
| Size | Price | Stock | Qty |
|---|---|---|---|
| 2mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
ML346 (CID767276) is a novel and potent activator of heat shock protein 70 (Hsp70) (EC50 = 4600 nM in HeLa cell toxicity assay). ML346 is a member of the barbituric acid class of substances that, in conformational disease models, induce HSF-1-dependent chaperone expression and restore folding of proteins.
| Targets |
HSP70 ( EC50 = 4.6 μM )
Protein homeostasis (proteostasis) network components: ML346 modulates the proteostasis network, potentially targeting molecular chaperones (e.g., heat shock proteins) and/or protein degradation pathways (e.g., ubiquitin-proteasome system, UPS); no specific IC₅₀, Ki, or EC₅₀ values for individual molecular targets were reported [1] - Mutant cystic fibrosis transmembrane conductance regulator (ΔF508-CFTR) protein: ML346 enhances the folding and trafficking of ΔF508-CFTR to the plasma membrane; the EC₅₀ for restoring ΔF508-CFTR-mediated chloride ion transport in CFBE41o⁻ cells (a human bronchial epithelial cell line expressing ΔF508-CFTR) was approximately 1.2 μM [1] - Mutant huntingtin (mHtt) protein (with expanded polyglutamine repeats): ML346 reduces the aggregation of mHtt (Q44-GFP) in HEK293T cells; no specific EC₅₀ value was reported [2] |
|---|---|
| ln Vitro |
ML346 is a newly discovered Hsp70 activator, exhibiting an EC50 of 4600 nM in HeLa cells. Proteostasis, CFTR-mediated iodide conductance, and proper folding of proteins expressed in two distinct cellular compartments are all restored by ML346 at 10 μM. In WT MEF cells, ML346 (Compound F1) strongly induces Hsp70, the oxidative stress response genes HO1 and GCLM, as well as a 2.5-fold upregulation of BiP. It also induces multiple responses. ML346 (0.5-25 μM) doubles the protection against H2O2-induced apoptosis and shows cytoprotective effects in cells following a 35-minute severe heat shock.
Restoration of ΔF508-CFTR function in bronchial epithelial cells: In CFBE41o⁻ cells (expressing ΔF508-CFTR), ML346 (0.1-10 μM) dose-dependently increased plasma membrane localization of ΔF508-CFTR (detected via immunofluorescence and cell surface biotinylation/Western blot). At 10 μM, plasma membrane ΔF508-CFTR levels were increased by ~3.5-fold compared to vehicle control. Concurrently, ML346 restored chloride ion transport (measured via Ussing chamber assay) with an EC₅₀ of ~1.2 μM; at 10 μM, chloride transport was ~60% of that observed in cells expressing wild-type CFTR [1] - Reduction of mHtt aggregation in HEK293T cells: In HEK293T cells transiently transfected with mHtt-Q44-GFP (a fluorescently tagged mHtt variant), ML346 (1-20 μM) dose-dependently decreased the number of mHtt aggregates (detected via fluorescence microscopy). At 20 μM, the percentage of cells with mHtt aggregates was reduced from ~45% (vehicle control) to ~12%. This effect was not associated with decreased mHtt protein expression (measured via Western blot) or cytotoxicity (assessed via MTT assay) [2] - Enhancement of α-synuclein clearance in SH-SY5Y cells: In SH-SY5Y cells (human neuroblastoma cell line) expressing α-synuclein-GFP (a protein linked to Parkinson’s disease), ML346 (5-20 μM) increased the clearance of α-synuclein-GFP (measured via flow cytometry and Western blot). At 20 μM, α-synuclein-GFP protein levels were reduced by ~40% compared to control, without affecting cell viability [2] |
| ln Vivo |
Efficacy in ΔF508-CFTR transgenic mouse model: In ΔF508-CFTR knock-in mice (6-8 weeks old), ML346 was administered via oral gavage at a dose of 50 mg/kg once daily for 7 days. Lung tissues were collected, and ΔF508-CFTR localization was assessed via immunofluorescence. ML346 significantly increased plasma membrane localization of ΔF508-CFTR in bronchial epithelial cells (by ~2.8-fold vs. vehicle control) and improved lung chloride transport (measured via ex vivo Ussing chamber assay) by ~35% [1]
- Efficacy in mHtt transgenic Drosophila model: In Drosophila melanogaster expressing mHtt (Q93) in the nervous system (which exhibit locomotor deficits), ML346 was administered via food at a concentration of 10 μM for 10 days. Locomotor activity (measured via climbing assay) was significantly improved: the percentage of flies able to climb 8 cm in 10 seconds increased from ~30% (vehicle control) to ~65%. Additionally, ML346 reduced mHtt aggregate formation in fly brains (detected via immunohistochemistry) by ~40% [2] |
| Enzyme Assay |
The procedure involves incubating HeLa cells for three or six hours with either DMSO (negative control), the PRs A1, A3, and ML346 (F1), or the positive controls MG132 (10 μM) and lactacystin (6 μM). After lysing the cells for five minutes on ice in homogenization buffer (50 mM Tris-HCl, pH7.5, 250 mM sucrose, 5 mM MgCl2, 2 mM ATP, 1 mM DTT, 0.5 mM EDTA, and 0.025% digitonin), the total protein content of the whole cell extract is calculated. The assay buffer (50 mM Tris-HCl, pH 7.5, 40 mM KCl, 5 mM MgCl2, 0.5 mM ATP, 1 mM DTT, 0.05 mg/mL BSA) is mixed with 3 μg of whole cell extracts in a black 96-well plate. The reaction is started by adding a 2× (200 μM) fluorogenic peptide substrate Suc-LLVY-AMC. Fluorescence is measured with a Synergy H4 multi-mode microplate reader every ten minutes[2].
Ubiquitin-proteasome system (UPS) activity assay: The assay was performed using a fluorogenic proteasome substrate (e.g., Suc-LLVY-AMC, specific for the chymotrypsin-like activity of the 20S proteasome) in HEK293T cell lysates. Reaction mixtures contained 50 μg of cell lysate, 50 mM Tris-HCl (pH 7.5), 2 mM ATP, 5 mM MgCl₂, and different concentrations of ML346 (0.1-20 μM). After preincubation at 37°C for 15 minutes, 100 μM Suc-LLVY-AMC was added, and fluorescence intensity (excitation: 380 nm; emission: 460 nm) was measured every 5 minutes for 1 hour. Proteasome activity was calculated as the rate of fluorescence increase relative to vehicle control. ML346 (10-20 μM) increased proteasome activity by ~25-35% [2] - Heat shock protein 70 (Hsp70) ATPase activity assay: Recombinant human Hsp70 was used, and the assay was based on the detection of inorganic phosphate (Pi) released from ATP hydrolysis. Reaction mixtures contained 2 μM Hsp70, 50 mM HEPES (pH 7.4), 10 mM KCl, 2 mM MgCl₂, 1 mM ATP, and ML346 (0.5-10 μM). Mixtures were incubated at 37°C for 30 minutes, and Pi concentration was measured using a colorimetric Pi detection kit. ML346 (5-10 μM) increased Hsp70 ATPase activity by ~40-50%, indicating enhanced chaperone function [1] |
| Cell Assay |
HeLa cells are plated in 100 μL of DMEM supplemented with 10% FBS and 1% Pen/Strep/Neo at a density of 10,000 cells per well in black 96-well plates. Before adding the compound, the plates are incubated for 16 hours at 37°C, 5% CO2, and 95% relative humidity. To the sample or control wells, 1 μL of hit compounds (ML346) in DMSO or DMSO alone is added, respectively. After that, the plates are kept in the incubator for a full day. Following incubation, 200 μL of PBS is used to wash the cells twice, and 200 μL of calcein AM (1 μg/mL) solution is added to each well. Following a 45-minute incubation period at 37°C and 5% CO2, the cells are fluoresced using an Analyst GT multimode reader. The expressed percentage of cytotoxicity is in relation to wells containing cells that were treated with DMSO alone (100%)[2].
ΔF508-CFTR plasma membrane localization assay in CFBE41o⁻ cells: CFBE41o⁻ cells were cultured in DMEM/F-12 medium supplemented with 10% FBS until 80% confluence. Cells were treated with ML346 (0.1-10 μM) or vehicle for 24 hours. For cell surface biotinylation: cells were washed with ice-cold PBS, incubated with sulfo-NHS-SS-biotin (a membrane-impermeable biotin reagent) on ice for 30 minutes, and then quenched with glycine. Cell lysates were prepared, and biotinylated proteins were pulled down with streptavidin-agarose beads. Western blot was performed using an anti-CFTR antibody to detect plasma membrane ΔF508-CFTR. For immunofluorescence: cells were fixed with 4% paraformaldehyde, permeabilized (or not, for surface staining), incubated with anti-CFTR antibody and a fluorescent secondary antibody, and imaged via confocal microscopy. Plasma membrane fluorescence intensity was quantified using image analysis software [1] - mHtt aggregation assay in HEK293T cells: HEK293T cells were transfected with a plasmid encoding mHtt-Q44-GFP using a transfection reagent. After 24 hours, cells were treated with ML346 (1-20 μM) or vehicle for 48 hours. Cells were fixed with 4% paraformaldehyde, and GFP fluorescence was imaged via fluorescence microscopy. The number of cells with mHtt aggregates (defined as bright, punctate GFP foci) was counted in 10 random fields per well (n=3 wells per group). Cell viability was assessed via MTT assay: cells were incubated with MTT reagent for 4 hours, formazan crystals were dissolved in DMSO, and absorbance at 570 nm was measured [2] - α-synuclein clearance assay in SH-SY5Y cells: SH-SY5Y cells were transfected with α-synuclein-GFP plasmid. After 24 hours, cells were treated with ML346 (5-20 μM) or vehicle for 36 hours. For Western blot: cell lysates were prepared, and α-synuclein-GFP levels were detected using an anti-GFP antibody, with GAPDH as a loading control. For flow cytometry: cells were trypsinized, washed with PBS, and GFP fluorescence intensity was measured using a flow cytometer. The mean fluorescence intensity (MFI) was normalized to vehicle control to assess α-synuclein clearance [2] |
| Animal Protocol |
ΔF508-CFTR knock-in mouse model experiment: Male and female ΔF508-CFTR knock-in mice (6-8 weeks old, 20-25 g) were randomly divided into vehicle and ML346 groups (n=6 per group). ML346 was dissolved in a solution of 10% DMSO and 90% corn oil to a concentration of 5 mg/mL. Mice were administered ML346 via oral gavage at a dose of 50 mg/kg once daily for 7 days; the vehicle group received 10% DMSO/90% corn oil alone. On day 8, mice were euthanized, and lungs were excised. For ex vivo Ussing chamber assay: lung slices containing bronchial segments were mounted in Ussing chambers, and transepithelial chloride current was measured before and after addition of a CFTR activator. For immunofluorescence: lung sections were fixed, stained with anti-CFTR antibody and fluorescent secondary antibody, and imaged via confocal microscopy [1] - mHtt transgenic Drosophila model experiment: Transgenic Drosophila melanogaster (UAS-mHtt-Q93-GFP; elav-Gal4) were reared on standard cornmeal-agar food at 25°C. Newly eclosed adult flies (3-5 days old) were divided into vehicle and ML346 groups (n=50 per group). ML346 was dissolved in ethanol and added to molten food to a final concentration of 10 μM; the vehicle group received ethanol alone (final ethanol concentration <1%, non-toxic). Flies were fed the treated food for 10 days, with food replaced every 2 days. Locomotor activity was assessed via climbing assay: flies were placed in a vertical tube, and the number of flies that climbed 8 cm in 10 seconds was counted (3 trials per group). After behavioral testing, fly brains were dissected, fixed, stained with anti-GFP antibody, and imaged via fluorescence microscopy to quantify mHtt aggregates [2] |
| Toxicity/Toxicokinetics |
In vitro cytotoxicity: In CFBE41o⁻, HEK293T and SH-SY5Y cells, ML346 (at a concentration up to 20 μM, treated for 48 hours) did not reduce cell viability (MTT assay: viability >90%, compared with solvent control group) [1][2]
- In vivo toxicity: In ΔF508-CFTR knock-in mice (oral administration of 50 mg/kg, 7 days), ML346 did not cause significant changes in body weight, liver function indicators (ALT, AST) or kidney function indicators (BUN, creatinine) compared with solvent control group. In transgenic fruit flies (feed concentration 10 μM, 10 days), no increase in mortality was observed (survival rate >85%, compared with solvent control group) [1][2] |
| References |
|
| Additional Infomation |
ML346 is a novel small molecule protein homeostasis regulator developed by the Molecular Library Project of the National Institutes of Health (NIH) to treat protein conformation disorders—diseases caused by protein misfolding, aggregation, or impaired transport [1]
- Mechanism of action: ML346 regulates the protein homeostasis network through two complementary pathways: (1) enhancing the ATPase activity of molecular chaperones (e.g., Hsp70) to promote the correct folding of misfolded proteins (e.g., ΔF508-CFTR); (2) enhancing the activity of the ubiquitin-proteasome system (UPS) to accelerate the degradation of toxic aggregates (e.g., mHtt, α-synuclein) [1][2] - Potential therapeutic indications: ML346 is indicated in cystic fibrosis (ΔF508-CFTR mutation), Huntington's disease (mHtt... It has shown preclinical efficacy in models of aggregation and other potential protein conformational diseases (e.g., Parkinson's disease, α-synucleinopathy); however, it has not yet received FDA approval or has clinical trial data reported [1][2] |
| Molecular Formula |
C14H12N2O4
|
|
|---|---|---|
| Molecular Weight |
272.26
|
|
| Exact Mass |
272.079
|
|
| Elemental Analysis |
C, 61.76; H, 4.44; N, 10.29; O, 23.51
|
|
| CAS # |
100872-83-1
|
|
| Related CAS # |
|
|
| PubChem CID |
767276
|
|
| Appearance |
Orange to red solid powder
|
|
| Density |
1.4±0.1 g/cm3
|
|
| Index of Refraction |
1.669
|
|
| LogP |
0.88
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
4
|
|
| Rotatable Bond Count |
3
|
|
| Heavy Atom Count |
20
|
|
| Complexity |
452
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
O(C([H])([H])[H])C1C([H])=C([H])C(=C([H])C=1[H])/C(/[H])=C(\[H])/C(/[H])=C1\C(N([H])C(N([H])C\1=O)=O)=O
|
|
| InChi Key |
IXYLVJHFJKDHRM-NSCUHMNNSA-N
|
|
| InChi Code |
InChI=1S/C14H12N2O4/c1-20-10-7-5-9(6-8-10)3-2-4-11-12(17)15-14(19)16-13(11)18/h2-8H,1H3,(H2,15,16,17,18,19)/b3-2+
|
|
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.25 mg/mL (4.59 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.6730 mL | 18.3648 mL | 36.7296 mL | |
| 5 mM | 0.7346 mL | 3.6730 mL | 7.3459 mL | |
| 10 mM | 0.3673 mL | 1.8365 mL | 3.6730 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
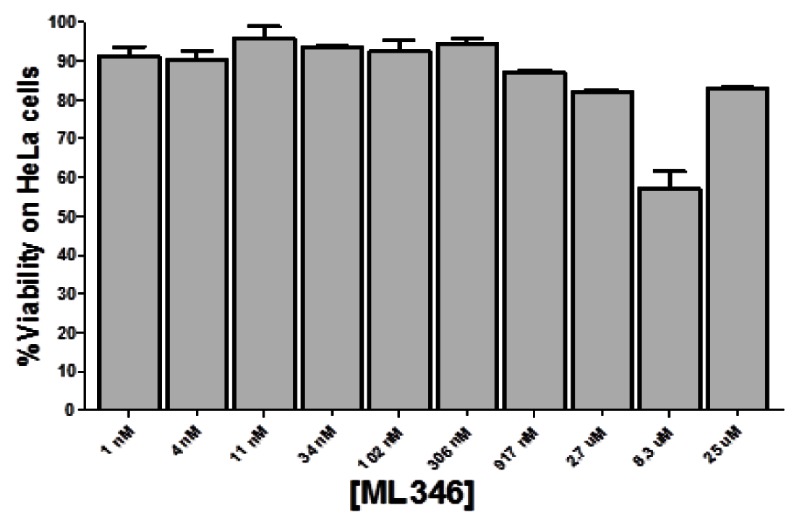 Probe ML346 is not toxic to HeLa cells.Probe Reports from the NIH Molecular Libraries Program |
|---|
 Probe ML346 (“F1”) induces Hsp70 mRNA expression.Probe Reports from the NIH Molecular Libraries Program |
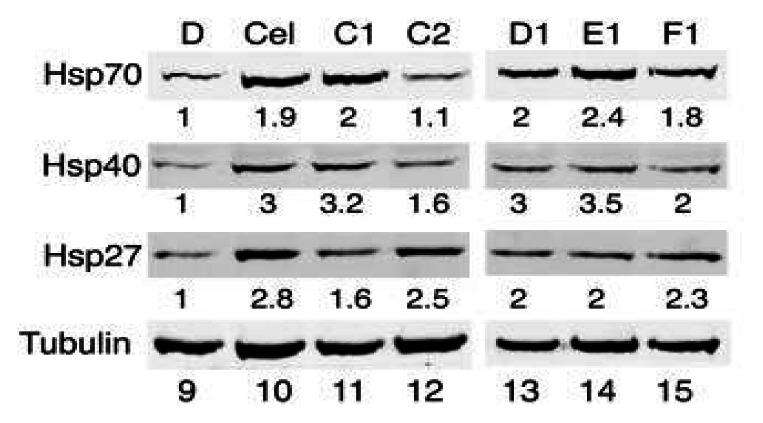 Probe ML346 (“F1”) induces chaperone protein expression.Probe Reports from the NIH Molecular Libraries Program |
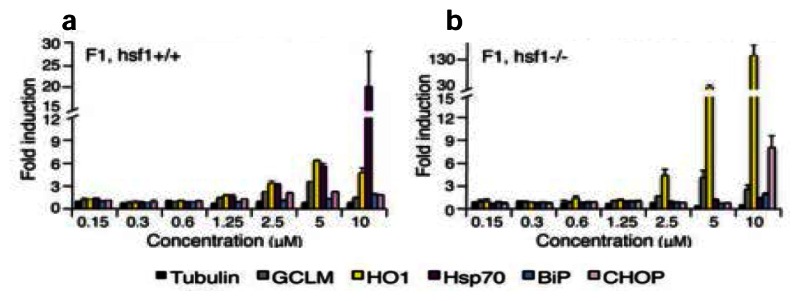 a,Wild type (hsf-1+/+) andb,HSF-1 null (hsf-1−/−) (MEFs) were treated for 4 h with probeML346(compound F1) at the indicated concentrations. |
|---|
 Probe ML346 restores proteostasis. |
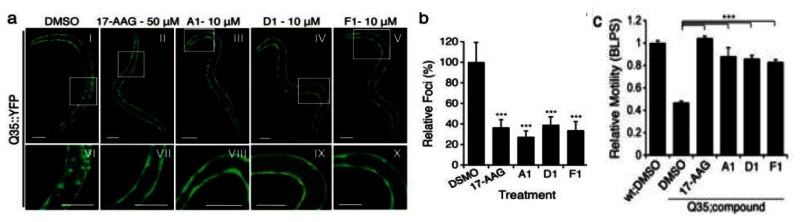 Probe ML346 (compound F1) reduces aggregation/toxicity inC. elegansmodels of diseases associated with polyQ expansions.Probe Reports from the NIH Molecular Libraries Program |