL | 15.0204 mL | |
| 5 mM | 0.3004 mL | 1.5020 mL | 3.0041 mL | |
| 10 mM | 0.1502 mL | 0.7510 mL | 1.5020 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
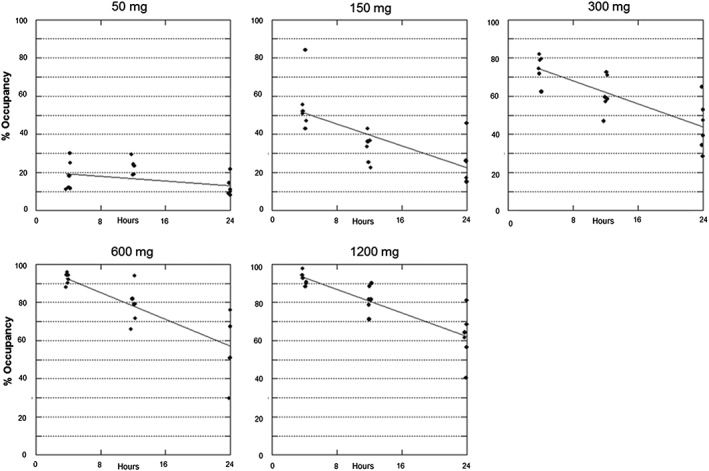 Individual BTK occupancy by PRN1008 dose level (Part A). Solid line represents fit of a linear regression model to estimate loss of occupancy over time.Br J Clin Pharmacol.2017 Nov;83(11):2367-2376. |
|---|
Duration of BTK occupancy (squares) in relation to the plasma concentration profile of PRN1008 (circles), following final dose on day 10 of a 600mg once daily dosing regimen in the multiple ascending dose study.Br J Clin Pharmacol.2017 Nov;83(11):2367-2376. |
Exposure–response relationship between 4‐hour BTK occupancy and PRN1008 maximum observed concentration (Part A).Br J Clin Pharmacol.2017 Nov;83(11):2367-2376. |