
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
Purity: ≥98%
MPTP hydrochloride is a dopaminergic neurotoxin and cause selective destruction of dopaminergic neurons in animal models of parkinsonism. MPTP prevented ICR mice's (20–30 g) phrenic nerve–hemidiaphragm preparations from exhibiting nerve-evoked twitches. MPTP, TC, and PP all had twitch inhibition IC50 values of 53, 0.7, and 123 uM, respectively.
| Targets |
Dopamine neurotoxin
Mitochondrial complex I (NADH-ubiquinone oxidoreductase) (IC50 for MPP+ (metabolite of MPTP hydrochloride) = 0.7 μM in isolated mouse brain mitochondria) [3] - Dopamine transporter (DAT), facilitating entry of MPP+ (metabolite of MPTP hydrochloride) into dopaminergic neurons [4] |
|---|---|
| ln Vitro |
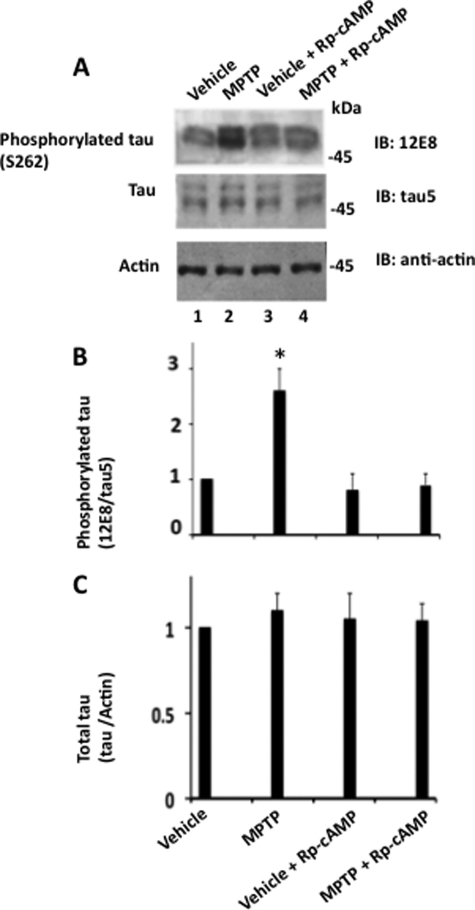
In vitro activity: Upon exposure to all MPTP doses, the morphology of glioma and N2AB-1 cells remained unchanged. Furthermore, MPTP therapy had no effect on the proliferation of C6 glioma cells[3]. In human neuroblastoma M17 cells, MPTP stimulates both tau phosphorylation and apoptosis. For human neuroblastoma M17 cells, MPTP strongly stimulates Tau phosphorylation at Ser262. Within our M17 human neuroblastoma cells, MPTP led to a dose-dependent rise in intracellular α-synuclein levels. MPTP appears to promote Tau phosphorylation in the brain by activating both PKA and GSK3β[4]. MPTP hydrochloride (100 μM) was metabolized to MPP+ by astrocytes, and the conditioned medium containing MPP+ induced death of dopaminergic neurons (N27 cells) with 40% viability reduction after 24 hours; this toxicity was blocked by DAT inhibitors [4] - MPP+ (metabolite of MPTP hydrochloride) inhibited mitochondrial complex I activity in isolated rat brain mitochondria, reducing NADH oxidation rate by 58% at 1 μM [3] - MPTP hydrochloride (50-200 μM) treatment of primary rat cortical neurons for 48 hours increased reactive oxygen species (ROS) production, decreased mitochondrial membrane potential, and induced apoptotic cell death (35% apoptotic rate at 150 μM) [1] |
| ln Vivo |
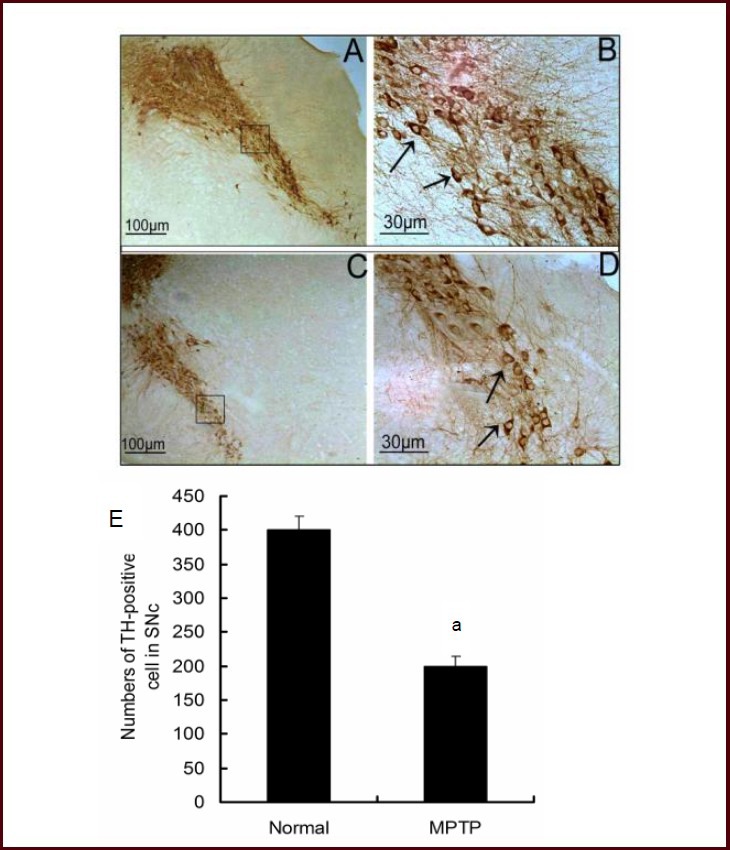
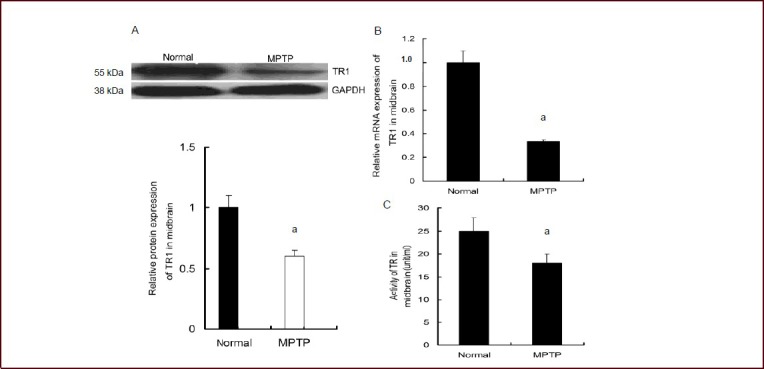
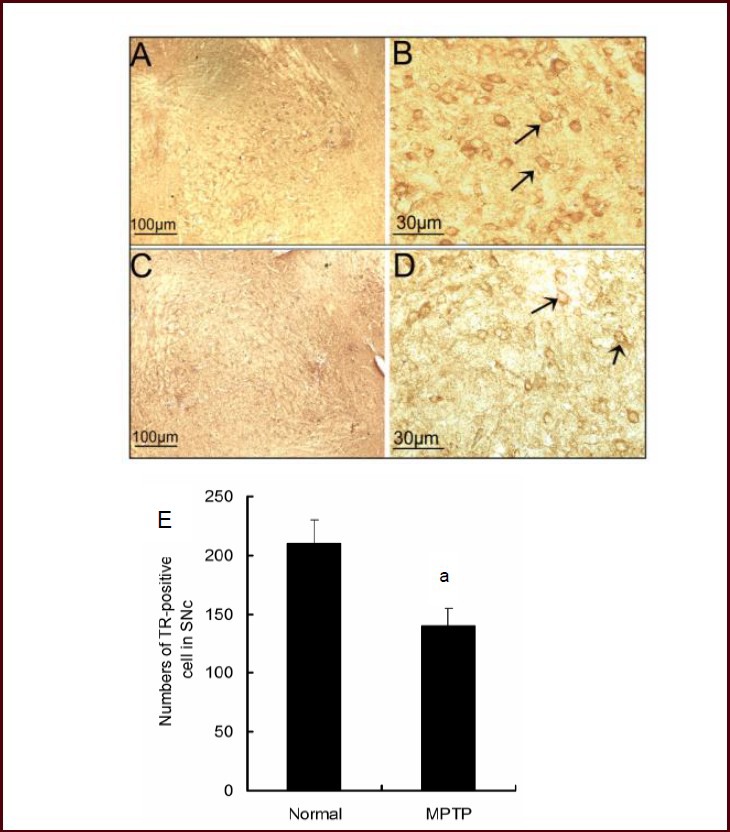
The number of tyrosine hydroxylase-positive neurons was decreased in the substantia nigra pars compacta of MPTP-treated mice. In the mouse midbrain, MPTP reduced the expression and activity of thioredoxin reductase 1, as well as the quantity of thioredoxin reductase 1-positive cells in the substantia nigra pars compacta. In human and nonhuman primates, the toxin MPTP administration can result in neurochemical, behavioral, and histopathological changes akin to those seen in Parkinsonian patients. Mice are less susceptible to MPTP than primates are. The most popular and reliable method of administering MPTP is systemic administration, which includes subcutaneous, intravenous, intraperitoneal, and intramuscular injection.Other routes of administration include gavage and stereotactic injection. Lipophilic protoxin MPTP has a quick blood-brain barrier crossing time after systemic injection. Once inside the brain, monoamine oxidase B transforms MPTP into 1-methyl-4-phenylpyridine[1]. Studies on humans, monkeys, and mice have demonstrated that MPTP is toxic to dopaminergic neurons in the nigrostriatal system and causes a persistent depletion of dopamine and its metabolites in the striatum[2].
C57BL/6 mice received MPTP hydrochloride (30 mg/kg, intraperitoneal injection, once daily for 5 days) induced Parkinson's disease (PD)-like symptoms: reduced locomotor activity (45% decrease in total distance traveled), loss of dopaminergic neurons in the substantia nigra pars compacta (SNpc) (60% reduction in tyrosine hydroxylase (TH)-positive neurons), and decreased dopamine content in the striatum (75% reduction) [1] - Pregnant CD-1 mice were administered MPTP hydrochloride (10 or 20 mg/kg, subcutaneous injection) on gestational day 12.5; fetal brains showed reduced TH-positive neurons in the midbrain (28% reduction at 20 mg/kg) and increased oxidative stress markers (4-hydroxynonenal levels elevated by 32%) [2] - Male C57BL/6 mice injected with MPTP hydrochloride (20 mg/kg, intravenous injection, single dose) showed peak MPP+ concentration in the striatum at 1 hour post-injection (12 ng/mg tissue), with selective damage to SNpc dopaminergic neurons (40% loss of TH-positive cells after 7 days) [3] |
| Enzyme Assay |
MPTP prevented phrenic nerve-hemidiaphragm preparations from ICR mice (20–30 g) from experiencing nerve-evoked twitches. The MPTP's inhibitory effect was amplified by PP 50 uM but not by PA 50 uM or TP 50 uM. The amplitude of twitch was inhibited by PP 100 uM alone. The MPTP, TC, and PP twitch inhibition IC50 values were 53, 0.7, and 123 uM, in that order. MPTP and TC had IC50 values of 18 and 0.3 uM, respectively, after pretreatment with PP 50 uM.
Isolated mouse brain mitochondria were suspended in reaction buffer and incubated with various concentrations of MPP+ (metabolite of MPTP hydrochloride) for 15 minutes at 37°C. NADH-ubiquinone oxidoreductase activity was measured by monitoring the decrease in absorbance at 340 nm (NADH oxidation) to calculate IC50 values [3] - Mitochondrial complex I activity assay was performed in rat brain homogenates treated with MPTP hydrochloride (in vitro conversion to MPP+ via added monoamine oxidase B). The reaction mixture contained NADH as substrate, and enzyme activity was quantified by spectrophotometric detection of ubiquinone reduction [4] |
| Cell Assay |
N2AB-1 and C6 glioma cells were plated using the culture medium previously mentioned in 24-well costar dishes (16 mm diameter), with 50,000 cells per well. Following a 24-hour period, duplicate medium containing different MPTP or MPP+ concentrations was added. After three days of treatment, both control and treated cells were trypsinized and their counts were measured using a hemocytometer each day.
Primary rat cortical neurons were cultured in neurobasal medium for 7 days, then treated with MPTP hydrochloride (50-200 μM) for 48 hours. ROS production was detected by DCFH-DA staining and flow cytometry; mitochondrial membrane potential was measured using JC-1 dye; apoptotic cells were identified by Annexin V-FITC/PI double staining [1] - N27 dopaminergic cells were cultured in RPMI 1640 medium and exposed to astrocyte-conditioned medium (pretreated with MPTP hydrochloride (100 μM) for 24 hours). Cell viability was assessed by MTT assay; Western blot was used to detect cleaved caspase-3 and Bcl-2/Bax ratio [4] - Primary mouse midbrain dopaminergic neurons were isolated and cultured, then treated with MPP+ (10 μM, metabolite of MPTP hydrochloride) for 24 hours. TH expression was detected by immunocytochemistry; intracellular ATP levels were measured by luciferase-based assay [2] |
| Animal Protocol |
The animal treatments are carried out in order to prepare the MPTP mouse model and the LPS rat model. In short, adult rats are given unilateral injections of LPS (0.5 μL of 10 μg/μL diluted in 0.9% saline) into the contralateral side of the same volume of 0.9% saline and into the medial forebrain bundle (MFB) at the following coordinates: AP-4.2 mm, L 1.5 mm, and V 7.8 mm. For five consecutive days, adult mice receive intraperitoneal injections of MPTP at a dose of 25 mg/kg. As a control, the same volume of saline is injected. Once the animals receive injections of MPTP or LPS, they are all sacrificed at weeks 1, 2, 3, or 4. The brain samples are gathered in preparation for the ensuing western blot and immunohistochemistry tests.
C57BL/6 mice (8-10 weeks old) were randomly divided into control and MPTP hydrochloride groups. The treatment group received intraperitoneal injections of 30 mg/kg MPTP hydrochloride (dissolved in normal saline) once daily for 5 consecutive days. On day 7 post-first injection, locomotor activity was tested using an open-field apparatus, then mice were euthanized to collect brain tissues (SNpc and striatum) for histopathological and biochemical analysis [1] - Pregnant CD-1 mice (gestational day 12) were assigned to control, 10 mg/kg, and 20 mg/kg MPTP hydrochloride groups. MPTP hydrochloride was dissolved in normal saline and administered via subcutaneous injection. On gestational day 18, mice were euthanized, fetal brains were dissected, and midbrain regions were processed for TH immunostaining and oxidative stress marker detection [2] - Male C57BL/6 mice (12 weeks old) were injected intravenously with a single dose of 20 mg/kg MPTP hydrochloride (dissolved in phosphate-buffered saline). At 1, 3, 6, and 24 hours post-injection, mice were euthanized to collect striatum, SNpc, liver, and kidney tissues for MPP+ concentration measurement by high-performance liquid chromatography (HPLC) [3] |
| ADME/Pharmacokinetics |
MPTP Metabolism [4]
MPTP metabolism is a complex multi-step process2. After systemic administration, the highly lipophilic MPTP can rapidly cross the blood-brain barrier. After entering the brain tissue, the protoxin MPTP is metabolized in non-dopaminergic cells by monoamine oxidase B (MAO-B) to 1-methyl-4-phenyl-2,3-dihydropyridinium, and then (possibly through spontaneous oxidation) further metabolized to 1-methyl-4-phenylpyridinium (MPP+), i.e., the active toxic compound. Subsequently, MPP+ is released into the extracellular space. Since MPP+ is a polar molecule, unlike the proto-MPTP, it cannot freely enter the cell, but depends on plasma membrane transport proteins to enter neurons such as dopaminergic neurons. MPP+ exhibits high affinity for plasma membrane dopamine transporters18, as well as norepinephrine and serotonin transporters. Once MPP+ enters dopaminergic neurons, it can be isolated into synaptosomal vesicles19 or accumulate in mitochondria20. This complex toxicokinetics means that to properly interpret the results of MPTP studies, it is necessary to assess key aspects of MPTP metabolism to ensure that observed effects are related to real and meaningful molecular events, rather than to interference from toxicological pharmacology. Assessments may include, but are not limited to, striatal MPP+ levels (described below), MAO-B activity, lactate levels, and the uptake of [3H]-dopamine and [3H]-MPP+. Protocols for these analyses can be found in the following references. MPTP hydrochloride is rapidly absorbed after administration via the parenteral route (intraperitoneal/intravenous) and distributed to the brain within 15 minutes [3] - In the brain, MPTP hydrochloride is metabolized to MPP+ by astrocyte-specific monoamine oxidase B (MAO-B); MPP+ is taken up by dopaminergic neurons via dopamine transporter (DAT) [4] - The elimination half-life of MPP+ in the striatum is 3.2 hours; MPP+ is mainly excreted in the urine as unchanged metabolites (65% of the administered dose is excreted within 24 hours) [3] - MPTP hydrochloride does not cross the placenta effectively, but after maternal administration, MPP+ (metabolites) can accumulate in fetal brain tissue [2] |
| Toxicity/Toxicokinetics |
Acute toxicity of MPTP hydrochloride in mice: LD50 was 38 mg/kg (intraperitoneal injection); mice died of respiratory failure within 24 hours [3]
- Selective neurotoxicity: MPTP hydrochloride (metabolized to MPP+) caused irreversible loss of dopaminergic neurons in the substantia nigra pars compacta (SNpc), leading to Parkinson's-like motor dysfunction [1] - Maternal administration of MPTP hydrochloride (≥10 mg/kg) during pregnancy can induce fetal neurotoxicity, including a decrease in the number of dopaminergic neurons in the fetal midbrain and an increase in oxidative stress [2] - No significant hepatotoxicity or nephrotoxicity was observed in mice after acute administration of MPTP hydrochloride (30 mg/kg, 5 days); plasma alanine aminotransferase (ALT) and creatinine levels remained within the normal range [1] |
| References | |
| Additional Infomation |
This study reports two animal models established in amphibians (frogs and salamanders). In these models, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) produced neurobehavioral and biochemical manifestations similar to those in humans, and also caused measurable alterations in at least one neural crest pigment cell—skin melanocytes. We believe this novel approach holds promise as an inexpensive and easily quantifiable model for studying the effects of MPTP on the central and peripheral nervous systems. We also demonstrate that monoamine oxidase inhibitors completely eliminate the toxic effects of MPTP in vivo, while catechol-O-methyltransferase inhibitors enhance these toxic effects. MPTP is oxidatively metabolized to toxic metabolites, but its presumed final metabolite, the 1-methyl-4-phenylpyridine ion (MPP+), is even more toxic than MPTP in this model, possibly through a different mechanism. [1]
This study investigated the enhancing effect of 4-phenylpyridine (a MAO-B inhibitor) on the neuromuscular blocking effect of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) in a mouse phrenic nerve-diaphragm model. MPTP blocked nerve-induced muscle twitching in a concentration-dependent manner (1-200 μM). 4-phenylpyridine (rather than pargyline or transphenylcyclopropane) enhanced this inhibitory effect of MPTP. Pretreatment with 50 μM 4-phenylpyridine reduced the IC50 values of MPTP and d-tubocurarine from 53 μM and 0.7 μM to 0.3 μM (the concentration required to inhibit 50% muscle twitching amplitude, respectively). 4-phenylpyridine also enhanced the inhibitory effect of MPTP and d-tubocurarine on acetylcholine (0.1 mM)-induced denervated mouse diaphragmatic contractions. 4-Phenylepyridine enhanced the inhibitory effect of α-cladosporin-induced muscle twitching and the specific binding of [125I]α-cladosporin to the mouse diaphragm, but 4-phenylpyridine pretreatment had no significant effect on the inhibition of [125I]α-cladosporin binding by MPTP and d-tubocurarine. Electrophysiological studies showed that 4-phenylpyridine enhanced the inhibitory effect of MPTP and d-tubocurarine on the amplitude of MEPP and EPPP. This indicates that 4-phenylpyridine enhances the neuromuscular blocking effect of MPTP and d-tubocurarine on postsynaptic nicotinic acetylcholine receptors. This finding suggests that the application of 4-phenylpyridine in MPTP-induced Parkinson's disease may be limited by its enhancement of MPTP neuromuscular blocking effect. [2] Background: Reactive astrogenesis is a prominent pathological feature of the brain in patients with Parkinson's disease (PD), but its progression and regulatory mechanisms are still unclear. This study discovered that growth arrest-specific 1 (Gas1) is a tumor growth inhibitory oncogene that can serve as a novel regulator of apoptosis in primary cultured reactive astrocytes and damaged substantia nigra. Methods: Animal models and cell culture methods were used in this study. Animal models treated with lipopolysaccharide (LPS) and 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) were employed. Gas1 expression in brain tissue was detected by immunohistochemistry and Western blotting. The role of Gas1 in the viability and apoptosis of reactive astrocytes and SH-SY5Y cells was analyzed using double labeling, CCK-8 assay, LDH, TUNEL assay, flow cytometry, and siRNA knockdown. Results: In LPS- or MPTP-damaged brain tissue, Gas1 expression was significantly increased in most reactive astrocytes. In the damaged substantia nigra, GFAP-positive astrocytes exhibited higher levels of cleaved caspase-3. In cell culture, upregulated Gas1 expression induced apoptosis in reactive astrocytes stimulated by LPS in combination with interferon-γ and tumor necrosis factor-α. This effect was confirmed by siRNA knockdown of Gas1 gene expression. Finally, it is noteworthy that the underlying signaling pathway was significantly associated with an increased Bax/Bcl-2 ratio, excessive production of reactive oxygen species, and activation of cleaved caspase-3. Conclusion: This study demonstrates that upregulation of inducible Gas1 promotes apoptosis in reactive astrocytes in damaged substantia nigra. The Gas1 signaling pathway may serve as a novel regulator of astrocyte proliferation and therefore a potential intervention target for inflammatory events in Parkinson's disease (PD). [3] View MoreThis protocol describes the method used to construct a reliable mouse model of Parkinson's disease (PD) using the neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). We discuss the specific details of the model, provide key references, and outline what researchers need to know to safely and successfully construct an MPTP-based mouse model of Parkinson's disease. The completion of this protocol depends on the MPTP dosing regimen used and the actual planned study, which typically lasts from 7 to 30 days. This protocol requires the implementation of safety measures and the purchase of several devices, which is a one-time investment worthwhile if the model is chosen to be used regularly. [4] Background: Studies have shown that neuroinflammation, in which activated microglia play a crucial role, is closely associated with the development of Parkinson's disease (PD). Therefore, modulating microglia activation is a potential therapeutic target for combating dopaminergic neurodegeneration in this neurological disease. Under physiological conditions, various soluble and membrane-associated inhibitory mechanisms help maintain the resting/surveillance phenotype of microglia. However, the presence of activated microglia in the brains of PD patients suggests that these mechanisms may be overloaded. We focused on a membrane-associated mechanism, namely the CD200-CD200R1 ligand-receptor pair. Methods: Using an acute MPTP-induced PD mouse model, we investigated the temporal patterns of CD200 and CD200R1 mRNA expression against the backdrop of MPTP-induced dopaminergic neurodegeneration and neuroinflammation. Dopaminergic damage was assessed by tyrosine hydroxylase (TH) immunoreactivity, and neuroinflammation was assessed by mRNA expression of inflammatory markers and immunohistochemistry of IBA1 and GFAP. This study investigated the effects of CD200-CD200R1 system regulation on MPTP-induced damage using CD200R1 agonist or CD200 knockout mice. Results showed that MPTP administration led to a gradual decrease in tyrosine hydroxylase (TH)-positive fibers in the striatum and TH-positive neurons in the substantia nigra pars compacta, accompanied by transient astrogenesis, microgliosis, and the expression of pro-inflammatory and anti-inflammatory markers. Following MPTP treatment, CD200 mRNA levels in the ventral midbrain rapidly decreased, while CD200R1 mRNA expression showed transient decreases in both early and late stages in this brain region. Conversely, CD200R1 expression in the striatum showed a transient increase. CD200R1 agonist administration inhibited MPTP-induced degeneration of dopaminergic neurons, while microglia in CD200-deficient mice showed earlier signs of activation. Conclusion: In summary, these findings provide evidence for the correlation between CD200-CD200R1 alterations, glial cell activation, and neuronal loss. CD200R1 stimulation reduced MPTP-induced loss of dopaminergic neurons, while CD200 deficiency led to earlier microglial activation, suggesting that enhancing CD200R1 signaling may be a possible approach to control neuroinflammation and neuronal death in Parkinson's disease (PD). [5] Parkinson's disease (PD) is a progressive neurodegenerative disease characterized by the loss of dopaminergic neurons in the substantia nigra (SN) and a decrease in dopamine levels in the striatum. Although the molecular mechanisms of dopaminergic neuronal death in PD are not well understood, neuroinflammation is also considered an important mediator in the pathogenesis and progression of PD. This study showed that in a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced PD mouse model, activation of the NLRP3 inflammasome in microglia was crucial for the loss of dopaminergic neurons and subsequent motor dysfunction. Specifically, NLRP3 deficiency significantly alleviated motor dysfunction and dopaminergic neuronal degeneration in MPTP-treated mice. Furthermore, NLRP3 deficiency also eliminated MPTP-induced microglia recruitment, interleukin-1β (IL-1β) production, and caspase-1 activation in the mouse substantia nigra (SN). In primary microglia and mixed glial cell cultures, MPTP/ATP treatment promoted the assembly and activation of the NLRP3 inflammasome by generating mitochondrial reactive oxygen species (ROS). In mouse bone marrow-derived macrophages, 1-methyl-4-phenylpyridinium (MPP+) consistently induced NLRP3 inflammasome activation upon ATP or nigericin treatment. These findings reveal novel initiating roles of the neurotoxins MPTP or MPP+ in NLRP3 activation. Subsequently, NLRP3 inflammasome-activated microglia induced severe neuronal death in a microglia-neuron co-culture model. In addition, microglia-specific expression of NLRP3 mutants based on Cx3Cr1CreER significantly exacerbated motor dysfunction and dopaminergic neuron loss in MPTP-treated mice. In summary, our results indicate that activation of the microglia NLRP3 inflammasome plays a key role in MPTP-induced neurodegeneration in Parkinson's disease. [6] References: [1]. Langston JW, Irwin I. MPTP neurotoxicity: an overview and characteristics of the toxic phases. II. Selective accumulation of MPP in the substantia nigra: a key (problem) of neurotoxicity. Life Sciences, 1985, 36, No. 3, 201-12. [2]. Hsu KS et al. 4-Phenylepyridine enhances the effect of MPTP on phrenic-diaphragmatic neuromuscular blockade in mice. Neuropharmacology, 1993, 32, No. 9, 877-83. [3]. Sun XL et al. In LPS and MPTP models, Gas1 upregulation can induce and promote apoptosis in reactive astrocytes of the substantia nigra. Journal of Neuroinflammation. 8 July 2016; 13(1):180. [4]. Jackson-Lewis V, Przedborski S. MPTP mouse Parkinson's disease model protocol. Nat Protoc. 2007;2(1):141-51. [5]. Rabaneda-Lombarte N et al. CD200R1 microglia inhibitory receptor as a therapeutic target in MPTP Parkinson's disease model. J Neuroinflammation. 6 April 2021; 18(1):88. [6]. Lee et al. MPTP-driven microglia NLRP3 inflammasome activation plays a central role in dopaminergic neurodegeneration. Cell Death Differ. Jan 2019; 26(2):213-228. MPTP hydrochloride is a synthetic compound widely used to induce experimental Parkinson's disease models in rodents and non-human primates, mimicking the pathological and behavioral characteristics of human Parkinson's disease [1]. The neurotoxic mechanism of MPTP hydrochloride involves three key steps: entry into the brain, conversion to MPP+ via MAO-B, and MPP+-mediated inhibition of mitochondrial complex I, leading to oxidative stress, ATP depletion, and apoptosis of dopaminergic neurons [4]. MPTP hydrochloride has no clinical therapeutic applications; its main use is in preclinical studies to investigate the pathogenesis of Parkinson's disease and evaluate potential neuroprotective agents [3]. |
| Molecular Formula |
C12H16CLN
|
|
|---|---|---|
| Molecular Weight |
209.72
|
|
| Exact Mass |
209.1
|
|
| CAS # |
23007-85-4
|
|
| Related CAS # |
|
|
| PubChem CID |
161406
|
|
| Appearance |
White to off-white solid powder
|
|
| Boiling Point |
258.6ºC at 760mmHg
|
|
| Melting Point |
254 - 257 °C
|
|
| Flash Point |
105.8ºC
|
|
| Vapour Pressure |
0.0136mmHg at 25°C
|
|
| LogP |
3.145
|
|
| Hydrogen Bond Donor Count |
1
|
|
| Hydrogen Bond Acceptor Count |
1
|
|
| Rotatable Bond Count |
1
|
|
| Heavy Atom Count |
14
|
|
| Complexity |
189
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
CN1CCC(=CC1)C2=CC=CC=C2.Cl
|
|
| InChi Key |
KOWJANGMTAZWDT-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C12H15N.ClH/c1-13-9-7-12(8-10-13)11-5-3-2-4-6-11;/h2-7H,8-10H2,1H3;1H
|
|
| Chemical Name |
1-methyl-4-phenyl-3,6-dihydro-2H-pyridine;hydrochloride
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.67 mg/mL (7.96 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 16.7 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.67 mg/mL (7.96 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 16.7 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 1.67 mg/mL (7.96 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 5%DMSO+ 40%PEG300+ 5%Tween 80+ 50%ddH2O: 2.1mg/ml (10.01mM) Solubility in Formulation 5: ≥ 100 mg/mL (476.83 mM) (saturation unknown) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.7683 mL | 23.8413 mL | 47.6826 mL | |
| 5 mM | 0.9537 mL | 4.7683 mL | 9.5365 mL | |
| 10 mM | 0.4768 mL | 2.3841 mL | 4.7683 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
 |
|---|
 |
 |
 |
|