
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
NMS-P937 (also know as NMS-P937; NMS1286937; NMS-P-937; NMS-P 937; Onvansertib) is an orally bioavailable, potent, selective, small-molecule Polo-like Kinase 1 (PLK1) inhibitor with potential antitumor activity. It exhibits 5000-fold selectivity over PLK2/PLK3 and an IC50 of 2 nM for inhibiting PLK1. NMS-1286937 selectively inhibits PLK1, causing reversible cell-cycle arrest at the G1 and G2 stages without apoptosis in normal cells, and selective G2/M cell-cycle arrest followed by apoptosis in a variety of tumor cells. Following oral administration, NMS-P937 demonstrated activity in vivo in the HCT116 xenograft model. The drug is currently being evaluated in Phase I clinical trials.
| Targets |
PLK1 (IC50 = 2 nM); FLT3 (IC50 = 510 nM); MELK (IC50 = 744 nM); CK2 (IC50 = 826 nM)
NMS-P937 (NMS1286937; Onvansertib) targets Polo-like kinase 1 (Plk1) with a Ki value of 0.8 nM and an IC50 value of 1.9 nM in recombinant kinase assays [1] NMS-P937 exhibits high selectivity for Plk1, with IC50 values > 10 μM for Plk2 and Plk3, and no significant inhibition of 45 other tested kinases (IC50 > 1 μM) [2] NMS-P937 specifically inhibits Plk1 kinase activity in primary CD56+ acute monoblastic leukemia cells, with no cross-reactivity to other mitotic kinases at therapeutic concentrations [3] |
|---|---|
| ln Vitro |
NMS-P937 demonstrates broad-spectrum antiproliferative activity against cell lines from lymphomas, leukemias, and solid tumors. In A2780 cells, NMS-P937 potently induces a mitotic cell-cycle arrest that is followed by apoptosis.[2]
Against a panel of human solid tumor cell lines (A549, HCT116, MCF-7, PC3, MiaPaCa-2), NMS-P937 showed potent antiproliferative activity with IC50 values ranging from 2.3 nM to 15.7 nM [1] - NMS-P937 induced G2/M cell cycle arrest in HCT116 cells, characterized by increased cyclin B1/Cdk1 complex levels and reduced phosphorylation of Plk1 substrates (Cdc25C, BubR1) [1] - Treatment with NMS-P937 (10 nM) for 48 hours triggered apoptosis in A549 cells, as evidenced by caspase-3/7 activation (3.2-fold increase vs vehicle) and PARP cleavage [1] - In hematologic malignancy cell lines (K562, MV4-11, Jurkat, Raji), NMS-P937 inhibited proliferation with IC50 values between 1.8 nM and 9.6 nM [2] - NMS-P937 synergized with paclitaxel, doxorubicin, and gemcitabine in solid tumor cell lines, with combination indices (CI) ranging from 0.3 to 0.7 [2] - In primary CD56+ acute monoblastic leukemia cells from patients (n=8), NMS-P937 suppressed cell viability with IC50 values of 3.1 nM to 8.4 nM, and induced apoptosis in 45-62% of cells at 10 nM [3] - NMS-P937 inhibited clonogenic growth of primary CD56+ acute monoblastic leukemia cells, reducing colony formation by 70-85% at 5 nM [3] |
| ln Vivo |
NMS-P937 (90 mg/kg/d i.v. or p.o.) significantly inhibits the growth of tumors in mice xenografted with human HCT116 colon adenocarcinoma cells.[1]
NMS-P937 inhibits xenograft tumor growth in mice with HT29, Colo205 colorectal, or A2780 ovarian xenograft tumors. Furthermore, when used with authorized cytotoxic medications, NMS-P937 promotes improved tumor regression and increases animal survival.[2] In HCT116 human colon cancer xenograft models (nu/nu mice), oral administration of NMS-P937 (25 mg/kg, q.d. for 14 days) resulted in 78% tumor growth inhibition (TGI), with no significant body weight loss [2] - In MV4-11 human acute myeloid leukemia (AML) xenograft models (SCID mice), intravenous NMS-P937 (10 mg/kg, q.o.d. for 21 days) induced 82% TGI and prolonged median survival by 56% vs vehicle [2] - In a disseminated CD56+ acute monoblastic leukemia mouse model (NOD/SCID mice), NMS-P937 (15 mg/kg, oral, q.d. for 28 days) reduced bone marrow and spleen leukemic cell infiltration by 68% and 73%, respectively, and extended median survival from 32 days to 57 days [3] - Tumor tissues from NMS-P937-treated mice showed reduced Ki-67 proliferation index (40% vs 75% in vehicle) and increased TUNEL-positive apoptotic cells (35% vs 8% in vehicle) [2][3] |
| Enzyme Assay |
Using a trans-phosphorylation assay, the potency of particular compounds and the inhibitory activity of putative kinase inhibitors are assessed. Under optimal buffer and cofactor conditions, a particular tyrosine kinase or serine-threonine kinase will trans-phosphorylate a specific peptide or protein substrate in the presence of ATP traced with 33P-γ-ATP. An excess of the ion exchange Dowex resin is added at the end of the phosphorylation reaction to capture over 98% of the unlabeled ATP and radioactive ATP; the resin then falls to the bottom of the reaction plate due to gravity. The phosphorylated substrate-containing supernatant is then extracted and put into a counting plate for analysis using b-counting. At 25 °C, an end-point assay lasting 60 minutes was used to assess the inhibitory potency of all the kinases that were tested. The substrate and ATP concentrations were maintained at 2 x αKm and >5 x αKm, respectively.
Recombinant Plk1 catalytic domain kinase assay: The assay was performed in reaction buffer containing ATP (10 μM) and a fluorescently labeled peptide substrate. Serial concentrations of NMS-P937 (0.1 nM to 1 μM) were added, and the mixture was incubated at 30°C for 60 minutes. Phosphorylated substrate was detected by fluorescence polarization, and Ki/IC50 values were calculated using nonlinear regression [1] - Kinase selectivity panel assay: NMS-P937 (1 μM and 10 μM) was tested against a panel of 48 human kinases (including Plk2, Plk3, Aurora A/B, CDK1) using the same fluorescence polarization method. Inhibition rates were determined relative to vehicle controls, and IC50 values were calculated for kinases showing > 50% inhibition at 10 μM [2] - Primary cell Plk1 activity assay: Lysates from primary CD56+ acute monoblastic leukemia cells were incubated with NMS-P937 (0.5 nM to 20 nM) and Plk1-specific substrate. Kinase activity was measured by radiolabeled ATP incorporation, and IC50 values were derived from dose-response curves [3] |
| Cell Assay |
In suitable medium supplemented with 10% fetal calf serum, cells are seeded into 96- or 384-well plates at densities ranging from 10,000 to 30,000/cm2 for adherent cells and 100,000/mL for nonadherent cells. Cells were treated in duplicate with NMS-P937 serial dilutions after a 24-hour period, and the CellTiter-Glo Assay (Promega) was used to determine the viable cell count 72 hours later. Using the Assay Explorer MDL sigmoidal fitting algorithm, IC50 values were determined. At least two independent experiments were conducted.
Antiproliferative assay: Solid tumor or hematologic malignancy cells were seeded in 96-well plates (3×103 to 5×103 cells/well) and treated with serial concentrations of NMS-P937 (0.1 nM to 100 nM) for 72 hours. Cell viability was assessed by a colorimetric assay based on tetrazolium salt reduction, and IC50 values were calculated from sigmoidal dose-response curves [1][2] - Cell cycle analysis: Cells were treated with NMS-P937 (5 nM to 20 nM) for 24-48 hours, harvested, fixed with 70% ethanol, stained with propidium iodide, and analyzed by flow cytometry to determine the percentage of cells in G1, S, and G2/M phases [1][2] - Apoptosis assay: Cells were exposed to NMS-P937 (10 nM to 30 nM) for 48 hours, stained with annexin V-FITC and propidium iodide, and analyzed by flow cytometry. Caspase activation was detected by Western blot using antibodies against cleaved caspase-3, -7, and PARP [1][3] - Clonogenic assay: Primary CD56+ acute monoblastic leukemia cells were plated in methylcellulose-based medium with NMS-P937 (1 nM to 10 nM). Colonies (> 50 cells) were counted after 14 days of incubation, and colony formation efficiency was calculated relative to vehicle controls [3] - Combination assay: Solid tumor cells were treated with NMS-P937 (0.5 nM to 5 nM) in combination with paclitaxel (0.1 nM to 1 nM), doxorubicin (0.5 nM to 5 nM), or gemcitabine (10 nM to 100 nM) for 72 hours. Cell viability was measured, and combination indices were determined using the Chou-Talalay method [2] |
| Animal Protocol |
Female Hsd athymic nu/nu mice, aged 5 to 6 weeks (average weight: 20–22 g), are used for carcinoma xenograft studies. Subcutaneous inoculation is used to inoculate the colorectal HCT116, HT29, ovarian human carcinoma A2780, and colono205 cell lines. Treatment begins the day after randomization and involves giving vehicle or NMS-P937 to mice with a palpable tumor (100-200 mm 3 ) according to prescribed doses and schedules. Tumor growth inhibition (TGI) is calculated and tumor dimensions are routinely measured using Vernier callipers. Reduction of body weight is used to assess toxicity. Severe combined immunodeficient mice (SCID; average weight: 20–22 g) that are 5–6 weeks old are used for leukemia research. Treatments begin with subcutaneous injections of the AmL cell line HL-60 (5×10 6 cells) and end when the tumor size reaches 200 to 250 mm 3 . TGI and tumor dimensions are evaluated. Treatments for disseminated models begin two days after intravenous injection of 5x10 6 AmL primary cells (AmL-PS). Every day, mice are checked for disease-related symptoms, and the median survival period for each group is calculated.
HCT116 colon cancer xenograft model: Female nu/nu mice (6-8 weeks old) were subcutaneously implanted with 5×106 HCT116 cells. When tumors reached 100-150 mm3, mice were randomized into groups (n=6/group) and administered NMS-P937 orally (12.5, 25, 50 mg/kg) or vehicle (0.5% carboxymethylcellulose + 0.1% Tween 80) once daily for 14 days. Tumor volume and body weight were measured every 2 days [2] - MV4-11 AML xenograft model: Female SCID mice (6-8 weeks old) were intravenously injected with 1×107 MV4-11 cells. Seven days post-inoculation, mice were randomized (n=8/group) and given NMS-P937 intravenously (5, 10, 20 mg/kg) or vehicle once every other day for 21 days. Tumor burden was monitored by bioluminescence imaging, and survival was recorded [2] - Disseminated CD56+ acute monoblastic leukemia model: Female NOD/SCID mice (6-8 weeks old) were intravenously injected with 2×106 primary human CD56+ acute monoblastic leukemia cells. Ten days post-inoculation, mice were randomized (n=10/group) and administered NMS-P937 orally (15 mg/kg) or vehicle once daily for 28 days. Mice were euthanized at endpoint, and bone marrow/spleen leukemic infiltration was quantified by flow cytometry [3] |
| ADME/Pharmacokinetics |
In mice, after oral administration of NMS-P937 (25 mg/kg), the Cmax was 3.2 μM, the AUC0-24h was 18.7 μM·h, and the oral bioavailability was 58% [2]. In mice, after intravenous administration of NMS-P937 (10 mg/kg), the clearance was 9.4 mL/min/kg, the volume of distribution (Vss) was 1.3 L/kg, and the terminal half-life (t1/2) was 8.2 h [2]. NMS-P937 has good water solubility (≥150 μM) and high human plasma protein binding (92%) [2]. In rats, after oral administration of NMS-P937 (20 mg/kg), the Cmax was 2.8 μM, and the AUC0-24h was 15.3 μM. μM·h, oral bioavailability was 52% [2]
|
| Toxicity/Toxicokinetics |
In repeated oral toxicity studies in mice (28 days, 12.5–50 mg/kg/day), NMS-P937 caused mild, reversible myelosuppression (25–30% decrease in white blood cell count at 50 mg/kg), but did not cause significant changes in body weight, organ weight, or histopathological changes in liver, kidney, heart, or spleen tissues [2]. - NMS-P937 did not inhibit human cytochrome P450 enzymes (CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP3A4) at concentrations up to 20 μM, suggesting a low likelihood of drug interactions [2]. - In dogs, oral administration of NMS-P937 (10 mg/kg/day for 14 days) showed no significant toxicity, and no significant changes were observed in plasma. The drug concentration remained above the in vitro IC50 for 12 hours after administration [2].
|
| References |
|
| Additional Infomation |
Onvansertib is being investigated in the clinical trial NCT03303339 (Onvansertib in combination with low-dose cytarabine or decitabine for the treatment of adult patients with acute myeloid leukemia (AML). Onvansertib is an orally bioavailable ATP-competitive inhibitor that inhibits polo-like kinase 1 (PLK1; PLK-1; STPK13) and possesses potential antitumor activity. After administration, onvansertib selectively binds to and inhibits PLK1, thereby interfering with mitosis and inducing selective G2/M phase cell cycle arrest in PLK1-overexpressing tumor cells, ultimately leading to apoptosis. PLK1, named after the polo gene in Drosophila, is a serine/threonine kinase crucial for the regulation of mitosis and plays a key role in tumor cell proliferation. PLK1 expression is upregulated in various tumor cell types, and high expression is associated with increased tumor invasiveness and poor prognosis.
NMS-P937 is a synthetic 4,5-dihydro-1H-pyrazolo[4,3-h]quinazoline derivative designed as a potent and selective PLK1 inhibitor[1]. - The antitumor activity of NMS-P937 is achieved by blocking the PLK1-dependent mitotic process, resulting in G2/M phase cell cycle arrest, mitotic catastrophe, and ultimately inducing apoptosis in cancer cells[1][2][3]. - NMS-P937 has shown efficacy in both solid tumors and hematologic malignancies, including acute myeloid leukemia and CD56+ acute monocytic leukemia, indicating its broad antitumor potential[2][3]. - Good oral bioavailability and pharmacokinetic properties also support its antitumor activity. NMS-P937 has good properties and low toxicity, making it a promising candidate drug for clinical development in cancer treatment[2]. |
| Molecular Formula |
C24H27F3N8O3
|
|
|---|---|---|
| Molecular Weight |
532.52
|
|
| Exact Mass |
532.215
|
|
| Elemental Analysis |
C, 54.13; H, 5.11; F, 10.70; N, 21.04; O, 9.01
|
|
| CAS # |
1034616-18-6
|
|
| Related CAS # |
1263293-37-3 (fumarate); 1034616-18-6;
|
|
| PubChem CID |
49792852
|
|
| Appearance |
Off-white to light yellow solid powder
|
|
| Density |
1.6±0.1 g/cm3
|
|
| Boiling Point |
757.8±70.0 °C at 760 mmHg
|
|
| Flash Point |
412.1±35.7 °C
|
|
| Vapour Pressure |
0.0±2.7 mmHg at 25°C
|
|
| Index of Refraction |
1.692
|
|
| LogP |
0.79
|
|
| Hydrogen Bond Donor Count |
3
|
|
| Hydrogen Bond Acceptor Count |
12
|
|
| Rotatable Bond Count |
7
|
|
| Heavy Atom Count |
38
|
|
| Complexity |
817
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
FC(OC1C([H])=C([H])C(=C([H])C=1N([H])C1=NC([H])=C2C(C3=C(C(C(N([H])[H])=O)=NN3C([H])([H])C([H])([H])O[H])C([H])([H])C2([H])[H])=N1)N1C([H])([H])C([H])([H])N(C([H])([H])[H])C([H])([H])C1([H])[H])(F)F
|
|
| InChi Key |
QHLVBNKYJGBCQJ-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C24H27F3N8O3/c1-33-6-8-34(9-7-33)15-3-5-18(38-24(25,26)27)17(12-15)30-23-29-13-14-2-4-16-20(22(28)37)32-35(10-11-36)21(16)19(14)31-23/h3,5,12-13,36H,2,4,6-11H2,1H3,(H2,28,37)(H,29,30,31)
|
|
| Chemical Name |
1-(2-hydroxyethyl)-8-[5-(4-methylpiperazin-1-yl)-2-(trifluoromethoxy)anilino]-4,5-dihydropyrazolo[4,3-h]quinazoline-3-carboxamide
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2 mg/mL (3.76 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2 mg/mL (3.76 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2 mg/mL (3.76 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8779 mL | 9.3893 mL | 18.7786 mL | |
| 5 mM | 0.3756 mL | 1.8779 mL | 3.7557 mL | |
| 10 mM | 0.1878 mL | 0.9389 mL | 1.8779 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
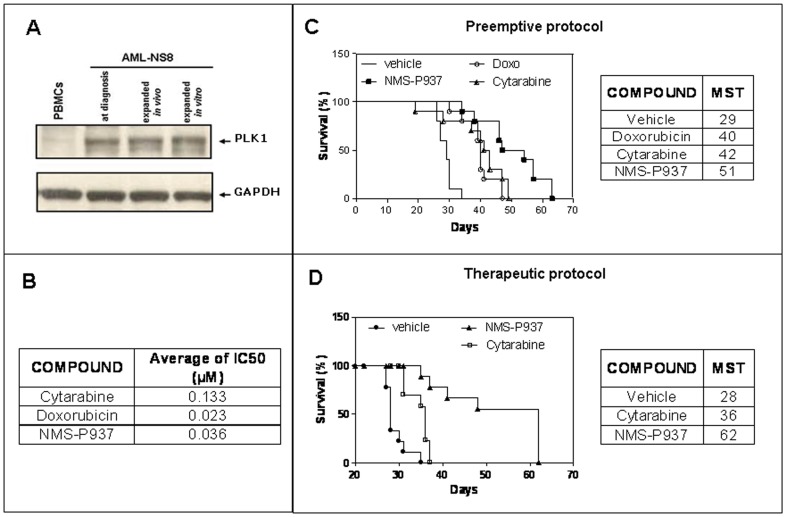 PLK1 expression in AML-NS8 cells and activity of PLK1 inhibitor NMS-937in vitroandin vivo.PLoS One.2013;8(3):e58424. |
|---|
Leukaemic infiltration of meninges and soft tissues from mice treated with NMS-P937 and cytarabine following a therapeutic schedule.PLoS One.2013;8(3):e58424. |
Mechanism of action of PLK1 inhibitorin vitroandin vivo.PLoS One.2013;8(3):e58424. |