
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
Purity: ≥98%
Mirabegron (formerly YM-178; YM178; Myrbetriq; Betanis; Betmiga) is a potent and selective β3-adrenoceptor agonist with similar effects to antimuscarinic medications. It stimulates the β3-adrenoceptor with an EC50 of 22.4 nM. The medication mirabegron is authorized for the management of overactive bladder. When the β3 adrenergic receptor in the bladder's detrusor muscle is activated by mirabegron, the muscle relaxes and the bladder's capacity increases.
| Targets |
β3-adrenoceptor ( EC50 = 22.4 nM )
β3-adrenoceptor (EC50 = 24 nM for cAMP accumulation; Ki = 7.4 nM for radioligand binding) [1] |
|---|---|
| ln Vitro |
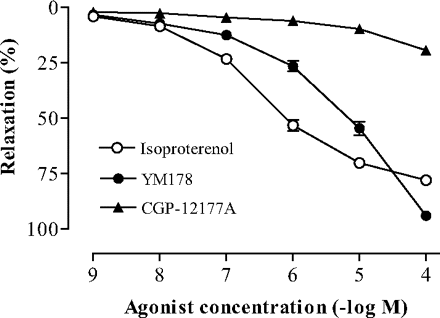
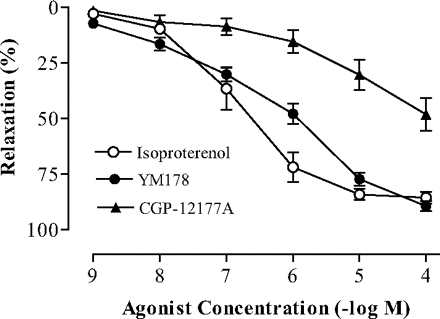
In vitro activity: Mirabegron concentration-dependently increases the accumulation of cAMP in CHO cells expressing human 3-adrenoceptors (ARs) with I.A. of 0.8. Mirabegron barely affects 1- and 2-ARs agonistically. With EC50 values of 5.1 μM and 0.78 μM, respectively, mirabegron concentration-dependently relaxes rat and human bladder smooth muscle strips precontracted with 10-6 M or 10-7 M carbachol. Mirabegron's maximal relaxant effects are 94.0% and 89.4% of those of carbachol, respectively. Mirabegron is a time-dependent inhibitor of CYP2D6 in the presence of NADPH, as demonstrated by the fact that after 30 minutes of preincubation, the IC50 value in human liver microsomes dropped from 13 to 4.3 μM. Mirabegron inhibits CYP2D6 in part through an irreversible or quasi-irreversible metabolism-dependent mechanism.
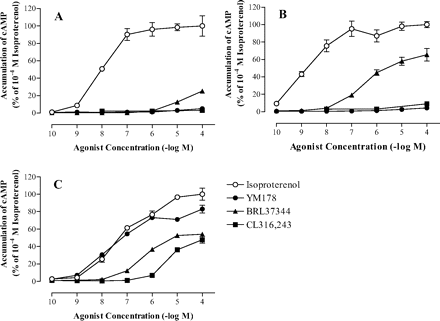
Mirabegron (YM178) is a selective β3-adrenoceptor agonist with potent activity on bladder smooth muscle. In isolated rat bladder strips, it induced concentration-dependent relaxation of carbachol-precontracted muscles, with an EC50 of 31 nM. Maximal relaxation at 1 μM was ~85%, which was blocked by the β3-adrenoceptor antagonist L-748,337 [1] It showed high selectivity for β3-adrenoceptors, with negligible activity on β1 (EC50 > 10 μM) and β2 (EC50 > 10 μM) adrenoceptors in cAMP accumulation assays. In radioligand binding studies, it exhibited minimal affinity for β1 (Ki = 1200 nM) and β2 (Ki = 890 nM) receptors [1] In cultured rat bladder smooth muscle cells, Mirabegron (YM178) (10-1000 nM) dose-dependently increased intracellular cAMP levels, with a maximal 3.8-fold increase at 1 μM, confirming β3-adrenoceptor-mediated signaling [1] |
| ln Vivo |
Mirabegron causes rats given anesthesia to experience a dose-dependent decrease in the frequency of rhythmic bladder contractions. 3 mg intravenously/kg. Mirabegron reduces the frequency to two counts per minute. Mirabegron does not lessen the rhythmic bladder contraction's amplitude. In rats, mirabegron reduces both bladder microcontractions and primary bladder afferent activity. Mechanosensitive single-unit afferent activities (SAAs) of Aδ fibers in response to bladder filling are inhibited by mirabegron (0.3 and 1 mg/kg). Only at 1 mg/kg of Mirabegron treatment do SAAs of C-fibers decrease. The administration of mirabegron suppresses both the mean bladder pressure and the number of microcontractions when the bladder is in an isovolumetric state. Mirabegron effectively aids in the storage of the bladder. In a dose-dependent manner, mirabegron lowers the intrathecal pressure at rest. Mirabegron reduces the frequency of non-voiding contractions dose-dependently, which is thought to be a sign of an aberrant response in bladder storage. Mirabegron shows no discernible effects on micturition pressure, threshold pressure, voided volume, residual volume, bladder capacity, or the amplitude of nonvoiding contractions.
In anesthetized rats, intravenous administration of Mirabegron (YM178) (0.1-1 mg/kg) dose-dependently increased bladder capacity and micturition volume. At 1 mg/kg, bladder capacity increased by ~40% and micturition volume by ~45% compared to vehicle, without significant changes in heart rate or blood pressure [1] In conscious rats with partial bladder outlet obstruction (a model of overactive bladder), oral administration of Mirabegron (YM178) (3-30 mg/kg) improved bladder function. At 30 mg/kg, it reduced micturition frequency by ~30% and increased average voided volume by ~35%, reversing obstruction-induced bladder hyperactivity [1] |
| Enzyme Assay |
We evaluated the pharmacological characteristics of (R)-2-(2-aminothiazol-4-yl)-4′-{2-[(2-hydroxy-2-phenylethyl)amino]-ethyl} acetanilide (YM178). YM178 increased cyclic AMP accumulation in Chinese hamster ovary (CHO) cells expressing human β3-adrenoceptor (AR). The half-maximal effective concentration (EC50) value was 22.4 nM. EC50 values of YM178 for human β1- and β2-ARs were 10,000 nM or more, respectively. The ratio of intrinsic activities of YM178 versus maximal response induced by isoproterenol (nonselective β-AR agonist) was 0.8 for human β3-ARs, 0.1 for human β1-ARs, and 0.1 for human β2-ARs. [1]
β3-adrenoceptor radioligand binding assay: Prepare membrane homogenates from Chinese hamster ovary (CHO) cells expressing human β3-adrenoceptors. Incubate homogenates with [3H]-CGP12177 (a non-selective β-adrenoceptor ligand) and various concentrations of Mirabegron (YM178) (0.1-1000 nM) at 25°C for 60 minutes. Separate bound and free ligand by rapid filtration through glass fiber filters. Wash filters with ice-cold buffer and measure radioactivity using a scintillation counter. Calculate Ki value from competition binding curves [1] cAMP accumulation assay: Seed CHO cells expressing human β3, β1, or β2 adrenoceptors in 96-well plates and culture until confluent. Treat cells with Mirabegron (YM178) (0.01-100 μM) for 30 minutes in the presence of a phosphodiesterase inhibitor. Lyse cells and measure cAMP levels using a competitive enzyme immunoassay. Calculate EC50 values for each receptor subtype [1] |
| Cell Assay |
In every well of a 24-well culture plate, 10 5 CHO cells are deposited and allowed to grow. A pH 7.4 Hanks balanced salt solution containing 0.1 mM 3-isobutyl-1-methylxanthine is added to each well of the medium three days later. 250 μL of 0.2 M HCl is added to end the incubation of the cells after they have been exposed to each compound (isoproterenol, Mirabegron, BRL37344, and CL316,243) for 10 minutes at 37°C. The compounds are added at final concentrations of 10 -10 to 10 - M. Through radioimmunoassay with a 125 I-cAMP assay system and a gamma counter, the concentration of cAMP in the reaction mixture is determined. The reaction is stopped by adding 400 μL of buffer solution after 50 microliters of reaction mixture and 50 μL of succinyl agent are incubated for 10 minutes at room temperature. For 24 hours at 4°C, 50 microliters of succinylated sample are incubated with 50 μL of 125 I-cAMP and 50 μL of anti-cAMP antibody. After the incubation period, add 250 μL of charcoal suspension, and centrifuge at 2800g for 10 minutes at 4°C. After being transferred into a tube, 250 microliters of supernatant are counted for one minute using a gamma counter. The maximal response of each compound is used to calculate the intrinsic activity (I.A.) relative to isoproterenol for each β-adrenoceptor agonist.
Bladder smooth muscle cell cAMP assay: Isolate bladder smooth muscle cells from adult rats, seed in 24-well plates, and culture in Dulbecco’s modified Eagle’s medium for 5-7 days. Serum-starve cells for 24 hours, then treat with Mirabegron (YM178) (10-1000 nM) for 20 minutes. Add lysis buffer to harvest cells, and determine cAMP concentration using a fluorescence-based assay kit [1] Bladder strip relaxation assay: Dissect bladder tissues from rats, cut into longitudinal strips (2-3 mm wide), and mount in organ baths containing oxygenated Krebs-Ringer solution at 37°C. Precontract muscles with carbachol (1 μM) until a stable contraction is achieved. Add Mirabegron (YM178) (0.1-1000 nM) in a cumulative manner and record tension changes using an isometric transducer. Calculate relaxation percentage relative to precontraction amplitude [1] |
| Animal Protocol |
It uses Wistar rats, both male (350–400 g) and female (225–290 g). In this investigation, the free-form doses of 0.03, 0.1, 0.3, 1, and 3 mg/kg for mirabegron and 0.0272, 0.0907, 0.272, 0.907, and 2.72 mg/kg for oxybutynin were employed.
Rats Anesthetized rat bladder function study: Adult male rats are anesthetized and implanted with a transurethral catheter for bladder filling and pressure recording, and a jugular vein catheter for drug administration. After stabilization, Mirabegron (YM178) is dissolved in physiological saline and administered intravenously at doses of 0.1, 0.3, or 1 mg/kg. Bladder capacity, micturition volume, and voiding pressure are recorded for 60 minutes post-administration [1] Partial bladder outlet obstruction (PBOO) rat model: Adult male rats are anesthetized, and the urethra is partially ligated to induce bladder outlet obstruction. Two weeks after surgery, rats are randomly divided into vehicle and treatment groups. Mirabegron (YM178) is suspended in 0.5% carboxymethylcellulose and administered orally at 3, 10, or 30 mg/kg once daily for 7 days. Conscious rats are placed in metabolic cages to record micturition frequency and voided volume over 24 hours [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
The absolute bioavailability of mirabegron via oral administration was 29% at a 25 mg dose and 35% at a 50 mg dose. The time to peak concentration (Tmax) for sustained-release tablets and suspensions was approximately 3.5 hours, while that for granules was 4–5 hours. The increases in Cmax and AUC were greater than the increases in dose—from 50 mg to 100 mg, Cmax and AUC increased 2.9-fold and 2.6-fold, respectively; while from 50 mg to 200 mg, Cmax and AUC increased 8.4-fold and 6.5-fold, respectively. Steady-state plasma concentrations of mirabegron were reached approximately 7 days after once-daily administration. In healthy volunteers, approximately 55% of the radioactive material was recovered in the urine and 34% in the feces after administration of 160 mg of radiolabeled mirabegron. Approximately 25% of the unmetabolized mirabegron was recovered in the urine but not in the feces. Mirabelon is primarily cleared via active tubular secretion, with glomerular filtration also playing a role. After intravenous administration, the apparent steady-state volume of distribution (Vd) of mirabegron is 1670 L, indicating its widespread distribution. The total plasma clearance after intravenous administration is approximately 57 L/h, with renal clearance accounting for about 25%, or approximately 13 L/h. Metabolism/Metabolites Mirabelon is extensively metabolized through various mechanisms, but after oral administration, the parent drug remains the main circulating component. Possible metabolic pathways and metabolites include amide hydrolysis (M5, M16, M17), glucuronidation (mirabegron O-glucuronide, N-glucuronide, N-carbamoylglucuronide, M12), and secondary amine oxidation or dealkylation (M8, M9, M15). The enzymes responsible for the oxidative metabolism of mirabegron are believed to be CYP3A4 and CYP2D6, while the UDP-glucuronyltransferases responsible for the binding reaction have been identified as UGT2B7, UGT1A3, and UGT1A8. Other enzymes that may be involved in mirabegron metabolism include butylcholinesterase and possible alcohol dehydrogenases. Biological Half-Life The mean terminal elimination half-life for adult patients receiving mirabegron for overactive bladder is approximately 50 hours. For pediatric patients receiving granule formulations for neurogenic detrusor overactive bladder, the mean terminal elimination half-life is approximately 26–31 hours. |
| Toxicity/Toxicokinetics |
Hepatotoxicity
In pre-registration clinical trials, elevated serum transaminases were uncommon and mild in patients treated with mirabegron, with an incidence similar to the placebo group. No clinically significant liver injury occurred in thousands of treated patients. Since mirabegron's approval and widespread use, there have been no published reports of hepatotoxicity. However, the mirabegron product information mentions occasional elevations in ALT and AST during treatment, as well as one case of Stevens-Johnson syndrome with elevated transaminases. Therefore, mirabegron may cause liver injury as part of a systemic hypersensitivity reaction. Probability Score: E (Unproven but suspected rare cause of clinically significant liver injury). Pregnancy and Lactation Effects ◉ Overview of Use During Lactation There is currently no information on the use of mirabegron during lactation. Due to its moderately high protein binding and relatively low bioavailability, breastfed infants may be exposed to lower amounts of the drug. If a mother needs to take mirabegron, this is not a reason to stop breastfeeding, but until more data is available, especially during the nursing of newborns or preterm infants, she may prefer to choose other medications. ◉ Effects on breastfed infants No relevant published information was found as of the revision date. ◉ Effects on lactation and breast milk No relevant published information was found as of the revision date. Protein binding Mirabelon has a protein binding rate of approximately 71% in plasma, primarily binding to albumin and α-1-acid glycoprotein. Acute intravenous toxicity studies in rats showed no deaths at doses up to 10 mg/kg [1]. No significant changes in heart rate, systolic/diastolic blood pressure, or ECG parameters were observed at therapeutic doses (0.1–1 mg/kg, intravenously) under anesthesia. In rats[1], Miraberon (YM178) has a plasma protein binding rate of approximately 71% in rats[1]. |
| References | |
| Additional Infomation |
Mirabegron is a monocarboxylic acid amide formed by the condensation of the carboxyl group of 2-amino-1,3-thiazol-4-ylacetic acid and the aniline group of (1R)-2-{[2-(4-aminophenyl)ethyl]amino}-1-phenylethanol. It is used to treat overactive bladder. It is a β-adrenergic agonist. It belongs to the 1,3-thiazolides, aromatic amides, ethanolamines, and monocarboxylic acid amides. Mirabelon is a sympathomimetic β3-adrenergic receptor agonist used to relax bladder smooth muscle and treat urinary frequency and incontinence. Mirabelon stands out among the many drugs used to treat overactive bladder because, unlike other drugs (such as solifenacin and dafinacin), it lacks significant anticholinergic activity. Anticholinergic activity is both the reason these drugs are effective and the reason for their widespread adverse reactions. Compared to other existing treatment options, mirabegron has a relatively favorable adverse event profile, and its mechanism of action is complementary to previous anticholinergic drugs, thus allowing it to be used in combination with solifenacin for refractory cases. Mirabelon was first approved by the U.S. Food and Drug Administration (FDA) in 2012 under the brand name Myrbetriq for the treatment of overactive bladder in adults. Subsequently, its extended-release granule formulation was approved in March 2021 for the treatment of neurogenic detrusor overactivity in children. Mirabelon is also used in other parts of the world, including Canada, the European Union, and Japan. Mirabelon is a β3-adrenergic agonist. Its mechanism of action is as a β3-adrenergic receptor agonist, cytochrome P450 2D6 inhibitor, cytochrome P450 3A inhibitor, and P-glycoprotein inhibitor. Mirabelon is a β3-adrenergic receptor agonist used to treat overactive bladder. Mirabelon does not cause elevated liver enzymes or clinically significant acute liver injury. Mirabelon is an orally bioavailable β3-adrenergic receptor (ADRB3) agonist with muscle relaxant, neuroprotective, and potential antitumor activities. After oral administration, mirabegron binds to and activates ADRB3, leading to smooth muscle relaxation. Mirabelon also restores sympathetic stimulation in the mesenchymal stem cell (MSC) microenvironment, inhibits the expansion of JAK2-mutant hematopoietic stem cells (HSCs), and blocks the progression of myeloproliferative neoplasms (MPNs). The lack of sympathetic stimulation in the MSC and HSC microenvironments is closely associated with the development and progression of MPNs.
Drug Indications Mirabelon is indicated for the treatment of overactive bladder (OAB), symptoms of which include urinary urgency, frequency, and urge incontinence, either alone or in combination with solifenacin. It is also indicated for the treatment of neurogenic detrusor overactivity (NDO) in children aged 3 years and older and weighing 35 kg or more. Relief of urinary urgency symptoms. Adult patients with overactive bladder may experience increased urination frequency and/or urge incontinence. Treatment of neurogenic detrusor overactive bladder. Treatment of idiopathic overactive bladder. Mechanism of Action: Mirabelon is a potent and selective β3-adrenergic receptor agonist. Activation of β3 receptors relaxes the detrusor smooth muscle during the storage phase of the bladder fill-void cycle, thereby increasing bladder capacity and relieving urinary urgency and frequency. Pharmacodynamics: Mirabelon exerts its pharmacological effect by promoting relaxation of bladder smooth muscle, thereby increasing bladder capacity and relieving urinary urgency. Mirabelon does not appear to have adverse effects on the mean maximum urinary flow rate or mean detrusor pressure at maximum urinary flow rate in patients with lower urinary tract symptoms and bladder outlet obstruction (BOO), but caution should be exercised in patients with BOO due to reports of significant urinary retention. In addition, mirabezone increases blood pressure and heart rate in a dose-dependent manner, and should therefore be used with caution in patients with severe uncontrolled hypertension or other patients who may be at risk of increased blood pressure and heart rate. Mirabelon (YM178) is a novel selective β3-adrenergic receptor agonist for the treatment of overactive bladder[1]. Its mechanism of action is to activate β3-adrenergic receptors in bladder smooth muscle, thereby increasing intracellular cAMP levels, leading to muscle relaxation, increased bladder capacity and improved urination[1]. It is more selective for β3-adrenergic receptors than for β1 and β2 subtypes, thereby minimizing the cardiovascular side effects (e.g., tachycardia, hypertension) associated with non-selective β-adrenergic receptor agonists[1]. Preclinical efficacy in a PBOO-induced overactive bladder model supports its potential for treating bladder dysfunction. Caused by urethral obstruction or idiopathic overactive bladder[1] |
| Molecular Formula |
C21H24N4O2S
|
|
|---|---|---|
| Molecular Weight |
396.51
|
|
| Exact Mass |
396.161
|
|
| Elemental Analysis |
C, 63.61; H, 6.10; N, 14.13; O, 8.07; S, 8.09
|
|
| CAS # |
223673-61-8
|
|
| Related CAS # |
(Rac)-Mirabegron-d5; 1215807-38-7
|
|
| PubChem CID |
9865528
|
|
| Appearance |
White to light yellow solid powder
|
|
| Density |
1.3±0.1 g/cm3
|
|
| Boiling Point |
690.0±55.0 °C at 760 mmHg
|
|
| Melting Point |
138-140°C
|
|
| Flash Point |
371.1±31.5 °C
|
|
| Vapour Pressure |
0.0±2.3 mmHg at 25°C
|
|
| Index of Refraction |
1.681
|
|
| LogP |
1.29
|
|
| Hydrogen Bond Donor Count |
4
|
|
| Hydrogen Bond Acceptor Count |
6
|
|
| Rotatable Bond Count |
9
|
|
| Heavy Atom Count |
28
|
|
| Complexity |
467
|
|
| Defined Atom Stereocenter Count |
1
|
|
| SMILES |
S1C(N([H])[H])=NC(=C1[H])C([H])([H])C(N([H])C1C([H])=C([H])C(=C([H])C=1[H])C([H])([H])C([H])([H])N([H])C([H])([H])[C@@]([H])(C1C([H])=C([H])C([H])=C([H])C=1[H])O[H])=O
|
|
| InChi Key |
PBAPPPCECJKMCM-IBGZPJMESA-N
|
|
| InChi Code |
InChI=1S/C21H24N4O2S/c22-21-25-18(14-28-21)12-20(27)24-17-8-6-15(7-9-17)10-11-23-13-19(26)16-4-2-1-3-5-16/h1-9,14,19,23,26H,10-13H2,(H2,22,25)(H,24,27)/t19-/m0/s1
|
|
| Chemical Name |
2-(2-amino-1,3-thiazol-4-yl)-N-[4-[2-[[(2R)-2-hydroxy-2-phenylethyl]amino]ethyl]phenyl]acetamide
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (5.25 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (5.25 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.08 mg/mL (5.25 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: Mirabegron (10 mg/ml) dissolved in DMSO (50%, vol%) and polyethylene glycol (50%, vol%) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5220 mL | 12.6100 mL | 25.2200 mL | |
| 5 mM | 0.5044 mL | 2.5220 mL | 5.0440 mL | |
| 10 mM | 0.2522 mL | 1.2610 mL | 2.5220 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Study to Test the Long Term Safety and Efficacy of the Beta-3 Agonist Mirabegron (YM178) in Patients With Symptoms of Overactive Bladder
CTID: NCT00688688
Phase: Phase 3 Status: Completed
Date: 2024-11-20
 |
|---|
 |
 |