
| Size | Price | Stock | Qty |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
Purity: ≥98%
Pancuronium dibromide (OrgNA97; NA-97; Org NA 97; Pavulon), a neuromuscular relaxant used in euthanasia, is a bis-quaternary steroid and a competitive nicotinic acetylcholine receptor antagonist with an IC50 of 5.5 nM. As a neuromuscular blocking agent, Pancuronium bromide is more potent than curare but has less effect on the circulatory system and on histamine release. Pancuronium dibromide interrupts neuromuscular transmission by competing with acetylcholine for receptor sites on the motor end-plate.
| Targets |
Pre-junctional muscarinic receptors (antagonist) [1]
- Post-junctional muscarinic receptors (antagonist) [1] - Sodium channels (inhibitory activity) [2] |
|---|---|
| ln Vitro |
Pancuronium's effect on transmembrane sodium conductance is examined in chick embryonic dorsal root ganglion neurons. Pancuronium, when externally perfused at concentrations between 50 μM and 1 mM, quickly and reversibly inhibits current flow. With a half-effective dose of 170 μM, inhibition is concentration-dependent but not voltage-dependent. By interacting with the sodium channels in both the resting and open states, pancuronium may lower the sodium current[2].
Incubation of isolated guinea-pig lung tissue with Pancuronium dibromide (1-10 μM) antagonized pre- and post-junctional muscarinic receptors, inhibiting acetylcholine-induced bronchoconstriction by 65% and reducing acetylcholine release from pre-junctional terminals by 40% [1] - Pancuronium dibromide (10-100 μM) dose-dependently inhibited sodium current in chick embryo sensory neurons, with 100 μM reducing peak sodium current amplitude by 58% without altering current kinetics [2] - In isolated rat phrenic nerve-diaphragm preparations, Pancuronium dibromide (0.1-1 μM) blocked somatic neuromuscular transmission, inhibiting twitch response to nerve stimulation by 72% at 1 μM [3] - Pancuronium dibromide (5 μM) antagonized cholinergic autonomic neurotransmission in isolated rat atria, reducing acetylcholine-induced bradycardia by 55% [3] |
| ln Vivo |
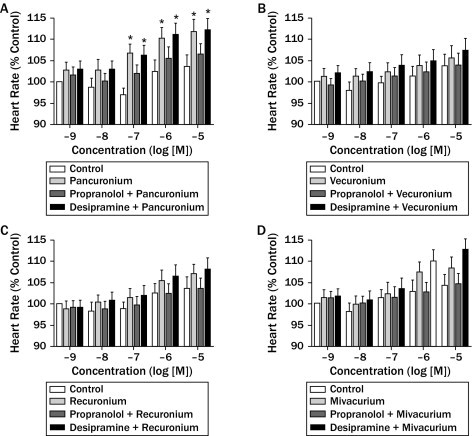
In male guinea pigs weighing 250–300 g, pancuronium (0.5 mg/kg) administered intravenously reverses the bradycardia caused by vagal nerve stimulation and injected acetylcholine (ACh). Vagally-induced bronchoconstriction is amplified by pancuronium (0.04 mg/kg) at levels that result in 100% neuromuscular blockade[1]. Rat anococcygeus and vas deferens exhibit pancuronium-mediated adrenergic nerve stimulation potentiation[3].
Intravenous injection of Pancuronium dibromide (0.1 mg/kg) to rats produced dose-dependent neuromuscular blockade, with maximum suppression of gastrocnemius muscle twitch response (80%) at 5 minutes post-administration, lasting for 30 minutes [3] - In anesthetized rats, Pancuronium dibromide (0.2 mg/kg, iv) altered autonomic function, increasing heart rate by 25% and reducing vagally mediated bradycardia by 60%, consistent with muscarinic receptor antagonism [3] |
| Enzyme Assay |
Muscarinic receptor binding assay: Membrane fractions were prepared from guinea-pig lung tissue. Pancuronium dibromide (0.01-100 μM) was incubated with membranes and [³H]quinuclidinyl benzilate (muscarinic ligand) at 25°C for 60 minutes. Unbound ligand was removed by filtration, and bound radioactivity was quantified to assess competitive antagonism [1]
- Autonomic receptor binding assay: Membrane fractions from rat atrial tissue were prepared. Pancuronium dibromide (0.1-10 μM) was incubated with membranes and [³H]acetylcholine at 37°C for 45 minutes. Bound radioactivity was measured after filtration to evaluate receptor blocking efficacy [3] |
| Cell Assay |
Sodium current inhibition assay: Chick embryo sensory neurons were dissociated and cultured for 2-3 days. Pancuronium dibromide (1-100 μM) was added to the culture medium, and sodium currents were recorded using whole-cell patch-clamp technique. Current amplitude and kinetics were analyzed to assess inhibitory effects [2]
|
| Animal Protocol |
NA
guinea-pig Neuromuscular transmission model: Male Wistar rats (10 weeks old) were anesthetized, and the gastrocnemius muscle was instrumented to record twitch responses to sciatic nerve stimulation. Pancuronium dibromide (0.05-0.2 mg/kg) was administered intravenously, and twitch suppression was measured for 60 minutes [3] - Autonomic function model: Anesthetized rats were implanted with electrodes to record heart rate. Pancuronium dibromide (0.1-0.3 mg/kg, iv) was administered, and vagally induced bradycardia (via cervical vagus nerve stimulation) was measured to assess autonomic neurotransmission blockade [3] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
241 to 280 mL/kg Plasma clearance = 1.1–1.9 mL/min/kg Both the liver and kidneys participate in the degradation and excretion of pancuronium bromide… After intravenous injection, the effect is felt within 3 minutes in adults and within 90 seconds in children. …The plasma half-life may be slightly less than 2 hours. Pancuronium bromide is primarily excreted unchanged in the urine. Following maternal administration, pancuronium bromide is rapidly transported across the placenta, but the fetal-to-maternal drug concentration ratio is extremely low. In 7 patients who received intravenous injection of pancuronium bromide, plasma drug concentrations conformed to a two-compartment kinetic model, with a β-phase half-life between 90 and 162 minutes. The average central compartment volume was 100 mL/kg, and the total volume of distribution was 261 mg/kg. In patients with chronic renal failure, plasma clearance was significantly reduced, while total and central compartment volumes were significantly increased. For more complete data on the absorption, distribution, and excretion of pancuronium (a total of 6 metabolites), please visit the HSDB record page. Metabolism/Metabolites Hepatic metabolism. In cats, 8 hours after intravenous administration of pancuronium, 58% of the dose of pancuronium was unmetabolized in urine, bile, and liver, 14.5% in the 3-hydroxy derivative, 7% in the 17-hydroxy derivative, and 4.5% in the 3,17-dihydroxy derivative. Biological half-life: 1.5 to 2.7 hours. Plasma half-life may be slightly less than 2 hours. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation
◉ Overview of Drug Use During Lactation There is currently no information regarding the use of pancuronium bromide during lactation. Due to its high polarity and poor oral absorption, it is unlikely to enter breast milk or the infant's bloodstream in high concentrations. When multiple anesthetics are used during surgery, follow the advice of the most likely problematic drug used during the procedure. ◉ Effects on Breastfed Infants No relevant published information was found as of the revision date. ◉ Effects on Lactation and Breast Milk No relevant published information was found as of the revision date. Protein Binding 77% to 91% Drug Interactions From a clinical perspective, the most important pharmacological interactions of these drugs are with certain general anesthetics, certain antibiotics, and anticholinesterase compounds. Neuromuscular blocking agents …Diethyl ether and succinylcholine can enhance and prolong the action of pancuronium bromide. Ether has a stabilizing effect on the postsynaptic membrane, thus exhibiting a synergistic effect with competitive blockers. …Fluorane, cyclopropane, fluoroquinolones, methoxyflurane, and enflurane also have a synergistic effect with competitive blockers, but to a weaker degree. /Neuromuscular competitive blockers/ Aminoglycoside antibiotics produce neuromuscular blocking effects by inhibiting the release of acetylcholine from preganglionic terminals (through competition with Ca²⁺) and stabilizing the postsynaptic membrane. Calcium salts can antagonize this blocking effect, but the antagonistic effect of anticholinesterase drugs is unstable. Tetracycline antibiotics can also produce neuromuscular blocking effects, possibly through chelation of calcium ions. Other antibiotics with neuromuscular blocking effects include polymyxin B, colistin, clindamycin, and lincomycin. /Neuromuscular Blocking Agents/ For more complete data on interactions of pancuronium bromide (27 in total), please visit the HSDB record page. Non-human Toxicity Values> Oral LD50 in rats: 202 mg/kg Intraperitoneal LD50 in rats: 479 μg/kg Subcutaneous LD50 in rats: 436 μg/kg Intravenous LD50 in rats: 153 μg/kg For more complete data on non-human toxicity values of pancuronium bromide (10 in total), please visit the HSDB record page. Pancuronium bromide (100 μM) showed no significant cytotoxicity after 24 hours of exposure in chicken embryo sensory neurons, as assessed by trypan blue exclusion [2]. In vivo administration to rats at therapeutic doses (0.1–0.2 mg/kg, intravenous) did not cause significant toxicity, but high doses (>1 mg/kg) led to respiratory depression due to severe neuromuscular blockade [3]. |
| References |
|
| Additional Infomation |
Pancuronium bromide is a steroidal ester with its 5α-androstane skeleton having acetoxy disubstituted positions at C-3α and C-17β, and 1-methylpiperidin-1-yl disubstituted positions at 2β and 16β. It is a non-depolarizing curare-like muscle relaxant. It has dual effects of muscle relaxation, cholinergic antagonism, and nicotinic antagonism. It is a steroidal ester and an acetate ester. It is a bisquaternary ammonium steroid and also a competitive nicotinic antagonist. As a neuromuscular blocking agent, it is more potent than curare, but has less effect on the circulatory system and histamine release. Pancuronium bromide is a non-depolarizing neuromuscular blocking agent. The physiological effects of pancuronium bromide are achieved through non-depolarizing neuromuscular blockade. Pancuronium bromide is a synthetic, long-acting bisquaternary ammonium steroid and also a non-depolarizing neuromuscular blocking agent with muscle relaxant effects. Pancuronium bromide competitively binds to and blocks nicotinic acetylcholine receptors at the neuromuscular junction, thereby preventing the binding of acetylcholine (ACh) and leading to skeletal muscle relaxation and paralysis. It is a competitive nicotinic receptor antagonist. As a neuromuscular blocking agent, it is more potent than curare but has less effect on the circulatory system and histamine release. See also: Pancuronium bromide (in salt form). Pancuronium bromide monohydrate (its active ingredient). Drug Indications For muscle relaxation during anesthesia and surgery. Mechanism of Action Non-depolarizing neuromuscular blocking agents inhibit neuromuscular transmission by competing with acetylcholine for cholinergic receptors at the motor endplate, thereby reducing the endplate's response to acetylcholine. This type of neuromuscular blockade can usually be antagonized by anticholinesterase drugs. Low concentrations (5 × 10⁻⁸ g/ml or lower) of pancuronium bromide had no effect on the presynaptic pathway of the phrenic nerve-diaphragm in mice. At high concentrations (5 × 10⁻⁷ g/ml), pancuronium bromide inhibited neurotransmitter release to 26% of the control group in the fibronectin formulation and to 40% of the control group in the high-magnesium formulation. Postsynaptic effects were observed at 5 × 10⁻⁷ g/ml, inhibiting postsynaptic effects to 16% and 22% of the control group, respectively. Pancuronium bromide had no effect on directly induced action potentials and membrane potential constants. Therefore, at paralyzing doses, the presynaptic and postsynaptic effects of pancuronium bromide are crucial for the overall therapeutic efficacy of neuromuscular inhibition. This study investigated the pharmacodynamics of D-tubocurarine (D-TC), pancuronium bromide, metoculin, and galamine in a rat phrenic nerve-hemiadiaphragm model using a vascular perfusion method at 25, 31, and 37 °C. Compared to 37 °C, the ED50 of D-TC, metoculin, and galamine increased nearly twofold at 25 °C. Pancuronium bromide did not show this relationship. The slope of the dose-response curves was not affected by temperature; however, the slopes of metoculin and D-TC were lower than those of pancuronium bromide and galamine. Therefore, in rats, pancuronium bromide retains its potency at low temperatures, while the potency of other muscle relaxants decreases. Furthermore, the slopes of the dose-response curves for metoculin and D-TC were also lower under these experimental conditions.
Therapeutic Uses Non-depolarizing neuromuscular drugs; Nicotine antagonists The primary clinical use of neuromuscular blocking agents is as adjuncts to surgical anesthesia to achieve relaxation of skeletal muscles (especially abdominal wall muscles)...Muscle relaxation is also valuable in various orthopedic surgeries, such as the correction of dislocations and the alignment of fractures. /Neuromuscular Blocking Agents/ ...For patients with cardiovascular disease or bronchial asthma, this product may be safer to use than any other neuromuscular blocking agent. ...It has actually been used to treat status asthmaticus to relax muscles, thereby facilitating artificial respiration and reducing oxygen demand. ...The duration of action at the usual dose is generally 30-60 minutes... Neuromuscular blocking agents/have been used in combination with general anesthetics to assist in laryngoscopy, bronchoscopy, and esophagoscopy. /Neuromuscular Blocking Agents/ For more complete data on the therapeutic uses of pancuronium bromide (9 in total), please visit the HSDB record page. Drug Warnings Neuromuscular blocking agents are potentially dangerous drugs. Therefore, these drugs should only be administered to patients by extensively trained anesthesiologists and other clinicians in facilities equipped with respiratory and cardiovascular resuscitation equipment.Neuromuscular Blocking Agents …This drug should be used with caution in patients with kidney or liver disease. Specific doses of pancuronium bromide may have reduced effects in patients with high plasma globulin levels (e.g., patients with liver disease). Extra caution should be exercised when using muscle relaxants in dehydrated or critically ill patients. /Neuromuscular Blocking Agents/ For more complete data on drug warnings for pancuronium bromide (17 in total), please visit the HSDB records page. Pancuronium bromide is a non-depolarizing neuromuscular blocker with additional anticholinergic (muscarinic receptor antagonist) activity [1,3] - It exerts its neuromuscular blocking effect by inhibiting somatic neuromuscular transmission and affects autonomic function by blocking presynaptic and postsynaptic muscarinic receptors [3] - The drug inhibits sodium channels in sensory neurons, suggesting that it may have an effect on neuronal excitability beyond neuromuscular and cholinergic pathways [2] - Clinically, it is used as an adjunct to anesthesia to induce skeletal muscle relaxation during surgery, its action mediated by a dual action on the neuromuscular junction and muscarinic receptors. [3] |
| Molecular Formula |
C35H60N2O4.2BR
|
|
|---|---|---|
| Molecular Weight |
732.67
|
|
| Exact Mass |
730.292
|
|
| CAS # |
15500-66-0
|
|
| Related CAS # |
16974-53-1 (cation);15500-66-0 (bromide);22189-36-2 (bromide hydrate);
|
|
| PubChem CID |
441289
|
|
| Appearance |
Off-white to pink solid powder
|
|
| Melting Point |
214 - 217ºC
|
|
| LogP |
0.036
|
|
| Hydrogen Bond Donor Count |
0
|
|
| Hydrogen Bond Acceptor Count |
4
|
|
| Rotatable Bond Count |
6
|
|
| Heavy Atom Count |
41
|
|
| Complexity |
1000
|
|
| Defined Atom Stereocenter Count |
10
|
|
| SMILES |
CC(=O)O[C@H]1C[C@@H]2CC[C@@H]3[C@@H]([C@]2(C[C@@H]1[N+]4(CCCCC4)C)C)CC[C@]5([C@H]3C[C@@H]([C@@H]5OC(=O)C)[N+]6(CCCCC6)C)C
|
|
| InChi Key |
NPIJXCQZLFKBMV-YTGGZNJNSA-L
|
|
| InChi Code |
InChI=1S/C35H60N2O4.2BrH/c1-24(38)40-32-21-26-13-14-27-28(35(26,4)23-31(32)37(6)19-11-8-12-20-37)15-16-34(3)29(27)22-30(33(34)41-25(2)39)36(5)17-9-7-10-18-36;;/h26-33H,7-23H2,1-6H3;2*1H/q+2;;/p-2/t26-,27+,28-,29-,30-,31-,32-,33-,34-,35-;;/m0../s1
|
|
| Chemical Name |
1,1-((2S,3S,5S,8R,9S,10S,13S,14S,16S,17R)-3,17-diacetoxy-10,13-dimethylhexadecahydro-1H-cyclopenta[a]phenanthrene-2,16-diyl)bis(1-methylpiperidin-1-ium) bromide
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (3.41 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (3.41 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (3.41 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 100 mg/mL (136.49 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3649 mL | 6.8244 mL | 13.6487 mL | |
| 5 mM | 0.2730 mL | 1.3649 mL | 2.7297 mL | |
| 10 mM | 0.1365 mL | 0.6824 mL | 1.3649 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
 |
|---|
|