
| Size | Price | Stock | Qty |
|---|---|---|---|
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
Purity: ≥98%
Olopatadine HCl (Pataday; Patanase; KW-4679; ALO-4943A; ALO4943A; KW4679), the hydrochloride salt of olopatadine, is a 2nd-generation, selective histamine H1 receptor (IC50 of 559 μM for the release of histamine) antagonist and also a mast cell stabilizer used to treat itching associated with allergic conjunctivitis (eye allergies), it may also be used for the treatment of steroid rebound (red skin syndrome).
| Targets |
Histamine receptor
Histamine H1 receptor (H1R) (human H1R, Ki=0.16 nM; rat H1R, Ki=0.25 nM; guinea pig H1R, Ki=0.31 nM) [2,3] |
|---|---|
| ln Vitro |
In vitro activity: Olopatadine is one of the second-generation histamine H1 receptor antagonists that are treated for allergic disorders. Olopatadine significantly inhibited the ear swelling and the increased production of IL-4, IL-1beta, IL-6, GM-CSF and NGF in the lesioned ear. Olopatadine was highly and rapidly absorbed in healthy human volunteers. The urinary excretion of olopatadine accounted for not less than 58% and the contribution of metabolism was considerably low in the clearance of olopatadine in humans. Olopatadine is one of the few renal clearance drugs in antiallergic drugs. Olopatadine was shown to be useful for the treatment of allergic rhinitis and chronic urticaria in double-blind clinical trials. AL-4943A inhibits histamine release in a concentration-dependent fashion (IC50 = 559 microM) from human conjunctival mast cell preparations in vitro. Passive anaphylaxis in guinea pig conjunctiva was attenuated by AL-4943A applied 30 min prior to intravenous or topical ocular antigen challenge (ED50 values 0.0067% and 0.0170%, w/v, respectively).
Radioligand binding assay with human/rat/guinea pig H1R-expressing cell membranes showed Olopatadine HCl (ALO4943A; KW4679) competitively bound to H1R with high affinity, displacing [3H]-pyrilamine in a concentration-dependent manner [2,3] - Rat peritoneal mast cells activated with compound 48/80 (1 μg/mL) were treated with Olopatadine HCl (ALO4943A; KW4679) (0.1 μM-10 μM). It dose-dependently inhibited histamine release, with 68% inhibition at 10 μM and IC50=0.8 μM [2] - Human conjunctival epithelial cells stimulated with TNF-α (10 ng/mL) were treated with Olopatadine HCl (ALO4943A; KW4679) (1 μM-50 μM). At 20 μM, it reduced IL-6 and IL-8 secretion by 55% and 62% respectively, via inhibiting NF-κB activation [3] - Mouse skin fibroblasts treated with oxazolone (100 μM) to induce inflammatory responses were pre-treated with Olopatadine HCl (ALO4943A; KW4679) (5 μM-50 μM). 30 μM concentration inhibited cyclooxygenase-2 (COX-2) mRNA expression by 48% and prostaglandin E2 (PGE2) production by 53% [1] |
| ln Vivo |
Oxazolone-induced contact dermatitis model in mice: Topical application of Olopatadine HCl (ALO4943A; KW4679) (0.1%, 0.3%, 1% cream) twice daily for 7 days dose-dependently reduced skin erythema, edema, and thickness. 1% cream reduced skin thickness by 65% and inflammatory cell infiltration (eosinophils, neutrophils) by 58% compared to vehicle [1]
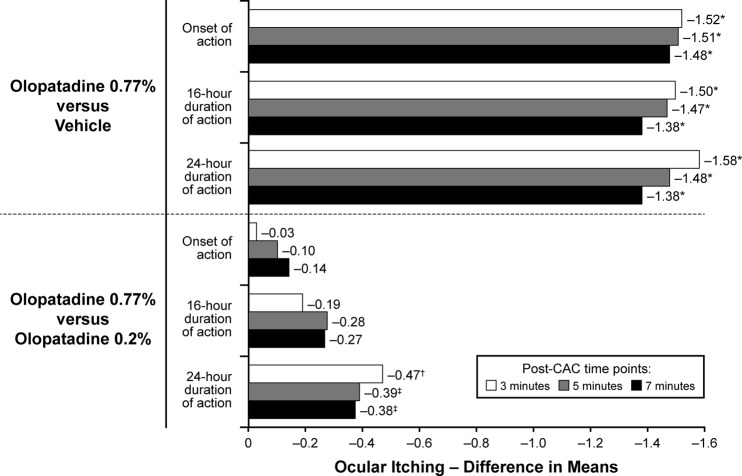
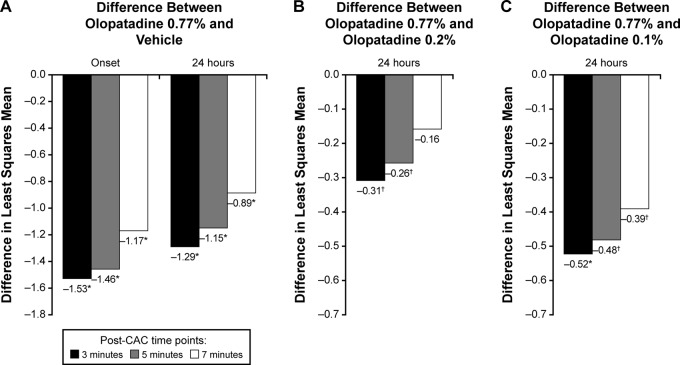
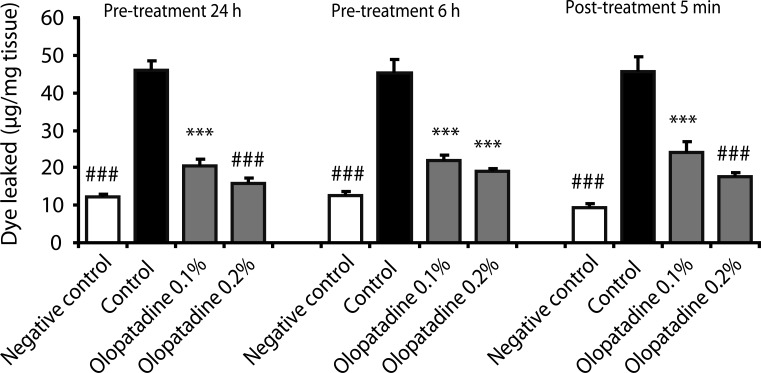
- Experimental allergic conjunctivitis (EAC) model in rats: Topical ocular administration of Olopatadine HCl (ALO4943A; KW4679) (0.1%, 0.5% eye drops) twice daily for 5 days inhibited conjunctival redness, itching, and eosinophil recruitment by 45-72% in a dose-dependent manner. The effect lasted for 8 hours post-administration [3] - Clinical trial in patients with allergic conjunctivitis: Topical Olopatadine HCl (ALO4943A; KW4679) (0.1% eye drops) twice daily for 2 weeks reduced ocular symptoms (itching, redness, tearing) by 70% compared to placebo. [2] |
| Enzyme Assay |
H1R binding assay: Prepare membrane fractions from HEK293 cells expressing human/rat/guinea pig H1R or animal brain tissue. Incubate membranes with [3H]-pyrilamine (0.5 nM) and various concentrations of Olopatadine HCl (ALO4943A; KW4679) (0.01 nM-100 nM) at 25°C for 60 minutes. Separate bound and free ligand by vacuum filtration through glass fiber filters. Measure radioactivity with a liquid scintillation counter and calculate Ki values using the Cheng-Prusoff equation [2,3]
|
| Cell Assay |
Mast cell histamine release assay: Isolate rat peritoneal mast cells by peritoneal lavage. Resuspend cells in culture medium and pre-treat with Olopatadine HCl (ALO4943A; KW4679) (0.1 μM-10 μM) for 15 minutes. Stimulate with compound 48/80 (1 μg/mL) for 30 minutes. Centrifuge to collect supernatant and measure histamine concentration via fluorometric assay [2]
- Conjunctival epithelial cell cytokine assay: Seed human conjunctival epithelial cells in 24-well plates and incubate until 80% confluent. Pre-treat with Olopatadine HCl (ALO4943A; KW4679) (1 μM-50 μM) for 1 hour, then stimulate with TNF-α (10 ng/mL) for 24 hours. Collect supernatant and quantify IL-6/IL-8 via ELISA; extract nuclear protein to detect NF-κB p65 activation via Western blot [3] - Skin fibroblast inflammatory assay: Seed mouse skin fibroblasts in 6-well plates and incubate for 24 hours. Pre-treat with Olopatadine HCl (ALO4943A; KW4679) (5 μM-50 μM) for 1 hour, then stimulate with oxazolone (100 μM) for 18 hours. Extract total RNA to detect COX-2 mRNA via RT-PCR; measure PGE2 in supernatant via ELISA [1] |
| Animal Protocol |
Contact dermatitis mouse model: Female BALB/c mice (20-25 g) were sensitized with 1% oxazolone on the abdomen on day 0. On day 7, challenge with 0.5% oxazolone on the ears to induce dermatitis. Olopatadine HCl (ALO4943A; KW4679) cream (0.1%, 0.3%, 1%) was topically applied to the ears twice daily for 7 days. Measure ear thickness daily; collect ear tissues for histopathological analysis and inflammatory cell counting [1]
- EAC rat model: Male Wistar rats (150-200 g) were sensitized with ovalbumin (100 μg) + aluminum hydroxide (2 mg) via intraperitoneal injection on days 0 and 7. On day 14, topical ocular ovalbumin (1% solution) was administered to induce EAC. Olopatadine HCl (ALO4943A; KW4679) eye drops (0.1%, 0.5%) were applied twice daily for 5 days. Score conjunctival symptoms (redness, itching) and count eosinophils in conjunctival tissues [3] - Pharmacokinetic rat experiment: Male Sprague-Dawley rats (200-250 g) were fasted for 12 hours. Olopatadine HCl (ALO4943A; KW4679) was administered via oral gavage (10 mg/kg) or intravenous injection (2 mg/kg). Blood samples were collected at predetermined time points, and plasma drug concentrations were determined by HPLC [2] |
| ADME/Pharmacokinetics |
Absorption: The oral bioavailability in the human body is 60-65%; the peak plasma concentration (Cmax) is reached 1-2 hours after oral administration (10 mg dose: Cmax = 15 ng/mL). Topical ocular administration results in very little systemic absorption (<1% of the dose) [2] - Distribution: The volume of distribution (Vd) in the human body is 1.2 L/kg; the brain/plasma concentration ratio is <0.05, indicating very low blood-brain barrier penetration [2] - Metabolism: It is mainly metabolized in the liver by cytochrome P450 (CYP) 3A4 into inactive metabolites [2] - Excretion: 70% of the dose is excreted in the urine (40% as the original drug and 30% as metabolites), and 25% is excreted in the feces. The elimination half-life (t1/2) in humans is 8-10 hours [2] - Plasma protein binding rate: Olopatadine hydrochloride (ALO4943A; KW4679) has a plasma protein binding rate of 55-60% in human plasma [2]
|
| Toxicity/Toxicokinetics |
Acute toxicity: LD50 in rats and mice >2000 mg/kg (oral); no deaths or serious clinical symptoms (convulsions, respiratory depression) have been reported [2] - Chronic toxicity: No significant hepatotoxicity or hematologic abnormalities were observed in rats after 6 months of oral administration of olopatadine hydrochloride (ALO4943A; KW4679) (100 mg/kg/day) [2] - Clinical side effects: Topical ophthalmic use may cause mild eye irritation (burning, stinging) in 3-5% of patients; oral administration may cause headache (2-3%) and dry mouth (1-2%). No sedative effect at therapeutic doses [2,3] - Drug interactions: No significant interactions with CYP3A4 inhibitors or other antihistamines; does not enhance central nervous system depression [2]
|
| References |
|
| Additional Infomation |
Olopatadine hydrochloride is a dibenzoxazole drug. Olopatadine hydrochloride is the hydrochloride salt form of olopatadine, a selective histamine H1 receptor antagonist and mast cell stabilizer with dual action and anti-allergic activity. Olopatadine stabilizes mast cells and prevents the release of histamine from them. Furthermore, it blocks histamine H1 receptors, thus preventing histamine from binding to these receptors. Both of these actions prevent the effects of histamine on capillary, bronchial, and gastrointestinal smooth muscle, including histamine-induced vasodilation, increased capillary permeability, bronchoconstriction, and spastic contraction of gastrointestinal smooth muscle. This drug also prevents histamine-induced mucosal pain and itching. It is an antihistamine with mast cell stabilizing action, used as eye drops to treat allergic conjunctivitis. See also: Olopatadine (containing active ingredient); Mometasone furoate; Olopatadine hydrochloride (ingredient)... See more...
Drug Indications Treatment of ocular signs and symptoms of seasonal allergic conjunctivitis. Olopatadine hydrochloride (ALO4943A; KW4679) is a second-generation non-sedating histamine H1 receptor antagonist with a dual anti-allergic mechanism: competitive H1 receptor blocking and inhibition of mast cell histamine release [2,3]. It exerts its anti-inflammatory effect by inhibiting pro-inflammatory cytokines (IL-6, IL-8) and the COX-2/PGE2 pathway, enhancing the efficacy against allergic inflammatory diseases [1,3]. Indications include allergic conjunctivitis (topical ophthalmic agent) and allergic dermatitis (topical cream), relieving symptoms such as itching, redness, edema, and tearing [1,2,3]. Its low blood-brain barrier penetration and high H1 receptor selectivity prevent it from having a sedative effect, distinguishing it from first-generation antihistamines [2]. The drug has a rapid onset of action (within 1 hour of administration) and a long duration of action (12-24 hours), supporting once- or twice-daily administration [2,3]. |
| Molecular Formula |
C21H24CLNO3
|
|
|---|---|---|
| Molecular Weight |
373.87
|
|
| Exact Mass |
373.144
|
|
| Elemental Analysis |
C, 67.46; H, 6.47; Cl, 9.48; N, 3.75; O, 12.84
|
|
| CAS # |
140462-76-6
|
|
| Related CAS # |
Olopatadine-d3 hydrochloride; 1331635-21-2; (Z)-Olopatadine-d3 hydrochloride; Olopatadine; 113806-05-6
|
|
| PubChem CID |
5282402
|
|
| Appearance |
White to off-white solid powder
|
|
| Density |
1.221 g/cm3
|
|
| Boiling Point |
523ºC at 760mmHg
|
|
| Melting Point |
242-245ºC
|
|
| Flash Point |
270.1ºC
|
|
| Vapour Pressure |
9.65E-13mmHg at 25°C
|
|
| LogP |
4.391
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
4
|
|
| Rotatable Bond Count |
5
|
|
| Heavy Atom Count |
26
|
|
| Complexity |
488
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
Cl[H].O1C([H])([H])C2=C([H])C([H])=C([H])C([H])=C2/C(=C(\[H])/C([H])([H])C([H])([H])N(C([H])([H])[H])C([H])([H])[H])/C2C([H])=C(C([H])([H])C(=O)O[H])C([H])=C([H])C1=2
|
|
| InChi Key |
HVRLZEKDTUEKQH-NOILCQHBSA-N
|
|
| InChi Code |
InChI=1S/C21H23NO3.ClH/c1-22(2)11-5-8-18-17-7-4-3-6-16(17)14-25-20-10-9-15(12-19(18)20)13-21(23)24;/h3-4,6-10,12H,5,11,13-14H2,1-2H3,(H,23,24);1H/b18-8-;
|
|
| Chemical Name |
2-[(11Z)-11-[3-(dimethylamino)propylidene]-6H-benzo[c][1]benzoxepin-2-yl]acetic acid;hydrochloride
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.69 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.69 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (6.69 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 17.14 mg/mL (45.84 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6747 mL | 13.3736 mL | 26.7473 mL | |
| 5 mM | 0.5349 mL | 2.6747 mL | 5.3495 mL | |
| 10 mM | 0.2675 mL | 1.3374 mL | 2.6747 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT03186755 | Recruiting | Drug: Hyaluronic acid 0.05% & Ectoine 2.0% Drug: Olopatadine hydrochloride ophthalmic solution 0.1% |
Conjunctivitis, Allergic Conjunctivitis, Vernal |
Michael Marchand, MD | June 11, 2017 | Phase 4 |
| NCT06126952 | Recruiting | Drug: Treatment A: Azelastine hydrochloride 0.15% nasal spray (Azelair) Drug: Treatment B: Placebo (Azelastine 0.15% vehicle) nasal spray |
Seasonal Allergic Rhinitis | MEDA Pharma GmbH & Co. KG | October 30, 2023 | Phase 2 |
| NCT00775658 | Completed | Drug: olopatadine Drug: placebo |
Allergic Rhinitis Allergic Conjunctivitis |
Vanderbilt University Medical Center |
January 2008 | Not Applicable |
| NCT01697969 | Completed | Drug: Olopatadine hydrochloride ophthalmic solution, 0.2% |
Allergic Conjunctivitis | Alcon Research | September 2012 | Phase 4 |
| NCT00609128 | Completed | Drug: olopatadine | Allergic Conjunctivitis | University of Wisconsin, Madison |
September 2000 | Not Applicable |
 |
|---|
 |
 |