
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg | |||
| Other Sizes |
Purity: ≥98%
ML-161 is a novel and potent allosteric inhibitor of PAR1 (proteinase-activated receptor 1) on platelets with IC50 of 0.26 μM. ML161 exhibits a different selectivity with the reported ALK inhibitor crizotinib. ML161 treatment was found to inhibit the activation of thrombin-induced platelets in a dose-dependent manner through the detection of P-selectin expression in human platelets. ML161 demonstrated a significant suppression of P-selectin expression in a dose-dependent manner with an EC50 value of 0.3μM when tested with granule secretion. Additionally, it prevented SFLLRN-induced thrombus formation. With an IC50 value of 0.26 µM, ML-161 inhibits P-selectin'ssurfaceexpression that is induced by the peptide SFLLRN. When thrombin or a PAR1 peptide agonist were used to cause platelet aggregation, ML161 prevented it, but not when using multiple other platelet agonists.
| Targets |
PAR1 ( IC50 = 0.26 μM )
ML161 (Parmodulin 2) targets protease-activated receptor 1 (PAR1) as a reversible allosteric inhibitor, with an IC₅₀ value of 0.8 μM (PAR1-driven calcium mobilization assay in HUVECs) [1] ML161 (Parmodulin 2) inhibits PAR1-mediated platelet aggregation with an IC₅₀ value of 1.2 μM (arachidonic acid-induced platelet aggregation assay) [3] |
|---|---|
| ln Vitro |
In vitro activity: Parmodulin 2 (ML161; 10 µM; for 30 minutes) blocks proinflammatory signaling in endothelial HUVECs cells[3].
In HUVECs: ML161 (Parmodulin 2) (0.1–10 μM) dose-dependently inhibited PAR1 agonist (TFLLR-NH₂)-induced calcium mobilization, reducing fluorescence intensity by 35–90% (Fluo-4 AM staining, microplate reader) [1] - The compound blocked PAR1-mediated ERK1/2 and Akt phosphorylation in HUVECs: 1 μM reduced p-ERK1/2 by 65% and p-Akt by 58% (Western blot) [1] - In human platelets: ML161 (Parmodulin 2) (0.5–10 μM) inhibited arachidonic acid-induced aggregation by 28–82%, and thrombin-induced aggregation by 32–78% (light transmission aggregometry) [3] - It showed no significant binding to PAR2/PAR4 (IC₅₀ > 20 μM) or G protein-coupled receptors (GPCRs) like CXCR4 (Ki > 20 μM), demonstrating high PAR1 selectivity [1] - No obvious cytotoxicity to HUVECs or human platelets at concentrations up to 20 μM (MTT assay for HUVECs; viability >90% for platelets) [1][3] |
| ln Vivo |
Parmodulin 2 (ML161; 5 mg/kg; IV) significantly prevents platelet thrombus formation, with aa 73% reduction in AUC (area under the curve)[2].
Parmodulin 2 does not increase bleeding time; instead, it prevents platelet thrombus formation in vivo. Platelet aggregation is selectively inhibited by Parmodulin 2 via the α2A-adrenergic receptor and Par1[2]. In mouse carotid artery FeCl₃-induced thrombosis model: Intravenous injection of ML161 (Parmodulin 2) (5, 10 mg/kg) dose-dependently prolonged thrombosis occlusion time by 45% and 72% compared to vehicle control [3] - The compound reduced thrombus weight by 38% (5 mg/kg) and 62% (10 mg/kg) in rat arteriovenous shunt thrombosis model [3] - Unlike vorapaxar (a PAR1 orthosteric antagonist), ML161 (Parmodulin 2) (10 mg/kg iv) did not induce endothelial injury in mouse aorta (histopathological analysis: no increase in endothelial cell apoptosis or permeability) [3] - No significant bleeding risk was observed at effective antithrombotic doses (10 mg/kg iv): tail bleeding time increased by <20% compared to vehicle [3] |
| Enzyme Assay |
In order to determine possible targets of hit compounds, a high-throughput screen of the NIH-MLSMR compound collection and a number of secondary assays were used to find a 1,3-diaminobenzene scaffold that targets protease-activated receptor 1 (PAR1). We hereby present further studies of the structure-activity relationship (SAR) that characterize the conditions for activity at PAR1 and pinpoint analogues that are stable in plasma that exhibit nanomolar inhibition of PAR1-mediated platelet activation. Compound 4 was declared as a probe (ML161) with the NIH Molecular Libraries Program. This substance prevented platelet aggregation brought on by thrombin or a PAR1 peptide agonist, but not by a number of other platelet agonists. According to preliminary research, ML161 is an allosteric inhibitor of PAR1. The identification of antithrombotics with an enhanced safety profile may benefit from these findings.
PAR1-driven calcium mobilization assay: HUVECs were loaded with Fluo-4 AM fluorescent probe, pretreated with serial dilutions of ML161 (Parmodulin 2) for 30 minutes, then stimulated with PAR1-specific agonist TFLLR-NH₂ (10 μM). Fluorescence intensity was measured in real-time to quantify calcium influx, and IC₅₀ was calculated [1] - PAR1 allosteric binding assay: Recombinant PAR1 extracellular domain was immobilized on sensor chips. ML161 (Parmodulin 2) was injected at different concentrations, and binding affinity was measured by surface plasmon resonance (SPR) to confirm allosteric interaction (no competition with orthosteric ligand thrombin) [1] |
| Cell Assay |
HUVEC calcium mobilization and signaling assay: HUVECs were seeded in 96-well plates (2×10⁴ cells/well), loaded with Fluo-4 AM, treated with ML161 (Parmodulin 2) (0.1–10 μM), and stimulated with TFLLR-NH₂. Fluorescence was detected; cells were lysed for Western blot analysis of p-ERK1/2 and p-Akt [1]
- Platelet aggregation assay: Human platelets were isolated from fresh blood, resuspended in buffer, pretreated with ML161 (Parmodulin 2) (0.5–10 μM) for 15 minutes, then induced with arachidonic acid (100 μM) or thrombin (0.1 U/mL). Aggregation was monitored by light transmission at 620 nm [3] - PAR subtype selectivity assay: PAR2/PAR4-expressing HEK293 cells were loaded with Fluo-4 AM, treated with ML161 (Parmodulin 2) (0.1–20 μM), and stimulated with respective agonists (SLIGKV-NH₂ for PAR2; AYPGKF-NH₂ for PAR4) to assess cross-reactivity [1] |
| Animal Protocol |
C57BL/6J wild type mice
5 mg/kg (Pharmacokinetic Analysis) IV Mouse carotid artery thrombosis model: Male C57BL/6 mice (20–25 g) were anesthetized, and the left carotid artery was exposed. FeCl₃ solution was applied to the artery to induce thrombosis. ML161 (Parmodulin 2) was administered intravenously (5, 10 mg/kg) 15 minutes before FeCl₃ application. Blood flow was monitored with a Doppler flowmeter to record occlusion time [3] - Rat arteriovenous shunt thrombosis model: Male Sprague-Dawley rats (250–300 g) were implanted with an arteriovenous shunt. ML161 (Parmodulin 2) (5, 10 mg/kg iv) was given 15 minutes before shunt placement. After 30 minutes, the thrombus was collected and weighed [3] - Drug formulation: ML161 (Parmodulin 2) was dissolved in dimethyl sulfoxide (DMSO) and diluted with normal saline to a final DMSO concentration of ≤5% [1][3] - Endothelial injury assessment: Mice were euthanized 24 hours after drug administration. Aorta tissues were collected, fixed, and stained with hematoxylin-eosin (HE) and TUNEL to evaluate endothelial morphology and apoptosis [3] |
| Toxicity/Toxicokinetics |
In vitro toxicity: CC₅₀ > 20 μM in HUVEC cells; no significant platelet damage was observed at concentrations up to 20 μM (lactate dehydrogenase (LDH) release assay) [1][3]
- Acute in vivo toxicity: No death or behavioral abnormalities (drowsiness, hemorrhage) were observed in mice after intravenous injection of ML161 (Parmodulin 2) at doses up to 50 mg/kg [3] - Plasma protein binding: 85% (human plasma, ultrafiltration) [1] - No endothelial damage: Unlike vorapaxaar, ML161 (Parmodulin 2) (10 mg/kg intravenous injection) did not increase the permeability of mouse aortic endothelial cells or induce apoptosis [3] |
| References | |
| Additional Infomation |
2-Bromo-N-[3-(1-Oxobutylamino)phenyl]benzamide belongs to the benzamide class of compounds. ML161 (Parmodulin 2) is a synthetic small molecule reversible PAR1 allosteric inhibitor belonging to the Parmodulin class of compounds [1][3]. Its mechanism of action is to bind to the allosteric site on PAR1, thereby preventing thrombin or other PAR1 agonists from activating the receptor without interfering with the binding of the normal ligand [1]. It exerts its antithrombotic effect by inhibiting PAR1-mediated platelet aggregation and endothelial cell signaling. Because it does not cause endothelial damage, its safety is superior to normal PAR1 antagonists (such as vorapaxar) [3]. This compound is a promising candidate for antiplatelet therapy and has potential application value in preventing thrombosis-related diseases (such as myocardial infarction, stroke) [1][3].
|
| Molecular Formula |
C17H17BRN2O2
|
|
|---|---|---|
| Molecular Weight |
361.23
|
|
| Exact Mass |
360.047
|
|
| Elemental Analysis |
C, 56.52; H, 4.74; Br, 22.12; N, 7.75; O, 8.86
|
|
| CAS # |
423735-93-7
|
|
| Related CAS # |
|
|
| PubChem CID |
1048267
|
|
| Appearance |
White to off-white solid powder
|
|
| Density |
1.4±0.1 g/cm3
|
|
| Boiling Point |
465.7±30.0 °C at 760 mmHg
|
|
| Flash Point |
235.4±24.6 °C
|
|
| Vapour Pressure |
0.0±1.2 mmHg at 25°C
|
|
| Index of Refraction |
1.656
|
|
| LogP |
3.45
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
2
|
|
| Rotatable Bond Count |
5
|
|
| Heavy Atom Count |
22
|
|
| Complexity |
389
|
|
| Defined Atom Stereocenter Count |
0
|
|
| SMILES |
BrC1=C([H])C([H])=C([H])C([H])=C1C(N([H])C1=C([H])C([H])=C([H])C(=C1[H])N([H])C(C([H])([H])C([H])([H])C([H])([H])[H])=O)=O
|
|
| InChi Key |
DFOVLSMXPWPCFH-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C17H17BrN2O2/c1-2-6-16(21)19-12-7-5-8-13(11-12)20-17(22)14-9-3-4-10-15(14)18/h3-5,7-11H,2,6H2,1H3,(H,19,21)(H,20,22)
|
|
| Chemical Name |
2-bromo-N-[3-(butanoylamino)phenyl]benzamide
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.92 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (6.92 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (6.92 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7683 mL | 13.8416 mL | 27.6832 mL | |
| 5 mM | 0.5537 mL | 2.7683 mL | 5.5366 mL | |
| 10 mM | 0.2768 mL | 1.3842 mL | 2.7683 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
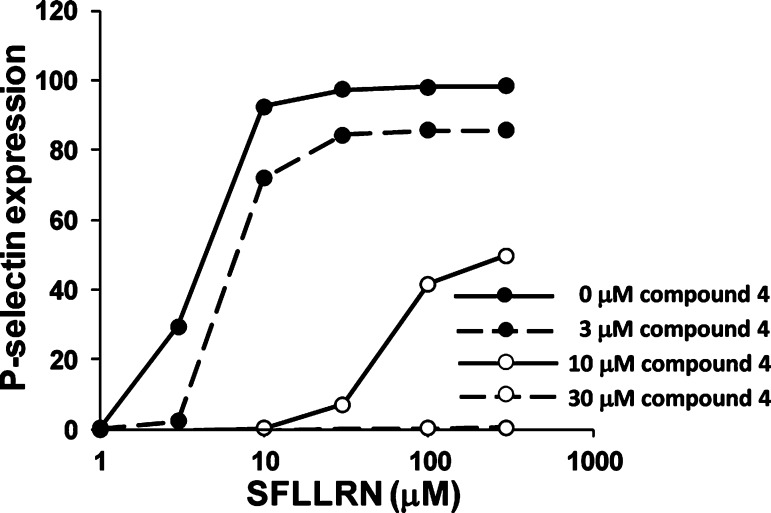 Dose–response curves of SFLLRN-induced P-selectin expression in the presence of varying concentrations of the PAR1 inhibitor4.ACS Med Chem Lett.2012 Mar 8;3(3):232-237. |
|---|
Synthesis of4(ML161).ACS Med Chem Lett.2012 Mar 8;3(3):232-237. |