
| Size | Price | Stock | Qty |
|---|---|---|---|
| 5g |
|
||
| 10g | |||
| 25g | |||
| Other Sizes |
Purity: ≥98%
Levodropropizine [(S)-(-)-Dropropizine; DF 526; DF-526; DF526; Levotuss], the S-isomer of dropropizine, is a histamine receptor inhibitor used as a cough suppressant. It inhibits the histamine receptor and has anti-allergic properties, which lessen coughing.
| Targets |
Histamine receptor
Transient Receptor Potential Vanilloid 1 (TRPV1) [4] Neurokinin 1 (NK1) receptor [4] |
|---|---|
| ln Vitro |
In vitro activity: Levodropropizine binds to alpha-adrenergic and H1-histaminic receptors with affinity.
|
| ln Vivo |
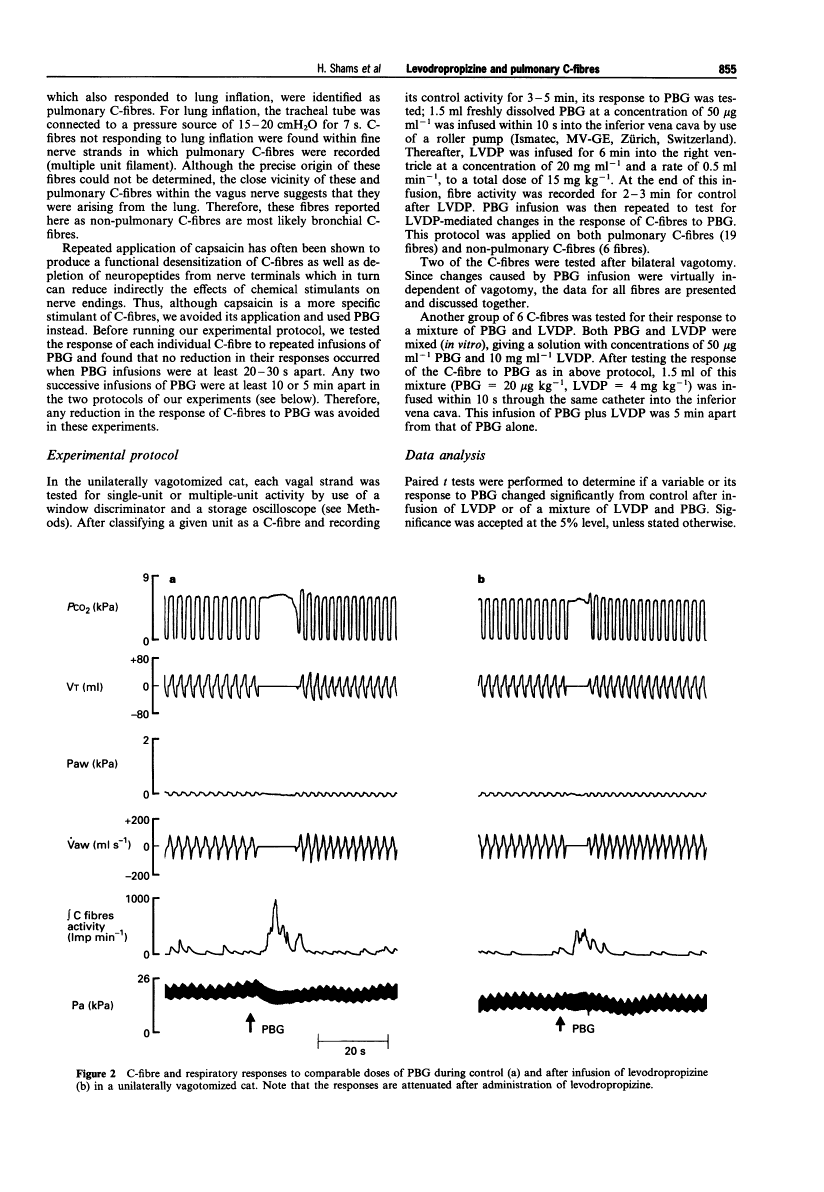
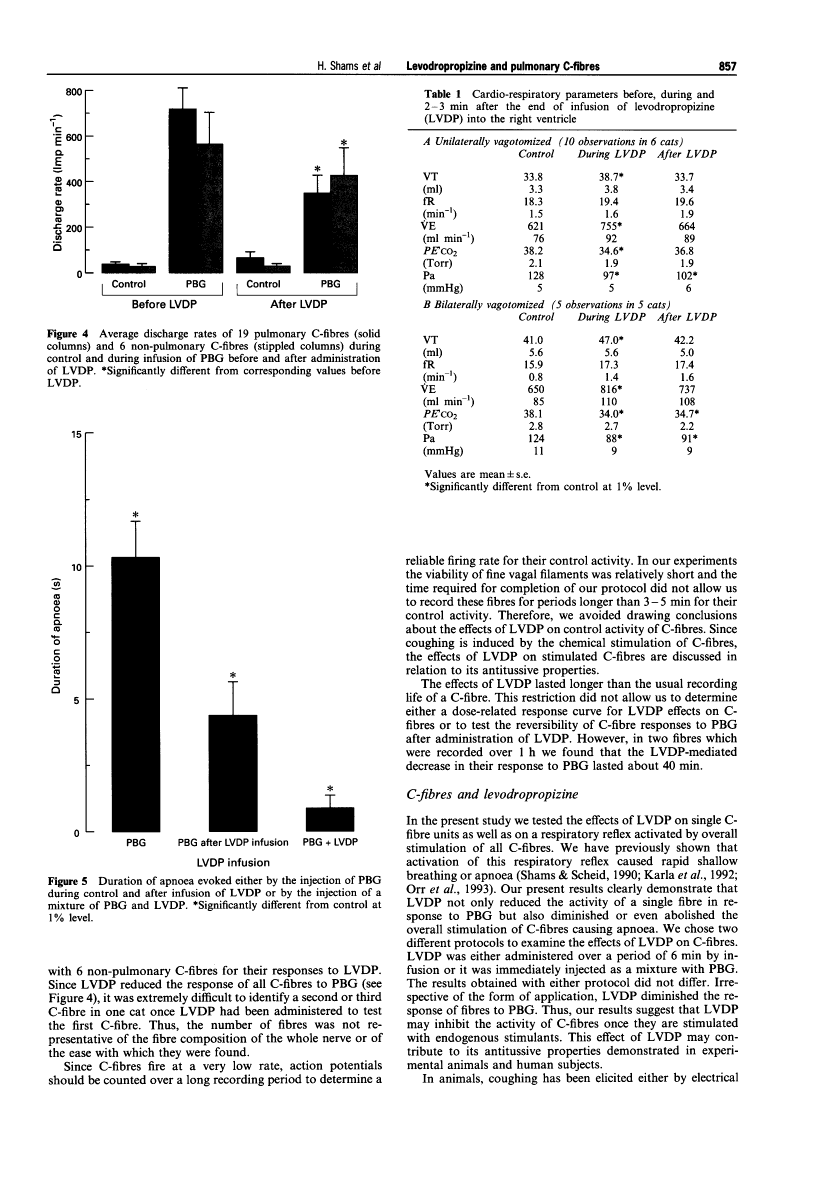
Levodropropizine does not cause physical dependence in rats and has less potent central sedative effects than racemate. Levodropropizine (15 mg/kg, intraperitoneally) decreases the length of apnea as well as the C-fiber'sreactionto phenylbiguanide. In pulmonary fibers, LVDP-induced suppression of the C-fibre response to PBG averages 50%, whereas in non-pulmonary fibers, it is 25%. Anaesthetized guinea pigs and rabbits demonstrate good antitussive activity when levodropropizine is administered. When it comes to inducing coughing in rabbits and guinea pigs, levodropropizine (i.v.) is similar to dropropizine and 1/10 to 1/20 as active as codeine. Orally administered levodropropizine has similar effects to dropropizine and codeine in terms of preventing coughing brought on by irritant aerosols. When electrically induced cough is given to guinea pigs, codeine (5 μg/50 μl i.c.v.) significantly reduces coughing, but levodropropizine (40 μg/50 μL i.c.v.) does not. A peripheral site of action associated with sensory neuropeptides is observed with levodropropizine. In the rat trachea, levodropropizine (10 mg/kg, 50 mg/kg, and 200 mg/kg) decreases the extravasation of Evans blue dye triggered by capsaicin in a dose-dependent manner. Levodropropizine (200 mg/kg) also prevents extravasation triggered by substance P, but it has no effect on extravasation triggered by platelet activating factor.
Meta-analysis of cough treatment: Oral administration of Levodropropizine (DF-526) (30 mg three times daily for adults, 1 mg/kg three times daily for children) reduced cough frequency by 58% in adults and 62% in children with acute/chronic cough, compared to placebo. The antitussive effect lasted for 6-8 hours per dose [1] - Guinea pig chronic cough model induced by cigarette smoke: Intragastric administration of Levodropropizine (DF-526) (10 mg/kg, 20 mg/kg) twice daily for 21 days dose-dependently reduced cough frequency by 45% and 68% respectively. It also inhibited airway neurogenic inflammation, with 52% reduction in substance P levels and 48% decrease in eosinophil infiltration in airway tissues [2] - Pentylenetetrazol (PTZ)-induced epilepsy model in rats: Intraperitoneal injection of Levodropropizine (DF-526) (50 mg/kg, 100 mg/kg) 30 minutes before PTZ administration (60 mg/kg, intraperitoneal) reduced seizure onset latency by 3.2-fold and seizure duration by 55% (100 mg/kg dose). It also increased the threshold of PTZ-induced convulsions [3] - Rat tracheal plasma extravasation model: Intravenous injection of Levodropropizine (DF-526) (1 mg/kg, 3 mg/kg, 10 mg/kg) 15 minutes before capsaicin (10 μg/kg, intravenous) or substance P (5 μg/kg, intravenous) administration dose-dependently reduced plasma extravasation in the trachea. The 10 mg/kg dose inhibited capsaicin-induced extravasation by 72% and substance P-induced extravasation by 65% [4] |
| Animal Protocol |
15 mg/kg, 10 mg/kg, 50 mg/kg and 200 mg/kg; i.v.
Rats Guinea pig chronic cough model: Male Hartley guinea pigs (300-350 g) were exposed to cigarette smoke (10 cigarettes/day, 2 times/day) for 28 days to induce chronic cough. Levodropropizine (DF-526) was dissolved in physiological saline and administered via intragastric gavage (10 mg/kg, 20 mg/kg) twice daily for 21 days (from day 8 to day 28). Record cough frequency within 10 minutes after smoke exposure; collect airway tissues to detect substance P levels and eosinophil infiltration [2] - PTZ-induced epilepsy rat model: Male Sprague-Dawley rats (200-250 g) were randomly divided into vehicle group and Levodropropizine (DF-526) groups (50 mg/kg, 100 mg/kg). The drug was dissolved in physiological saline and administered via intraperitoneal injection 30 minutes before PTZ (60 mg/kg, intraperitoneal) administration. Record seizure onset latency, duration, and severity; calculate convulsion threshold [3] - Rat tracheal plasma extravasation model: Male Wistar rats (180-220 g) were anesthetized with urethane. Levodropropizine (DF-526) was dissolved in physiological saline and administered via intravenous injection (1 mg/kg, 3 mg/kg, 10 mg/kg) 15 minutes before capsaicin (10 μg/kg, intravenous) or substance P (5 μg/kg, intravenous) injection. Evans blue (50 mg/kg, intravenous) was injected 5 minutes before the inducer. Thirty minutes later, rats were euthanized, tracheal tissues were harvested, and Evans blue content was measured to assess plasma extravasation [4] |
| ADME/Pharmacokinetics |
Absorption: The oral bioavailability in the human body is 80-85%; the peak plasma concentration (Cmax) is reached 1-1.5 hours after oral administration (30 mg dose: Cmax = 180 ng/mL) [1]
- Distribution: The volume of distribution (Vd) in the human body is 1.1 L/kg; it is widely distributed in tissues and hardly crosses the blood-brain barrier [1] - Metabolism: It is metabolized very little in the liver (<10% of the dose), and >90% is excreted unchanged [1] - Excretion: 70% of the dose is excreted in the urine (65% as unchanged drug and 5% as metabolites), and 25% is excreted in the feces. The elimination half-life (t1/2) in humans is 6-8 hours [1] - Plasma protein binding rate: The plasma protein binding rate of levopiperazine (DF-526) in human plasma is 15-20% [1] |
| Toxicity/Toxicokinetics |
Acute toxicity: LD50 in rats and mice > 2000 mg/kg (oral); no deaths or serious clinical symptoms (convulsions, respiratory depression) have been reported [1] - Chronic toxicity: No significant hepatotoxicity or hematologic abnormalities were observed in rats after 6 months of oral administration of levopipermine (DF-526) (100 mg/kg/day) [1] - Clinical side effects: Mild headache (2-3% of patients), nausea (1-2%) and diarrhea (1%) have been reported. No sedation or cognitive impairment was observed at therapeutic doses [1] - Drug interactions: No significant interactions were observed with other antitussives, antibiotics or central nervous system drugs [1]
|
| References |
|
| Additional Infomation |
Levopropiperazine belongs to the N-arylpiperazine class of drugs, with the structure N-phenylpiperazine, wherein the amino hydrogen is replaced by a 2,3-dihydroxypropyl group (S-enantiomer). It is a peripheral antitussive and can be used as an alternative to opioids. It has antitussive effects. It is an N-alkylpiperazine, N-arylpiperazine, and secondary alcohol. Levopropiperazine is being investigated in the clinical trial NCT01573663 (Drug Interaction Study of Ambroxol and Levopropiperazine). Levopropiperazine (DF-526) is a non-opioid antitussive with anti-neurogenic inflammatory and anticonvulsant activities [1,2,3,4]. Its core antitussive mechanism is the inhibition of airway neurogenic inflammation, blocking capsaicin and substance P-mediated sensory nerve activation, thereby reducing the cough reflex [2,4].
It exerts its anticonvulsant effect by modulating neuronal excitability in a pentylenetetrazole (PTZ)-induced epilepsy model, but its exact mechanism has not been fully elucidated[3]. Indications include acute and chronic cough in adults and children, with good tolerability and safety. [1] Unlike opioid antitussives, it does not carry the risk of addiction or respiratory depression. [1] It is rapidly absorbed, has a long elimination half-life, and provides sustained efficacy, supporting three-times daily dosing (30 mg each time) for adults and dosing according to weight for children. [1] |
| Molecular Formula |
C13H20N2O2
|
|
|---|---|---|
| Molecular Weight |
236.31
|
|
| Exact Mass |
236.152
|
|
| Elemental Analysis |
C, 66.07; H, 8.53; N, 11.85; O, 13.54
|
|
| CAS # |
99291-25-5
|
|
| Related CAS # |
Dropropizine; 17692-31-8; Levodropropizine-d8
|
|
| PubChem CID |
65859
|
|
| Appearance |
White to off-white solid powder
|
|
| Density |
1.2±0.1 g/cm3
|
|
| Boiling Point |
412.7±34.0 °C at 760 mmHg
|
|
| Melting Point |
102-106ºC
|
|
| Flash Point |
220.9±24.3 °C
|
|
| Vapour Pressure |
0.0±1.0 mmHg at 25°C
|
|
| Index of Refraction |
1.577
|
|
| LogP |
0.46
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
4
|
|
| Rotatable Bond Count |
4
|
|
| Heavy Atom Count |
17
|
|
| Complexity |
212
|
|
| Defined Atom Stereocenter Count |
1
|
|
| SMILES |
O([H])[C@]([H])(C([H])([H])O[H])C([H])([H])N1C([H])([H])C([H])([H])N(C2C([H])=C([H])C([H])=C([H])C=2[H])C([H])([H])C1([H])[H]
|
|
| InChi Key |
PTVWPYVOOKLBCG-ZDUSSCGKSA-N
|
|
| InChi Code |
InChI=1S/C13H20N2O2/c16-11-13(17)10-14-6-8-15(9-7-14)12-4-2-1-3-5-12/h1-5,13,16-17H,6-11H2/t13-/m0/s1
|
|
| Chemical Name |
(2S)-3-(4-phenylpiperazin-1-yl)propane-1,2-diol
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.03.00
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (10.58 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (10.58 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (10.58 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 50 mg/mL (211.59 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.2317 mL | 21.1586 mL | 42.3173 mL | |
| 5 mM | 0.8463 mL | 4.2317 mL | 8.4635 mL | |
| 10 mM | 0.4232 mL | 2.1159 mL | 4.2317 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT01573663 | Completed | Drug: Ambroxol Drug: Levodropropizine |
Healthy | Hanmi Pharmaceutical Company Limited |
February 2012 | Phase 1 |
| NCT01416480 | Completed | Drug: Theobromine 300mg Drug: Levodropropizine 10mg |
Acute Bronchitis | Ahn-Gook Pharmaceuticals Co.,Ltd | May 2010 | Phase 3 |
 |
|---|
 |
 |