
| Size | Price | Stock | Qty |
|---|---|---|---|
| 2mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg | |||
| Other Sizes |
Purity: ≥98%
LCL161 (LCL-161) is a novel and potent SMAC (second mitochondrial-derived activator of caspases) with potential anticancer activity. It works by tightly binding to and inhibiting a variety of IAP (inhibitor of apoptosis) proteins, including XIAP and c-IAP. With IC50 values of 35 and 0.4 nM, respectively, it inhibits cIAP1 in MDA-MB-231 cells and XIAP in HEK293 cells. With IC50 values of 10 and 19 μM, respectively, LCL161 demonstrated anti-proliferative activities in two human hepatocellular carcinoma (HCC) cells, Hep3B and PLC5.
| Targets |
XIAP (IC50 = 35 nM); cIAP1 (IC50 = 0.40 nM)
The target of LCL161 is the Inhibitor of Apoptosis Proteins (IAPs) family, a potent oral Smac mimetic that selectively binds to and antagonizes cIAP1 and XIAP (dual cIAP1/XIAP antagonist), with weak or no activity against other IAPs or non-IAP proteins. - For human cIAP1 BIR3 domain (HTRF binding assay): IC₅₀ = 1.2 nM [2] - For human XIAP BIR3 domain (HTRF binding assay): IC₅₀ = 22 nM [2] - For human cIAP2 BIR3 domain: IC₅₀ = 350 nM (weak affinity) [2] - For non-IAP proteins (e.g., Bcl-2, Mcl-1, survivin, caspases): Ki > 1000 nM (no significant binding) [2] - For cIAP1 degradation in HepG2 hepatocellular carcinoma cells (cell-based assay): EC₅₀ = 5 nM [2] |
|---|---|
| ln Vitro |
LCL161 has a strong affinity for inhibitors of apoptosis proteins (IAPs) and starts the destruction of cIAP1 and cIAP2 to further induce apoptosis by activating caspase. When given alone, LCL161 only weakly inhibits the growth of FLT3-ITD-expressing cells, with an IC50 ranging from 0.5 μM (Ba/F3-FLT3-ITD cells) to 4 μM (MOLM13-luc+ cells). When tested against Ba/F3-D835Y cells, LCL161's potency against the D835Y mutant is found to be significantly higher, with an IC50 of less than 50 nM. LCL161 and PKC412 treatment of MOLM13-luc+ cells significantly increases cell death compared to treatment with either agent alone, with Calcusyn combination indices indicating synergy. For MOLM13-luc+ cells, PKC412 and LCL161 cause apoptosis. In comparison to either agent alone, the combination of PKC412 and LCL161 increases the induction of apoptosis. By working well with PKC412 in combination, LCL161 can prevent stromal-mediated rescue of mutant FLT3-expressing cells. A 100 nM IC50 value for LCL161 prevents the growth of Ba/F3.p210 cells. Imatinib, an ABL inhibitor, and LCL161 work together synergistically to kill cells that express the BCR-ABL gene. Drug-resistant cells that express point mutations in the target proteins have also been shown to be active against LCL161. Ba/F3-derived cell lines that express FLT3-ITD and carry point mutations in the ATP-binding pocket of FLT3 can be mostly or completely eliminated by LCL161 at a concentration of 1000 nM. Additionally, LCL161 exhibits activity against Ba/F3 cells expressing various imatinib- and nilotinib-resistant BCR-ABL point mutations at concentrations ranging from 100 to 1000 nM.[1] Using 96 hours, LCL161 is compared to the 23 cell lines in the Pediatric Preclinical Testing Program (PPTP) in vitro panel. Only three of the 23 PPTP cell lines tested by LCL161 exhibit 50% growth inhibition at a concentration of 10 μM. Two of the three cell lines are T-cell ALL cell lines (COG-LL-317 and CCRF-CEM) and one is an anaplastic large cell lymphoma cell line (Karpas-299), with CCRF-CEM and Karpas-299 exhibiting the lowest relative IC50 values (0.25 and 1.6 μM, respectively). [2]
On human immune subsets, LCL161 exhibits immunomodulatory properties. With little impact on CD4 and CD8 T-cell survival or proliferation, LCL161-treated T lymphocytes exhibit significantly increased cytokine secretion upon activation. Naive T cell priming in vitro with synthetic peptides is significantly improved by LCL161 treatment of peripheral blood mononuclear cells. The phenotypic maturation of myeloid dendritic cells in response to LCL161 results in a diminished ability to cross-present a tumor antigen-based vaccine. The observed activation of the canonical and non-canonical NF-κB pathways in response to LCL161 and the subsequent upregulation of anti-apoptotic molecules are thought to be the possible mechanisms by which these effects are mediated. [3]
1. Antitumor activity against melanoma stem cells (MSCs): LCL161 (0.1–10 μM) inhibited proliferation of CD133⁺/ABCG2⁺ MSCs (melanoma stem cell population) with GI₅₀ = 0.8 μM, and reduced sphere formation by 80% at 2 μM (sphere formation assay). After 24-hour treatment with 5 μM, apoptotic cells (Annexin V-FITC/PI) increased from 3% (control) to 55%, and western blot showed cleavage of caspase-3/PARP and degradation of cIAP1 (>90% reduction) [1] 2. Efficacy in hepatocellular carcinoma (HCC) cells and synergy with Bcl-2 inhibitors: LCL161 (0.5–10 μM) inhibited viability of HepG2 and Huh7 HCC cells with GI₅₀ = 2.5 μM (HepG2) and 3.2 μM (Huh7). It synergized with the Bcl-2 inhibitor ABT-263 (0.1 μM) in HepG2 cells (combination index CI = 0.4): cell viability reduced by 85% vs. 40% (LCL161 alone) or 35% (ABT-263 alone). The combination enhanced cIAP1 degradation and caspase-9 activation (western blot) [2] 3. Induction of apoptotic signaling in solid tumor cells: LCL161 (1–5 μM) treated A549 (lung cancer) and MCF-7 (breast cancer) cells for 12 hours induced dose-dependent degradation of cIAP1 (70–90% reduction at 5 μM) and upregulation of cleaved caspase-3 (4–6-fold increase). No significant effect on XIAP protein levels was observed (consistent with its lower affinity for XIAP) [2] 4. Minimal activity on normal cells: LCL161 (up to 20 μM) had no significant antiproliferative effect on normal human hepatocytes (L02) or foreskin fibroblasts (NHFF) (cell viability >80% vs. control) [2] |
| ln Vivo |
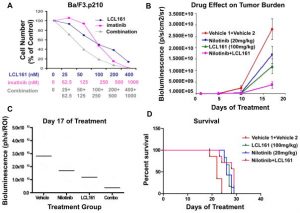
LCL161 significantly improves PKC412's capacity to prevent the development of Ba/F3-FLT3-ITD-luc+ cells in vivo. LCL161 has also been shown to effectively combat FLT3-ITD- and D835Y-expressing cells when combined with the common chemotherapeutic drugs Ara-c and Doxorubicin. Combining nilotinib and lcl161 can suppress leukemia growth in a way that is additive. Nilotinib (100 mg/kg) in high-moderate doses improves the in vivo effects on the burden of leukemia in mice when combined with LCL161 (100 mg/kg).[1] CL161 is evaluated against the in vivo panels of the Pediatric Preclinical Testing Program (PPTP) (30 or 75 mg/kg [solid tumors] or 100 mg/kg [ALL]) when given orally twice per week. About one-third of solid tumor xenografts (glioblastoma and osteosarcoma) show significant differences in EFS distribution in response to LCL161, but not ALL xenografts. There are no detectable objective tumor responses. LCL161 exhibits only modest single agent activity in vivo against the pediatric preclinical models investigated. [2]
1. Antitumor efficacy in melanoma stem cell xenografts: Male athymic nude mice (6–8 weeks old) were subcutaneously injected with 1×10⁶ CD133⁺/ABCG2⁺ MSCs. When tumors reached 100–150 mm³, mice were randomized into 3 groups (n=6/group): vehicle (0.5% methylcellulose), 25 mg/kg LCL161, 50 mg/kg LCL161 (oral gavage, once daily for 28 days). The 50 mg/kg group achieved 88% tumor growth inhibition (TGI); tumor weight was reduced by 82% vs. vehicle. IHC of tumor tissues showed reduced cIAP1 staining (75%) and increased cleaved caspase-3 staining (5-fold) [1] 2. Efficacy in HepG2肝癌 xenografts with Bcl-2 inhibitor: Female nude mice bearing HepG2 xenografts (120–160 mm³) were treated with LCL161 (50 mg/kg, oral, qd) + ABT-263 (10 mg/kg, oral, qd) for 21 days. The combination group showed 92% TGI, significantly higher than LCL161 alone (65% TGI) or ABT-263 alone (60% TGI). No significant increase in toxicity was observed vs. single agents [2] 3. Clinical pharmacodynamic effects (Phase I study): In a Phase I dose-escalation study of LCL161 in patients with advanced solid tumors (n=45), oral administration of 200–1200 mg once weekly showed dose-dependent inhibition of cIAP1 in peripheral blood mononuclear cells (PBMCs) (up to 70% reduction at 800 mg). Stable disease was observed in 12 patients (27%), with a median progression-free survival of 3.2 months [4] |
| Enzyme Assay |
1. HTRF Assay for cIAP1/XIAP BIR3 Binding: The assay was performed in 384-well plates using recombinant human cIAP1 BIR3 (20 nM) or XIAP BIR3 (50 nM) and a biotinylated Smac N-terminal peptide (10 nM, sequence: AVPIAQK-biotin). LCL161 was tested at serial concentrations (0.001–1000 nM) in assay buffer (25 mM HEPES pH 7.4, 150 mM NaCl, 0.05% BSA, 1 mM DTT). After incubation at 37°C for 1 hour, streptavidin-conjugated Eu³⁺ cryptate (10 nM) and anti-cIAP1/XIAP antibody conjugated with XL665 (5 nM) were added. FRET signals were measured at 620 nm (donor) and 665 nm (acceptor). IC₅₀ was defined as the concentration of LCL161 that inhibited 50% of the Smac-IAP BIR3 interaction [2]
2. cIAP1 Ubiquitination Assay: Recombinant human cIAP1 (50 nM) was incubated with LCL161 (0.1–10 μM), E1 (20 nM), E2 (UbcH5b, 100 nM), and ubiquitin (2 μM) in ubiquitination buffer (50 mM Tris-HCl pH 7.5, 10 mM MgCl₂, 2 mM ATP) at 37°C for 2 hours. The reaction was stopped with SDS loading buffer, and ubiquitinated cIAP1 was detected by western blot with anti-cIAP1 antibody. LCL161 induced dose-dependent cIAP1 polyubiquitination (maximal at 5 μM) [2] |
| Cell Assay |
In vitro testing is performed using DIMSCAN.
1. Antiproliferative Assay (GI₅₀ Determination): Cells (CD133⁺/ABCG2⁺ MSCs, HepG2, Huh7) were seeded in 96-well plates at a density of 1000–2000 cells/well and incubated overnight (37°C, 5% CO₂). LCL161 (0.1–10 μM) was added alone or with ABT-263 (0.1 μM), and cells were cultured for 72 hours. Cell viability was measured using the CellTiter-Glo Luminescent Assay (luminescence intensity proportional to ATP). GI₅₀ was calculated as the concentration inhibiting 50% cell growth vs. vehicle [1, 2] 2. Western Blot for IAP Degradation and Apoptosis Markers: Cells were seeded in 6-well plates (5×10⁵ cells/well) and treated with LCL161 (0.5–10 μM) for 4–24 hours. Cells were lysed in RIPA buffer containing protease/phosphatase inhibitors; lysates were separated by 12% SDS-PAGE and transferred to PVDF membranes. Membranes were blocked with 5% BSA, incubated overnight at 4°C with primary antibodies (cIAP1, XIAP, cleaved caspase-3, cleaved PARP, caspase-9, β-actin), then with HRP-conjugated secondary antibodies. Bands were visualized via ECL chemiluminescence [1, 2] 3. Apoptosis Detection by Flow Cytometry: CD133⁺/ABCG2⁺ MSCs or HepG2 cells were treated with LCL161 (1–5 μM) for 24 hours. Cells were harvested, washed with cold PBS, and stained with Annexin V-FITC and propidium iodide (PI) for 15 minutes at room temperature (dark). Stained cells were analyzed via flow cytometry; apoptotic cells were categorized as Annexin V-positive/PI-negative (early apoptosis) or Annexin V-positive/PI-positive (late apoptosis) [1, 2] 4. Melanoma Stem Cell Sphere Formation Assay: CD133⁺/ABCG2⁺ MSCs (1×10³ cells/well) were seeded in ultra-low attachment 96-well plates with stem cell medium (DMEM/F12 + 20 ng/mL EGF/bFGF + 1% B27). LCL161 (0.1–10 μM) was added, and spheres (>50 μm) were counted after 7 days. Sphere inhibition rate = [(vehicle spheres - treated spheres)/vehicle spheres] × 100% [1] |
| Animal Protocol |
Mice: Male NCr athymic nude mice (5-7 weeks of age) are used. Each mouse is inoculated s.c. in the dorsal flank with 1×106 Huh-7 cells suspended in 0.1 mL of serum-free medium containing 50% Matrigel. When tumors grow to 200–300 mm3, mice are given LCL161 (50 mg/kg) or SC-2001 (10 mg/kg) or both once daily via oral administration. Vehicle is received by controls. Weekly caliper measurements of tumors are performed, and the volume of each tumor is determined using the following formula: width2×length×0.52. LCL161 is a first-in-class oral Smac mimetic that has been shown to cause the cleavage of caspase 3 and the degradation of cIAP1 in mouse xenograft models.
Rats: LCL161 is given orally once weekly over the course of 21 days, with a starting dose of 10 mg (which is equal to one-tenth of the dose that caused severe toxicity in 10% of rats). Once-weekly and twice-daily LCL161 dosing are equally effective in the MDA-MB-231 triple-negative breast cancer xenograft model. Better tolerated with less weight loss is once per week. 1. Melanoma Stem Cell Xenograft Model: Male athymic nude mice (6–8 weeks old, 18–22 g) were acclimated to the laboratory (12 h light/dark cycle, 22±2°C) for 7 days. CD133⁺/ABCG2⁺ MSCs (1×10⁶ cells in 0.2 mL PBS/matrigel 1:1) were subcutaneously injected into the right flank. When tumors reached 100–150 mm³ (≈14 days post-injection), mice were randomized into 3 groups (n=6/group). LCL161 was formulated in 0.5% methylcellulose (w/v) in deionized water, doses 25 mg/kg and 50 mg/kg, administered via oral gavage once daily for 28 days. The vehicle group received the same volume of 0.5% methylcellulose. Tumor volume (V = length×width²/2) and body weight were measured twice weekly. At study end, mice were euthanized; tumors were excised for IHC (cIAP1, cleaved caspase-3) [1] 2. HepG2 Hepatocellular Carcinoma Combination Model: Female nude mice were injected subcutaneously with 5×10⁶ HepG2 cells (PBS/matrigel 1:1). When tumors reached 120–160 mm³, mice were divided into 4 groups (n=6/group): vehicle, LCL161 (50 mg/kg, oral, qd), ABT-263 (10 mg/kg, oral, qd), and combination. Treatment lasted 21 days. ABT-263 was formulated in 0.5% methylcellulose. Tumor volume and body weight were monitored as described above; at study end, tumors were weighed to calculate TGI [2] |
| ADME/Pharmacokinetics |
1. Oral bioavailability and human pharmacokinetics (Phase I study): In healthy subjects (n=12), a single oral dose of LCL161 (100–1200 mg) demonstrated dose-proportional pharmacokinetic characteristics. Key parameters: oral bioavailability (F) = 32% (compared to intravenous microdose), Cmax = 8.5 μM (1200 mg), Tmax = 3 hours, terminal half-life (t₁/₂) = 28 hours. Steady-state concentrations were reached on day 15 (once weekly) [3, 4]
2. Plasma protein binding: Human plasma (500 μL) was mixed with LCL161 (0.1–10 μM) and dialyzed at 37°C for 4 hours using a dialysis membrane with a molecular weight cutoff of 12–14 kDa. The concentration of free drug in dialysate was determined by liquid chromatography-tandem mass spectrometry (LC-MS/MS). The plasma protein binding rate was 98.2% [3] 3. Metabolism and CYP interaction: LCL161 is mainly metabolized by CYP3A4 in human liver microsomes (HLM). In vitro experiments: It has a time-dependent inhibitory effect on CYP3A4 (TDI) (IC₅₀ = 1.8 μM) and can induce the expression of CYP3A4 mRNA in primary human hepatocytes (2-fold increase at 10 μM). In healthy subjects, co-administration of LCL161 (800 mg) with midazolam (a CYP3A4 probe) increased the AUC of midazolam by 2.3 times (indicating CYP3A4 inhibition in vivo) [3] 4. Tissue distribution in mice: Mice were orally administered LCL161 (50 mg/kg) and sacrificed 3 hours later (Tmax). Tissues (liver, spleen, lung, tumor, brain) were homogenized in PBS; drug concentrations were determined by LC-MS/MS. Highest concentrations: liver (22.5 μM), spleen (18.3 μM); tumor (6.8 μM, tumor/plasma ratio = 1.2); brain (0.5 μM, brain/plasma ratio = 0.08) [2] |
| Toxicity/Toxicokinetics |
1. Clinical toxicity (Phase I study): In 45 patients with advanced solid tumors treated with LCL161 (once weekly, 200-1200 mg), the most common treatment-related adverse events (TRAEs) were fatigue (47%), nausea (33%), diarrhea (29%), and vomiting (22%). Dose-limiting toxicities (DLTs) occurred in the 1200 mg dose group: grade 3 diarrhea (n=2) and grade 3 fatigue (n=1). No grade 4 TRAEs or treatment-related deaths were reported. [4] 2. Preclinical toxicity in mice: Mice treated with LCL161 (50-100 mg/kg, orally, once daily for 28 days) did not show significant weight loss (<5%) or histopathological lesions in the liver, kidneys, spleen, or lungs. Serum ALT/AST, BUN, and creatinine were all within the normal range; complete blood count (CBC) showed no bone marrow suppression (white blood cell, red blood cell, and platelet counts were unchanged compared to the control group) [2]
3. Drug interaction risk: Because LCL161 has CYP3A4 inhibitory/inducing effects, there is a potential risk of interaction with CYP3A4 substrates (e.g., midazolam, cyclosporine). It is not recommended to use potent CYP3A4 inhibitors (e.g., ketoconazole) or inducers (e.g., rifampin) simultaneously, as they may increase or decrease the concentration of LCL161, respectively [3] |
| References | |
| Additional Infomation |
LCL161 is a small-molecule IAP inhibitor with potent antitumor activity against various solid tumors. In hepatocellular carcinoma (HCC), the efficacy of LCL161 treatment has been shown to be correlated with Bcl-2 expression. It acts as both an antitumor drug and an apoptosis inducer. LCL161 is an aromatic ketone compound belonging to the monofluorobenzene, N-acylpyrrolidine, 1,3-thiazole, and L-alanine derivative classes. LCL161 has been used in clinical trials for the treatment of various cancers, including leukemia, tumors, solid tumors, breast cancer, and ovarian cancer. LCL161 is a highly bioavailable SMAC mimic and an inhibitor of the IAP (inhibitor of apoptosis protein) protein family, possessing potential antitumor activity. The SMAC mimic LCL161 can bind to IAPs, such as X-linked IAP (XIAP) and cellular IAP 1 and 2. Since IAPs protect cancer cells from the apoptosis process, this drug may restore and promote apoptosis induction by activating apoptosis signaling pathways in cancer cells. Many cancer cell types overexpress IAPs, which inhibit apoptosis by binding to and inhibiting active caspases -3, -7, and -9, which play important roles in apoptosis (programmed cell death), necrosis, and inflammation. 1. Background: LCL161 is a first-in-class oral dual cIAP1/XIAP antagonist (Smac mimic) for the treatment of advanced solid tumors. Unlike intravenously administered Smac mimics (e.g., GDC-0152), its oral bioavailability allows for convenient outpatient administration, a key advantage for the treatment of chronic cancers [3, 4]. 2. Mechanism of Action: LCL161 binds to the BIR3 domain of both cIAP1 (high affinity) and XIAP (intermediate affinity). It induces autoubiquitination and proteasome degradation of cIAP1, releasing TRAF2 to activate the non-canonical NF-κB pathway; it can also displace caspase from XIAP, relieving caspase inhibition and activating apoptosis signaling pathway. Its dual activity enhances the efficacy against tumors with heterogeneous IAP expression [2]
3. Clinical development: LCL161 has completed a phase I clinical trial (NCT00979123) for patients with advanced solid tumors, showing good safety and preliminary efficacy (27% of patients had stable disease) [4]. Subsequently, researchers further evaluated its combination with other anticancer drugs (e.g., Bcl-2 inhibitors, chemotherapy drugs) to enhance efficacy, but no phase II/III clinical trial results were reported in the included literature [2, 4] 4. Therapeutic advantages: LCL161 can selectively target cancer cells and cancer stem cells (e.g., melanoma stem cells) without significant toxicity to normal cells. Its synergistic effect with Bcl-2 inhibitors can address the problem of intrinsic apoptosis resistance, a common mechanism of treatment failure in solid tumors [1, 2]. 5. Limitations: Major limitations include CYP3A4-mediated drug interactions (requiring dose adjustments or avoidance of certain drugs) and limited monotherapy efficacy in Phase I clinical trials (most patients achieved stable disease rather than objective response). Future research should focus on combination therapies to improve clinical benefit [3, 4]. |
| Molecular Formula |
C26H33FN4O3S
|
|
|---|---|---|
| Molecular Weight |
500.63
|
|
| Exact Mass |
500.225
|
|
| Elemental Analysis |
C, 62.38; H, 6.64; F, 3.79; N, 11.19; O, 9.59; S, 6.40
|
|
| CAS # |
1005342-46-0
|
|
| Related CAS # |
|
|
| PubChem CID |
24737642
|
|
| Appearance |
White to yellow solid powder
|
|
| Density |
1.2±0.1 g/cm3
|
|
| Boiling Point |
713.7±60.0 °C at 760 mmHg
|
|
| Flash Point |
385.4±32.9 °C
|
|
| Vapour Pressure |
0.0±2.3 mmHg at 25°C
|
|
| Index of Refraction |
1.577
|
|
| LogP |
3.78
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
7
|
|
| Rotatable Bond Count |
8
|
|
| Heavy Atom Count |
35
|
|
| Complexity |
757
|
|
| Defined Atom Stereocenter Count |
3
|
|
| SMILES |
S1C([H])=C(C(C2C([H])=C([H])C(=C([H])C=2[H])F)=O)N=C1[C@]1([H])C([H])([H])C([H])([H])C([H])([H])N1C([C@]([H])(C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H])N([H])C([C@]([H])(C([H])([H])[H])N([H])C([H])([H])[H])=O)=O
|
|
| InChi Key |
UFPFGVNKHCLJJO-SSKFGXFMSA-N
|
|
| InChi Code |
InChI=1S/C26H33FN4O3S/c1-16(28-2)24(33)30-22(17-7-4-3-5-8-17)26(34)31-14-6-9-21(31)25-29-20(15-35-25)23(32)18-10-12-19(27)13-11-18/h10-13,15-17,21-22,28H,3-9,14H2,1-2H3,(H,30,33)/t16-,21-,22-/m0/s1
|
|
| Chemical Name |
(2S)-N-[(1S)-1-cyclohexyl-2-[(2S)-2-[4-(4-fluorobenzoyl)-1,3-thiazol-2-yl]pyrrolidin-1-yl]-2-oxoethyl]-2-(methylamino)propanamide
|
|
| Synonyms |
LCL161; LCL 161; LCL-161
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (4.99 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.99 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (4.99 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9975 mL | 9.9874 mL | 19.9748 mL | |
| 5 mM | 0.3995 mL | 1.9975 mL | 3.9950 mL | |
| 10 mM | 0.1997 mL | 0.9987 mL | 1.9975 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT01968915 | Completed | Drug: LCL161 Drug: Paclitaxel |
Neoplasms | Novartis Pharmaceuticals | November 2013 | Phase 1 |
| NCT01617668 | Completed | Drug: LCL161 Drug: Paclitaxel |
Breast Cancer | Novartis Pharmaceuticals | August 2012 | Phase 2 |
| NCT01240655 | Completed | Drug: LCL161 Drug: Paclitaxel |
Solid Tumors | Novartis Pharmaceuticals | April 2011 | Phase 1 |
| NCT01098838 | Completed | Drug: LCL161 | Advanced Solid Tumors | Novartis Pharmaceuticals | April 2011 | Phase 1 |
| NCT03111992 | Completed | Drug: LCL161 Drug: CJM112 |
Multiple Myeloma | Novartis Pharmaceuticals | December 18, 2017 | Phase 1 |
|
|---|
|
 |