
| Size | Price | Stock | Qty |
|---|---|---|---|
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| 5g |
|
||
| Other Sizes |
Purity: =99.05%
Palbociclib HCl (Pfizer trade name Ibrance, also known as PD-0332991), the HCl salt of Palbociclib, is a highly selective, orally bioavailable pyridopyrimidine-derived inhibitor of CDK4/6 with potential antineoplastic activity. In cell-free experiments, it inhibits CDK4 and CDK6 with IC50s of 11 nM and 16 nM, respectively. Many tumor cells overexpress CDK4 and CDK6, and Pfizer's palbociclib is the first CDK4/6 inhibitor to be approved by the FDA as a cancer treatment in 2017. There is no evidence of any activity against PDGFR, EGFR, FGFR, CDK1/2/5, InsR, etc. In vitro, it is a strong anti-proliferative agent that induces an exclusive G1 arrest in Rb-positive tumor cells. It has been shown to cause G1 arrest in primary bone marrow cells and stop tumor growth in disseminated human myeloma xenografts.
| Targets |
DYRK1A (IC50 = 2000 nM); MAPK (IC50 = 8000 nM); Cdk4/cyclin D3 (IC50 = 9 nM); Cdk4/cyclin D1 (IC50 = 11 nM); Cdk6/cyclin D2 (IC50 = 16 nM)
Cyclin-dependent kinase 4 (CDK4) (IC50 = 11 nM, human; complexed with cyclin D1) [1][4] - Cyclin-dependent kinase 6 (CDK6) (IC50 = 16 nM, human; complexed with cyclin D3) [1][4] - No significant affinity for CDK1/2/5/7 (IC50 > 1000 nM) [1][4] |
|---|---|
| ln Vitro |
PD 0332991 has minimal impact on FGFR, PGFR, IR, EGFR, and other protein kinases. PD 0332991 is a Cdk4 non-ATP competitive inhibitor. Because of lowered Rb phosphorylation at Ser780, PD 0332991 inhibits MDA-MB-435 breast carcinoma cells with an IC50 of 66 nM. With IC50 values ranging from 0.04-0.17 μM, PD 0332991 inhibits the thymidine incorporation into the DNA of human leukemias and Rb-positive breast, colon, and lung carcinomas. Rb-negative cells exhibit no activity for PD 0332991. MDA-MB-453 breast and Colo-205 carcinoma cells accumulate cells in G1 in response to PD 0332991. Additionally, PD 0332991 exhibits activity in the immunocompetent model of 5T33MM myeloma cells, making the cells more susceptible to bortezomib-induced cell death.[2] PD 0332991 suppresses MDA-MB-175, ZR-75-30, CAMA-1, MDA-MB-134, HCC-202, and UACC-893, among other luminal ER-positive and HER2-amplified breast cancer cell lines. In these cell lines, PD 0332991 increases the effectiveness of trastuzumab and tamoxifen. In the tamoxifen-resistant MCF7 cells, PD 0332991 increases the sensitivity of tamoxifen.[3] A recent study demonstrates that malignant rhabdoid tumor (MRT) cell lines, such as MP-MRT-AN, KP-MRT-RY, G401, and KP-MRT-NS, can be suppressed by PD 0332991, and that the MRT cell lines' susceptibility to PD 0332991 is inversely connected with p16 expression.[4]
Palbociclib (PD-0332991) HCl is a potent, selective inhibitor of cyclin-dependent kinase 4/6 (CDK4/6) [1][2][3][4] - In human breast cancer cells (MCF-7, T47D) with cyclin D1 overexpression, Palbociclib (0.01-10 μM) dose-dependently inhibited cell proliferation with IC50 values of 0.04 μM and 0.06 μM, inducing G1 phase cell cycle arrest (G1 population increased by 60-70%) and reducing retinoblastoma protein (Rb) phosphorylation by 80% [1][3] - In human non-small cell lung cancer (NSCLC) cells (A549, H1299), Palbociclib (0.1-5 μM) suppressed proliferation with IC50 values of 0.3 μM and 0.5 μM, blocking CDK4/6-mediated Rb-E2F signaling [2] - Combined with letrozole (1 μM) in estrogen receptor-positive (ER+) breast cancer cells (MCF-7), Palbociclib (0.05 μM) synergistically reduced cell viability by 50% compared to single-agent treatment [3] - In CDK4-amplified glioblastoma cells (U87MG), Palbociclib (0.5-5 μM) inhibited colony formation by 75% and downregulated cyclin D1 expression by 45% [4] |
| ln Vivo |
PD 0332991 shows total tumor stasis in an MDA-MB-435 xenograft at 150 mg/kg. Additionally, PD 0332991 exhibits broad-spectrum antitumor activity in multiple human tumor xenografts through the down-regulation of genes under the transcriptional control of E2F and the removal of phospho-Rb and the proliferative marker Ki-67 from tumor tissue. [1]
Multiple myeloma (MM) remains incurable partly because no effective cell cycle-based therapy has been available to both control tumor cell proliferation and synergize with cytotoxic killing. PD 0332991 is an orally active small molecule that potently and specifically inhibits Cdk4 and Cdk6. It has been shown to induce rapid G(1) cell cycle arrest in primary human myeloma cells and suppress tumor growth in xenograft models. To improve therapeutic targeting of myeloma progression, we combined tumor suppression by PD 0332991 with cytotoxic killing by bortezomib, a proteasome inhibitor widely used in myeloma treatment, in the immunocompetent 5T33MM myeloma model. We show that 5T33MM tumor cells proliferate aggressively in vivo due to expression of cyclin D2, elevation of Cdk4, and impaired p27(Kip1) expression, despite inhibition of Cdk4/6 by p18(INK4c) and the maintenance of a normal plasma cell transcription program. PD 0332991 potently inhibits Cdk4/6-specific phosphorylation of Rb and cell cycle progression through G(1) in aggressively proliferating primary 5T33MM cells, in vivo and ex vivo. This leads to tumor suppression and a significant improvement in survival. Moreover, induction of G(1) arrest by PD 0332991 sensitizes 5T33MM tumor cells to killing by bortezomib. Inhibition of Cdk4/6 by PD 0332991, therefore, effectively controls myeloma tumor expansion and sensitizes tumor cells to bortezomib killing in the presence of an intact immune system, thereby representing a novel and promising cell cycle-based combination therapy.[2] In nude mice bearing MCF-7 ER+ breast cancer xenografts, oral Palbociclib (50 mg/kg/day for 28 days) reduced tumor volume by 65% and increased Rb hypophosphorylation in tumor tissues by 70% [1][3] - In mice with T47D breast cancer xenografts, oral Palbociclib (75 mg/kg/day) combined with letrozole (1 mg/kg/day) inhibited tumor growth by 80%, compared to 40% with letrozole alone [3] - In NSCLC A549 xenograft mice, intraperitoneal Palbociclib (30 mg/kg/day for 21 days) prolonged median survival by 40% and reduced tumor microvessel density by 35% [2] - It did not cause significant weight loss or systemic toxicity in xenograft models at therapeutic doses [1][3] |
| Enzyme Assay |
CDK assays are run in 96-well filter plates for kinetic analysis and IC50 calculations. By infecting insect cells with baculovirus, all CDK-cyclin kinase complexes are expressed and purified. A portion of pRb fused to GST (amino acids 792–928) serves as the substrate for the assays (GST•RB-Cterm). 20 mM Tris-HCl, pH 7.4, 50 mM NaCl, 1 mM dithiothreitol, 10 mM MgCl2, 25 μM ATP (for CDK4-cyclin D1, CDK6-cyclin D2, and CDK6-cyclin D3), 0.25 μCi of [γ-32P]ATP, 20 ng of enzyme, 1 μg of GST•RB-Cterm, and suitable dilutions of inhibitor are included in the overall reaction volume of 0.1 mL. After adding all the ingredients to the wells—aside from the [γ-32P]ATP—they are put on a plate mixer for two minutes. Addition of [γ-32P]ATP initiates the reaction, which is then incubated for 15 minutes at 25°C. In order to allow the substrate to precipitate, the reaction is stopped by adding 0.1 mL of 20% trichloroacetic acid and keeping the plate at 4°C for at least an hour. After five well washes with 0.2 mL of 10% trichloroacetic acid, radioactive incorporation is measured using a β plate counter.
CDK4/6 kinase activity assay: Recombinant human CDK4/cyclin D1 and CDK6/cyclin D3 complexes were individually incubated with [γ-³²P]-ATP, Rb-derived peptide substrate, and Palbociclib (0.001-1000 nM) at 30°C for 60 minutes. Phosphorylated substrates were separated by filtration and quantified by scintillation counting to calculate IC50 values [1][4] - CDK selectivity assay: Recombinant human CDK1/cyclin B, CDK2/cyclin E, CDK5/p25 complexes were incubated with Palbociclib (0.01-100 μM) under the same conditions as CDK4/6 assay to assess off-target inhibition [1][4] |
| Cell Assay |
In a 96-well plate, 2 × 10 4 cells are seeded per well and incubated for a full night. After being added to the wells, PD 0332991 (0.01-1 μM) is incubated for an additional 24 hours at 37 °C. [ 14 C]Thymidine (0.1 μCi) is added to each well and incorporation of the radiolabel is allowed to proceed for 72 hours. Using a β plate counter, one can determine the incorporerated radioactivity.
Forty-seven human breast cancer and immortalized cell lines representing the known molecular subgroups of breast cancer were treated with PD 0332991 to determine IC50 values. These data were analyzed against baseline gene expression data to identify genes associated with PD 0332991 response. Results: Cell lines representing luminal estrogen receptor-positive (ER+) subtype (including those that are HER2 amplified) were most sensitive to growth inhibition by PD 0332991 while nonluminal/basal subtypes were most resistant. Analysis of variance identified 450 differentially expressed genes between sensitive and resistant cells. pRb and cyclin D1 were elevated and CDKN2A (p16) was decreased in the most sensitive lines. Cell cycle analysis showed G0/G1 arrest in sensitive cell lines and Western blot analysis demonstrated that Rb phosphorylation is blocked in sensitive lines but not resistant lines. PD 0332991 was synergistic with tamoxifen and trastuzumab in ER+ and HER2-amplified cell lines, respectively. PD 0332991 enhanced sensitivity to tamoxifen in cell lines with conditioned resistance to ER blockade. Conclusions: These studies suggest a role for CDK4/6 inhibition in some breast cancers and identify criteria for patient selection in clinical studies of PD 0332991[3]. Malignant rhabdoid tumor (MRT) is a rare and highly aggressive neoplasm of young children. MRT is characterized by inactivation of integrase interactor 1 (INI1). Cyclin-dependent kinase 4 (CDK4), which acts downstream of INI1, is required for the proliferation of MRT cells. Here we investigated the effects of PD 0332991 (PD), a potent inhibitor of CDK4, against five human MRT cell lines (MP-MRT-AN, KP-MRT-RY, G401, KP-MRT-NS, KP-MRT-YM). In all of the cell lines except KP-MRT-YM, PD inhibited cell proliferation >50%, (IC(50) values 0.01 to 0.6 μM) by WST-8 assay, and induced G1-phase cell cycle arrest, as shown by flow cytometry and BrdU incorporation assay. The sensitivity of the MRT cell lines to PD was inversely correlated with p16 expression (r=0.951). KP-MRT-YM cells overexpress p16 and were resistant to the growth inhibitory effect of PD. Small interfering RNA against p16 significantly increased the sensitivity of KP-MRT-YM cells to PD (p<0.05). These results suggest that p16 expression in MRT could be used to predict its sensitivity to PD. PD may be an attractive agent for patients with MRT whose tumors express low levels of p16 [4]. Breast cancer cell proliferation assay: MCF-7/T47D cells were seeded in 96-well plates, treated with Palbociclib (0.001-50 μM) alone or combined with letrozole (1 μM) for 72 hours. Cell viability was measured by MTT assay, and IC50 values were calculated [1][3] - Cell cycle analysis: MCF-7 cells were treated with Palbociclib (0.1 μM) for 24 hours, stained with propidium iodide, and cell cycle distribution was analyzed by flow cytometry [1][3] - Rb phosphorylation assay: A549 cells were treated with Palbociclib (0.1-5 μM) for 12 hours. Rb phosphorylation (Ser780) was detected by Western blot and quantified [2] - Colony formation assay: U87MG cells were seeded in 6-well plates, treated with Palbociclib (0.5-5 μM) for 14 days. Colonies were stained with crystal violet and counted to assess clonogenic potential [4] |
| Animal Protocol |
Advanced stage human tumor xenografts including Colo-205, MDA-MB-435 breast, SF-295 glioblastoma, ZR-75-1 breast, PC-3 prostate, H125 lung, SW-620 colon, H23 lung and MDA-MB-468 breast (Rb negative) are established in severe combined immunodeficient mice.
0-150 mg/kg Given by gavage Multiple myeloma (MM) remains incurable partly because no effective cell cycle-based therapy has been available to both control tumor cell proliferation and synergize with cytotoxic killing. PD 0332991 is an orally active small molecule that potently and specifically inhibits Cdk4 and Cdk6. It has been shown to induce rapid G(1) cell cycle arrest in primary human myeloma cells and suppress tumor growth in xenograft models. To improve therapeutic targeting of myeloma progression, we combined tumor suppression by PD 0332991 with cytotoxic killing by bortezomib, a proteasome inhibitor widely used in myeloma treatment, in the immunocompetent 5T33MM myeloma model. We show that 5T33MM tumor cells proliferate aggressively in vivo due to expression of cyclin D2, elevation of Cdk4, and impaired p27(Kip1) expression, despite inhibition of Cdk4/6 by p18(INK4c) and the maintenance of a normal plasma cell transcription program. PD 0332991 potently inhibits Cdk4/6-specific phosphorylation of Rb and cell cycle progression through G(1) in aggressively proliferating primary 5T33MM cells, in vivo and ex vivo. This leads to tumor suppression and a significant improvement in survival. Moreover, induction of G(1) arrest by PD 0332991 sensitizes 5T33MM tumor cells to killing by bortezomib. Inhibition of Cdk4/6 by PD 0332991, therefore, effectively controls myeloma tumor expansion and sensitizes tumor cells to bortezomib killing in the presence of an intact immune system, thereby representing a novel and promising cell cycle-based combination therapy.[2] MCF-7 ER+ breast cancer xenograft model: Female nude mice (18-22 g) were subcutaneously inoculated with MCF-7 cells (5×10⁶ cells/mouse). When tumors reached 100 mm³, Palbociclib suspended in 0.5% CMC-Na was administered orally at 25, 50, 75 mg/kg/day for 28 days. Tumor volume and Rb phosphorylation were evaluated [1][3] - T47D breast cancer combination therapy model: Female nude mice (18-22 g) were subcutaneously inoculated with T47D cells (4×10⁶ cells/mouse). Tumor-bearing mice received oral Palbociclib (75 mg/kg/day) plus oral letrozole (1 mg/kg/day) for 21 days. Tumor growth inhibition was monitored [3] - A549 NSCLC xenograft model: Male nude mice (20-25 g) were subcutaneously inoculated with A549 cells (3×10⁶ cells/mouse). Palbociclib dissolved in saline was administered intraperitoneally at 30 mg/kg/day for 21 days. Survival time and tumor microvessel density were measured [2] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Palbociclib exhibits linear pharmacokinetics, reaching peak plasma concentrations 6–12 hours after oral administration. Its reported oral bioavailability is 46%, reaching steady state after 8 days, with a median cumulative ratio of 2.4. Absorption of palbociclib is significantly reduced on an empty stomach; therefore, it is recommended to take this medication with food. The primary route of elimination for palbociclib is through hepatic metabolism and excretion in feces, while renal clearance is minimal, accounting for only 17.5% of the eliminated dose. The mean apparent volume of distribution for palbociclib is 2583 L, indicating its extensive penetration into peripheral tissues. The mean apparent oral clearance of palbociclib is 63.1 L/h. Metabolism/Metabolites Palbociclib is primarily metabolized in the liver. Its metabolism is mainly catalyzed by cytochrome P450 isoenzyme 3A and sulfotransferase 2A1. Palbociclib is primarily metabolized through oxidation and sulfonation, with acylation and glucuronidation being minor reactions. After metabolism, palbociclib mainly forms inactive glucuronide and aminosulfonic acid conjugates. The main circulating metabolite is the glucuronide conjugate, accounting for 1.5% of the excreted dose. Biological Half-Life The mean plasma elimination half-life of palbociclib is 29 hours. Oral bioavailability: Approximately 46% in humans; approximately 55% in rats after oral administration of 50 mg/kg[1][3] -Elimination half-life: 24.3 hours in humans; 11.2 hours in rats[1] -Plasma protein binding rate: 93.8% in human plasma (concentration range: 0.1-10 μg/mL)[1] -Distribution: Volume of distribution (Vd) in rats = 2.4 L/kg, widely distributed in tumor tissues and peripheral tissues[1][2] -Metabolism: Mainly metabolized in the liver by CYP3A4 into inactive metabolites[1] -Excretion: 74% of the dose is excreted in feces as metabolites; 15% is excreted in urine; <2% is excreted unchanged[1] |
| Toxicity/Toxicokinetics |
Hepatotoxicity
Adverse events are relatively common in large clinical trials, leading to dose reductions in one-third of patients and discontinuation of treatment in 8%. Literature on the efficacy and safety of palbociclib rarely mentions elevated serum ALT or hepatotoxicity. In a study of women with refractory metastatic breast cancer, 6% of patients receiving palbociclib in combination with fulvestrant experienced elevated serum ALT (2% exceeding 5 times the upper limit of normal), compared to 3% in patients treated with fulvestrant alone (none exceeding 5 times the upper limit of normal). Since palbociclib's approval and widespread use, several reports have shown significant ALT elevations after 2 or 3 cycles of treatment, with improvement upon discontinuation but rapid relapse upon restarting. These patients had normal serum bilirubin and alkaline phosphatase levels and did not report any related symptoms. In addition, rare case reports have shown that patients with refractory metastatic breast cancer developed pseudocirrhosis 2 to 3 months after starting palbociclib, presenting with fatigue, jaundice, and ascites, with only mild elevations in serum transaminase and alkaline phosphatase levels. Imaging revealed severe hepatic nodules, but histological examination showed profibrotic changes in the necrotic metastatic areas without cirrhosis. Vascular changes were also present in the liver, suggesting hepatic sinusoidal obstruction syndrome, which may be due to the combined effects of rapid shrinkage of metastatic tissue and vascular damage. Pseudocirrhosis has also been reported with other highly effective antitumor therapies for liver metastases, but this is relatively rare. Probability Score: C (Possibly a rare cause of clinically significant liver injury, manifesting as pseudocirrhosis due to nodular transformation of the liver following necrosis of liver metastases). Use during pregnancy and lactation> ◉ Overview of use during lactation There is currently no information regarding the clinical use of palbociclib during lactation. Because palbociclib binds to plasma proteins at a rate of 85%, its concentration in breast milk may be low. However, its half-life is approximately 29 hours, which may allow it to accumulate in the infant. Furthermore, palbociclib is used in combination with letrozole or fulvestrant, which may increase the risk to the infant. The manufacturer recommends discontinuing breastfeeding during palbociclib treatment and for 3 weeks after the last dose. ◉ Effects on breastfed infants As of the revision date, no relevant published information was found. ◉ Effects on lactation and breast milk As of the revision date, no relevant published information was found. Protein binding Palbociclib binds to human plasma proteins at approximately 85% of the administered dose in vitro. Acute toxicity: oral LD50 in rats > 600 mg/kg; in mice > 500 mg/kg [1] - Subchronic toxicity (oral administration in rats over 28 days): no significant hepatotoxicity or nephrotoxicity was observed at doses up to 75 mg/kg/day; mild neutropenia (neutrophil count decrease ≤15%) occurred at a daily dose of 100 mg/kg [1] - In xenograft mice, no significant changes were observed in serum creatinine, BUN, ALT/AST, or body weight at therapeutic doses (25-75 mg/kg/day) [1][3] - Drug interactions: potent CYP3A4 inhibitors (e.g., ketoconazole) can inhibit this product, increasing AUC by 2.0-fold; no interaction with letrozole or tamoxifen [3] |
| References | |
| Additional Infomation |
Malignant rhabdomyosarcoma (MRT) is a rare and highly aggressive childhood tumor. MRT is characterized by inactivation of integrase-interacting protein 1 (INI1). Cyclin-dependent kinase 4 (CDK4), located downstream of INI1, is essential for MRT cell proliferation. This study investigated the effects of the potent CDK4 inhibitor PD 0332991 (PD) on five human MRT cell lines (MP-MRT-AN, KP-MRT-RY, G401, KP-MRT-NS, and KP-MRT-YM). Except for the KP-MRT-YM cell line, PD inhibited cell proliferation by more than 50% in all cell lines (IC50 values ranging from 0.01 to 0.6 μM as determined by WST-8 assay) and induced G1 phase cell cycle arrest (flow cytometry and BrdU incorporation assays). The sensitivity of MRT cell lines to PD was negatively correlated with p16 expression (r = 0.951). KP-MRT-YM cells overexpressed p16 and were resistant to the growth-inhibiting effect of PD. Small interfering RNA targeting p16 significantly increased the sensitivity of KP-MRT-YM cells to PD (p<0.05). These results suggest that p16 expression in MRT can be used to predict its sensitivity to PD. PD may be an attractive therapeutic agent for MRT patients with low p16 expression levels in their tumors. [4]
Cell lines representing luminal estrogen receptor-positive (ER+) subtypes (including HER2 amplified subtypes) were most sensitive to the growth-inhibiting effect of PD 0332991, while non-luminal/basal subtypes were the most resistant. Analysis of variance identified 450 differentially expressed genes between sensitive and resistant cells. In the most sensitive cell lines, pRb and cyclin D1 levels were elevated, while CDKN2A (p16) levels were decreased. Cell cycle analysis showed G0/G1 phase arrest in sensitive cell lines. Western blot analysis showed that Rb phosphorylation was blocked in sensitive cell lines but not in resistant cell lines. PD 0332991 had a synergistic effect with tamoxifen and trastuzumab in ER+ and HER2 amplified cell lines, respectively. PD 0332991 enhanced the sensitivity of tamoxifen in cell lines with conditional resistance to ER blockade. [3] Palbociclib belongs to the pyridopyrimidine class of compounds. Its chemical name is 2-{[5-(piperazin-1-yl)pyridin-2-yl]amino}pyrido[2,3-d]pyrimidin-7-one, with methyl, acetyl and cyclopentyl substituents at positions 5, 6 and 8, respectively. It is used in combination with letrozole to treat metastatic breast cancer. It is an EC 2.7.11.22 (cyclin-dependent kinase) inhibitor and an anti-tumor drug. It is a pyridopyrimidine compound, belonging to the aminopyridine, secondary amino, piperidine, aromatic ketone, cyclopentane, and tertiary amino groups. Palbociclib is a piperazine-based pyridopyrimidine compound that acts on cell cycle regulation mechanisms. It is a second-generation cyclin-dependent kinase inhibitor, selected from pyridopyrimidine compounds due to its superior physical and pharmaceutical properties. Palbociclib was developed by Pfizer after the discovery that cyclin-dependent kinases are key regulators of cell growth. It was initially approved by the FDA in March 2015 for the treatment of HR-positive, HER2-negative advanced or metastatic breast cancer. In April 2019, based on post-marketing reports and data confirming its safety and clinical efficacy in electronic health records, its indication was updated to include male patients. Palbociclib is a kinase inhibitor. Palbociclib's mechanism of action is as a kinase inhibitor and a cytochrome P450 3A inhibitor. Palbociclib is a unique cyclin-dependent kinase inhibitor used in combination with aromatase inhibitors to treat postmenopausal women with metastatic breast cancer. Palbociclib treatment can cause transient and usually mild elevations in serum transaminases and may lead to a rare form of liver injury called pseudocirrhosis. Pseudocirrhosis is caused by shrinkage of liver tumor metastases accompanied by profibrotic changes and vascular damage; it can be severe, rapidly progressive, and even fatal. Palbociclib is an orally administered cyclin-dependent kinase (CDK) inhibitor with potential antitumor activity. Palbociclib selectively inhibits cyclin-dependent kinases 4 (CDK4) and 6 (CDK6), thereby inhibiting phosphorylation of proteins in early G1 phase retinoblastoma (Rb) cells, leading to cell cycle arrest. This inhibits DNA replication and reduces tumor cell proliferation. CDK4 and CDK6 are serine/threonine kinases that are upregulated in various tumor cell types and play a key role in the regulation of cell cycle progression. See also: Palbociclib isothiocyanate (its active ingredient). Drug Indications Palbociclib, in combination with letrozole, is indicated as initial endocrine therapy for adult patients with advanced/metastatic breast cancer who have HER2-negative, hormone receptor (HR)-positive tumors. It is also approved in combination with fulvestrant for patients whose disease has progressed after prior endocrine therapy. According to the official labeling, palbociclib should be used in combination with an aromatase inhibitor (not limited to letrozole) as initial endocrine therapy for postmenopausal women or men. Breast cancer begins with a cluster of cancer cells that invade and destroy nearby breast tissue. This growth can spread to other parts of the body, a process known as metastasis. Depending on the location of the cancer cells, breast cancer can be classified as ductal carcinoma or lobular carcinoma. In addition, other types of breast cancer include inflammatory breast cancer, Paget's disease of the breast, triple-negative breast cancer, non-Hodgkin's lymphoma, and soft tissue sarcoma. Treatment for male breast cancer is generally the same as for breast cancer in postmenopausal women, and almost all cases are ductal carcinoma. FDA Label Ibrance is indicated for the treatment of hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative locally advanced or metastatic breast cancer: in combination with aromatase inhibitors; in combination with fulvestrant for women who have previously received endocrine therapy. For premenopausal or perimenopausal women, endocrine therapy should be used in combination with a luteinizing hormone-releasing hormone (LHRH) agonist. Treatment of Ewing's sarcoma Treatment of breast malignancies Mechanism of Action Palbociclib is a cyclin-dependent kinase 4/6 (CDK4/6) inhibitor whose mechanism of action is through binding to the ATP-binding pocket, with an IC50 value in the range of 9-15 nmol/L. Notably, it exhibits low or negligible activity against other kinases. CDK4/6 kinases, along with their co-regulatory partner cyclin D, participate in the G1-S phase transition. Therefore, inhibiting this step can halt cell cycle progression in cells where this pathway functions. This step involves the retinoblastoma protein phosphorylation pathway and the E2F transcription factor family. Palbociclib (PD-0332991) hydrochloride is a potent, selective CDK4/6 inhibitor used to treat hormone receptor-positive (HR+) and human epidermal growth factor receptor 2-negative (HER2-) breast cancer [1][3]. Its core mechanism includes inhibiting the CDK4/6-cyclin D complex, blocking Rb phosphorylation, inducing G1 phase cell cycle arrest, and inhibiting tumor cell proliferation [1][2][4]. Its therapeutic applications include first-line treatment of advanced HR+/HER2- breast cancer (in combination with aromatase inhibitors) and metastatic breast cancer (in combination with fulvestrant) [3]. It exhibits synergistic antitumor effects with endocrine therapy (letrozole, fulvestrant) by overcoming endocrine resistance in breast cancer [3]. Good oral bioavailability, long elimination half-life, and tumor-specific distribution support its use in chronic cancer treatment and combination therapy regimens [1][3]. |
| Molecular Formula |
C24H29N7O2.HCL
|
|---|---|
| Molecular Weight |
483.99
|
| Exact Mass |
483.214
|
| Elemental Analysis |
C, 59.56; H, 6.25; Cl, 7.33; N, 20.26; O, 6.61
|
| CAS # |
827022-32-2
|
| Related CAS # |
Palbociclib;571190-30-2;Palbociclib hydrochloride;571189-11-2;Palbociclib-d8;1628752-83-9;Palbociclib isethionate;827022-33-3;Palbociclib dihydrochloride;Palbociclib orotate;2757498-64-7
|
| PubChem CID |
11431660
|
| Appearance |
Yellow solid powder
|
| Boiling Point |
727ºC at 760 mmHg
|
| Flash Point |
393.5ºC
|
| LogP |
4.234
|
| Hydrogen Bond Donor Count |
3
|
| Hydrogen Bond Acceptor Count |
8
|
| Rotatable Bond Count |
5
|
| Heavy Atom Count |
34
|
| Complexity |
775
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
Cl.O=C1N(C2CCCC2)C2C(=CN=C(NC3C=CC(N4CCNCC4)=CN=3)N=2)C(C)=C1C(C)=O
|
| InChi Key |
STEQOHNDWONVIF-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C24H29N7O2.ClH/c1-15-19-14-27-24(28-20-8-7-18(13-26-20)30-11-9-25-10-12-30)29-22(19)31(17-5-3-4-6-17)23(33)21(15)16(2)32;/h7-8,13-14,17,25H,3-6,9-12H2,1-2H3,(H,26,27,28,29);1H
|
| Chemical Name |
6-acetyl-8-cyclopentyl-5-methyl-2-[(5-piperazin-1-ylpyridin-2-yl)amino]pyrido[2,3-d]pyrimidin-7-one;hydrochloride
|
| Synonyms |
Palbociclib; PD-332991; PD332991; PD 332991; PD0332991; PD-0332991; PD0332991 HCl, PD-0332991 hydrochloride; Palbociclib HCl; Palbociclib (hydrochloride); PD 0332991 HCl; Palbociclib (PD-0332991) HCl; PD-0332991 hydrochloride; BKC4F3Q5XL; PD 0332991; Palbociclib HCl; Trade name: Ibrance
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 0.54 mg/mL (1.12 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.4 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 0.54 mg/mL (1.12 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 5.4 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: Saline: 20 mg/mL Solubility in Formulation 4: 20 mg/mL (41.32 mM) in 0.5%HPMC 1%Tween80 (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. Solubility in Formulation 5: 4.17 mg/mL (8.62 mM) in Lactic acid buffer (50 mM, pH 4.0) (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0662 mL | 10.3308 mL | 20.6616 mL | |
| 5 mM | 0.4132 mL | 2.0662 mL | 4.1323 mL | |
| 10 mM | 0.2066 mL | 1.0331 mL | 2.0662 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT06003114 | Active Recruiting |
Drug: Palbociclib | Breast Cancer | Pfizer | September 2015 | |
| NCT03936270 | Active Recruiting |
Drug: Palbociclib 125mg Drug: Letrozole 2.5mg |
Ovarian Cancer | Latin American Cooperative Oncology Group |
January 27, 2020 | Phase 2 |
| NCT02738866 | Active Recruiting |
Drug: Palbociclib Drug: Fulvestrant |
Metastatic Breast Cancer | Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins |
October 25, 2016 | Phase 2 |
| NCT05069038 | Recruiting | Drug: Palbociclib 125mg | Breast Cancer | University of Nebraska | March 2, 2022 | Phase 2 |
| NCT04360941 | Recruiting | Drug: Palbociclib Drug: Avelumab |
ER+ Breast Cancer Recurrent Breast Cancer |
Royal Marsden NHS Foundation Trust |
August 11, 2020 | Phase 1 |
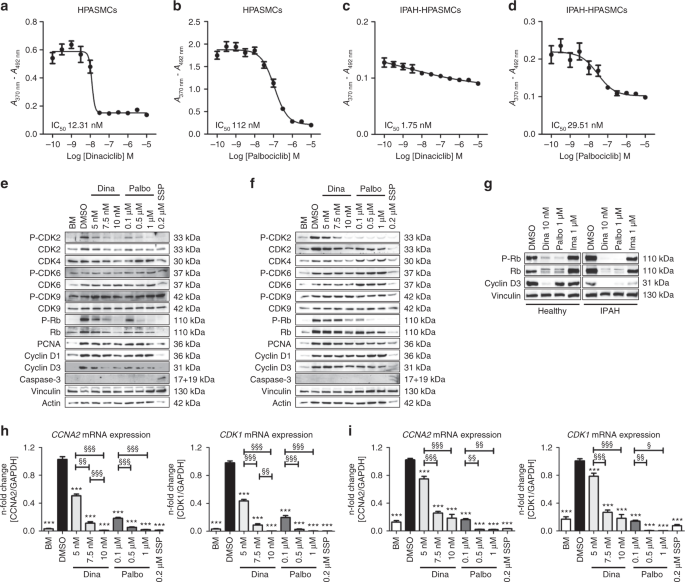 Evaluation of IC50concentrations of the CDK inhibitors dinaciclib and palbociclib on proliferation, and their effects on CDK-Rb-E2F signaling in human HPASMCs from healthy donors and IPAH patients.Nat Commun.2019May 17;10(1):2204. |
|---|
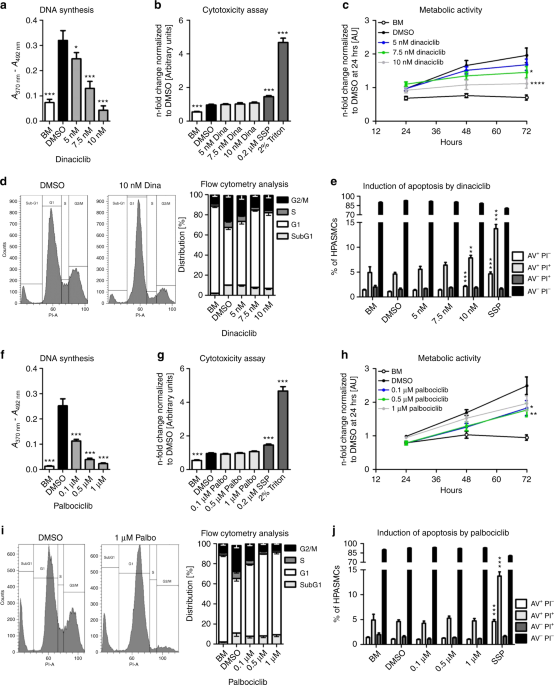 Effects of the CDK inhibitors dinaciclib and palbociclib on proliferation, cell cycle, and apoptosis.Nat Commun.2019May 17;10(1):2204. |
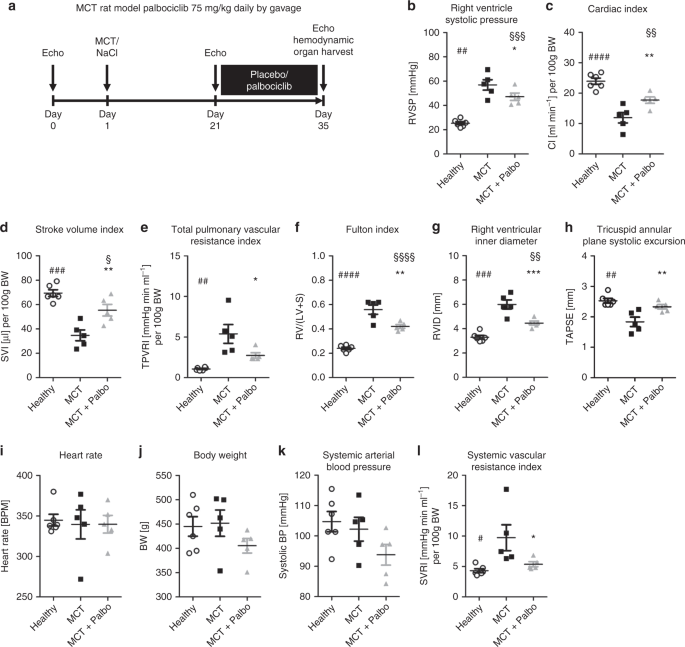 Effects of palbociclib on disease progression in the MCT rat model of pulmonary arterial hypertension.Nat Commun.2019May 17;10(1):2204. |
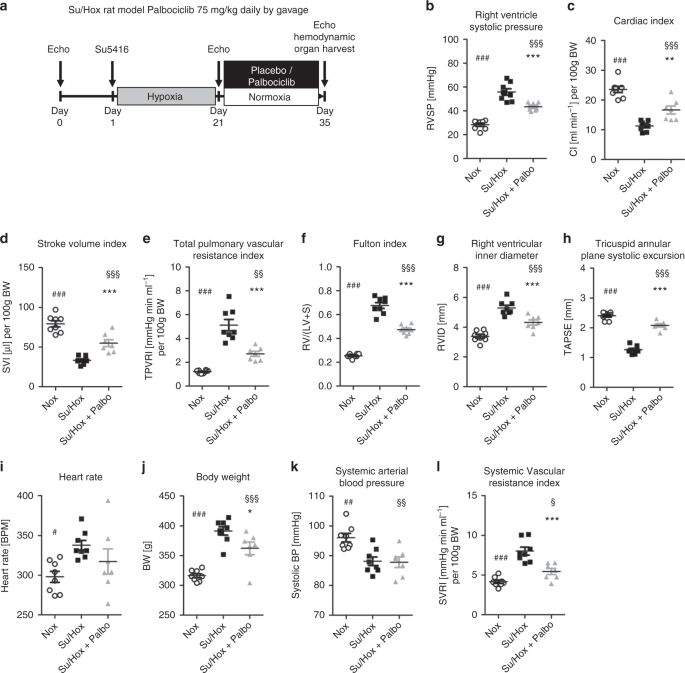 Effects of palbociclib on disease progression in the Su/Hox rat model of pulmonary arterial hypertension.Nat Commun.2019May 17;10(1):2204. |
|---|
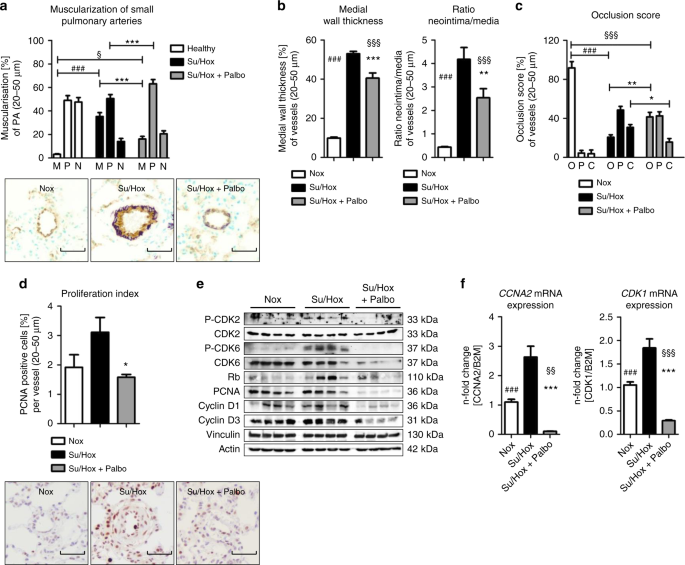 Ex vivo analyses of lung tissue for reversal of remodeling and in vivo drug efficacy in the Su/Hox model.Nat Commun.2019May 17;10(1):2204. |
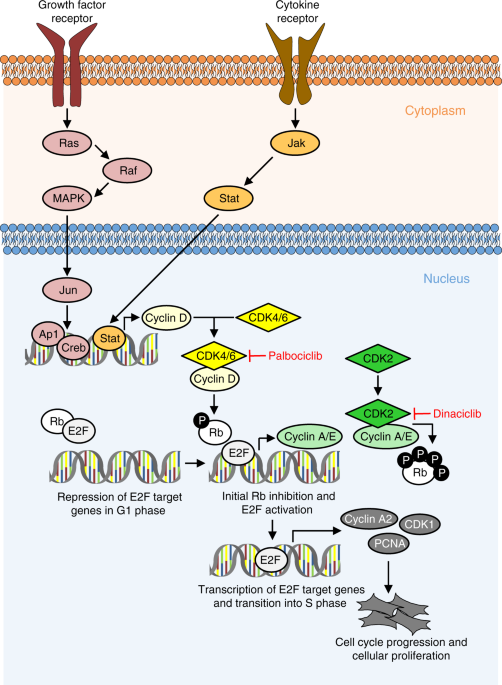 Proposed mechanism of action of palbociclib and dinaciclib in PAH. Multiple growth factors, cytokines, and mitogens induce the activation of cyclin-dependent kinases (CDKs), e.g., by increasing the expression of cyclin D1.Nat Commun.2019May 17;10(1):2204. |
|
|---|
|
|